Abstract
Ribosomal proteins (RPs) play pivotal roles in developmental regulation. The loss or mutation of ribosomal protein L11 (RPL11) induces various developmental defects. However, few RPs have been functionally characterized in Apis cerana cerana. In this study, we isolated a single copy gene, AccRPL11, and characterized its connection to brain maturation. AccRPL11 expression was highly concentrated in the adult brain and was significantly induced by abiotic stresses such as pesticides and heavy metals. Immunofluorescence assays demonstrated that AccRPL11 was localized to the medulla, lobula and surrounding tissues of esophagus in the brain. The post-transcriptional knockdown of AccRPL11 gene expression resulted in a severe decrease in adult brain than in other tissues. The expression levels of other brain development-related genes, p38, ERK2, CacyBP and CREB, were also reduced. Immunofluorescence signal attenuation was also observed in AccRPL11-rich regions of the brain in dsAccRPL11-injected honeybees. Taken together, these results suggest that AccRPL11 may be functional in brain maturation in honeybee adults.





Similar content being viewed by others
References
Ament SA, Corona M, Pollock HS, Robinson GE (2008) Insulin signaling is involved in the regulation of worker division of labor in honey bee colonies. Proc Natl Acad Sci USA 105:4226–4231. doi:10.1073/pnas.0800630105
Barakat A, Szick-Miranda K, Chang IF, Guyot R, Blanc G, Cooke R, Delseny M, Bailey-Serres J (2001) The organization of cytoplasmic ribosomal protein genes in the Arabidopsis genome. Plant Physiol 127:398–415. doi:10.1104/pp. 010265
Bhat KP, Itahana K, Jin A, Zhang Y (2004) Essential role of ribosomal protein L11 in mediating growth inhibition-induced p53 activation. EMBO J 23:2402–2412. doi:10.1038/sj.emboj.7600247
Bouakaz L, Bouakaz E, Murgola EJ, Ehrenberg M, Sanyal S (2006) The role of ribosomal protein L11 in class I release factor-mediated translation termination and translational accuracy. J Biol Chem 281:4548–4556. doi:10.1074/jbc.MS10433200
Brandt R, Rohlfing T, Rybak J, Krofczik S, Maye A, Westerhoff M, Hege H, Menzel R (2005) Three-dimensional average-shape atlas of the honeybee brain and its applications. J Comp Neurol 492:1–19. doi:10.1002/cne.20644
Byrne ME (2009) A role for the ribosome in development. Trends Plant Sci 14:512–519. doi:10.1018/j.tplants.2009.08.009
Chakraborty A, Uechi T, Higa S, Torihara H, Kenmochi N (2009) Loss of ribosomal protein L11 affects zebrafish embryonic development through a p53-dependent apoptotic response. PLoS One 4:e4152. doi:10.1371/journal.pone.0004152
Danilova N, Sakamoto KM, Lin S (2011) Ribosomal protein L11 mutation in zebrafish leads to haematopoietic and metabolic defects. Br J Haematol 152:217–228. doi:10.1111/j.1365-2141.2010.08396.x
Duncan KA, Jimenez P, Carruth LL (2009) The selective estrogen receptor-alpha co activator, RPL7, and sexual differentiation of the songbird brain. Psychoneuroendocrinol Suppl 1:S30–S38. doi:10.1016/j.psyneuen.2009.04.023
Dyer FC (2002) The biology of the dance language. Annu Rev Entomol 47:917–949. doi:10.1146/annurev.ento.47.091201.145306
Dyer AG, Paulk AC, Reser DH (2011) Color processing in complex environments: insights from the visual system of bees. Proc Biol Sci 278:952–959. doi:10.1098/rspb.2010.2412
Elias-Neto M, Soares MP, Simões ZL, Hartfelder K, Beyond MM (2010) Developmental characterization, function and regulation of a Laccase2 encoding gene in the honey bee, Apis mellifera (Hymenoptera, Paine). Insect Biochem Mol Biol 40:241–251. doi:10.1016/j.ibmb.2010.02.004
Franco R, Sánchez-Olea R, Reyes-Reyes EM, Panayiotidis MI (2009) Environmental toxicity, oxidative stress and apoptosis: ménage à trios. Mutat Res 674:3–22. doi:10.1016/j.mrgentox.2008.11.012
Guo SW, Liu MG, Long YL, Ren LY, Lu ZM, Yu HY, Hou JF, Li H, Gao CY, Cui XY, An YY, Li J, Zhao LF, Chen J (2007) Region- or state-related differences in expression and activation of extracellular signal-regulated kinases (ERKs) in naïve and pain-experiencing rats. BMC Neurosci 8:53. doi:10.1186/1471-2202-8-53
Jeffrey PL, Capes-Davis A, Dunn JM, Tolhurst O, Seeto G, Hannan AJ, Lin SL (2000) CROC-4: a novel brain specific transcriptional activator of c-foes expressed from proliferation through to maturation of multiple neuronal cell types. Mol Cell Neurosci 16:185–196. doi:10.1006/mcne.2000.0866
Kenmochi N (2003) Ribosome’s and ribosomal proteins. In: Cooper D, ed. Nature encyclopedia of the human genome 5:77-82
Lindström MS (2009) Emerging functions of ribosomal proteins in gene-specific transcription and translation. Biochem Biophys Res Commun 379:167–170. doi:10.1016/j.bbrc.2008.12.083
Mantamadiotis T, Lemberger T, Bleckmann SC, Kern H, Kretz O, Martin Villalba A, Tronche F, Kellendonk C, Gau D, Kapfhammer J, Otto C, Schmid W, Schütz G (2002) Disruption of CREB function in brain leads to neurodegeneration. Nat Genet 31:47–54. doi:10.1038/ng882
Marco Antonio DS, Guidugli-Lazzarini KR, do Nascimento AM, Simões ZL, Hartfelder K (2008) RNAi-mediated silencing of vitellogenin gene function turns honeybee (Apis mellifera) workers into extremely precocious foragers. Naturwissenschaften 95:953–961. doi:10.1007/s00114-008-0413-9
Martínez-Guitarte JL, Planelló R, Morcillo G (2007) Characterization and expression during development and under environmental stress of the genes encoding ribosomal proteins L11 and L13 in Chironomus riparius. Comp Biochem Physiol B Biochem Mol Biol 147:590–596. doi:10.1016/j.cbpb.2007.03.015
Marygold SJ, Coelho CM, Leevers SJ (2005) Genetic analysis of RpL38 and RpL5, two minute genes located in the centric heterochromatin of chromosome 2 of Drosophila melanogaster. Genetics 169:683–695. doi:10.1534/genetics.104.034124
Meng F, Zhang L, Kang M, Guo X, Xu B (2010) Molecular characterization, immunohistochemical localization and expression of a ribosomal protein L17 gene from Apis cerana cerana. Arch Insect Biochem Physiol 75:121–138. doi:10.1002/arch.20386
Murayama E, Kissa K, Zapata A, Mordelet E, Briolat V, Lin HF, Handin RI, Herbomel P (2006) Tracing hematopoietic precursor migration to successive hematopoietic organs during zebrafish development. Immunity 25:963–975. doi:10.1016/j.immuni.2006.10.015
Nelson CM, Ihle KE, Fondrk MK, Page RE, Amdam GV (2007) The gene vitellogenin has multiple coordinating effects on social organization. PLoS Biol 5:e62. doi:10.1371/journal.pbio.0050062
Oliveira CS, Rigon AP, Leal RB, Rossi FM (2008) The activation of ERK1/2 and p38 mitogen-activated protein kinases is dynamically regulated in the developing rat visual system. Int J Dev Neurosci 26:355–362. doi:10.1016/j.ijdevneu.2007.12.007
Oliver ER, Saunders TL, Tarlé SA, Glaser T (2004) Ribosomal protein L24 defect in belly spot and tail (Bst), a mouse Minute. Development 131:3907–3920. doi:10.1242/dev.01268
Page RE Jr, Scheiner R, Erber J, Amdam GV (2006) The development and evolution of division of labor and foraging specialization in a social insect (Apis mellifera L.). Curr Top Dev Biol 74:253–286. doi:10.1016/S0070-2153(06)74008-X
Paulk AC, Gronenberg W (2008) Higher order visual input to the mushroom bodies in the bee, Bombus impatiens. Arthropod Struct Dev 37:443–458. doi:10.1016/j.asd.2008.03.002
Planelló R, Martínez-Guitarte JL, Morcillo G (2007) Ribosomal genes as early targets of cadmium-induced toxicity in Chironomus riparius larvae. Sci Total Environ 373:113–121. doi:10.1016/j.scitotenv.2006.10.038
Quarello P, Garelli E, Carando A, Brusco A, Calabrese R, Dufour C, Longoni D, Misuraca A, Vinti L, Aspesi A, Biondini L, Loreni F, Dianzani I, Ramenghi U (2010) Diamond–Blackfan anemia: genotype–phenotype correlations in Italian patients with RPL5 and RPL11 mutations. Haematologica 95:206–213. doi:10.3324/haematol.2009.011783
Schröter U, Menzel R (2003) A new ascending sensory tract to the calyces of the honeybee mushroom body, the subesophageal-calycal tract. J Comp Neurol 465:168–178. doi:10.1002/cne.10843
Sinakevitch I, Mustard JA, Smith BH (2011) Distribution of the octopamine receptor AmOA1 in the honey bee brain. PLoS One 18:e14536. doi:10.1371/journal.pone.0014536
Tamura K, Dudley J, Nei M, Kumar S (2007) MEGA4: molecular evolutionary genetics analysis (MEGA) software version 4.0. Mol Biol Evol 24:1596–1599. doi:10.1093/molbev/msm092
Vogelstein B, Lane D, Levine AJ (2000) Surfing the p53 network. Nature 408:307–310. doi:10.1038/35042675
Wang M, Kang M, Guo X, Xu B (2010) Identification and characterization of two phospholipid hydroperoxide glutathione peroxidase genes from Apis cerana cerana. Comp Biochem Physiol C Toxicol Pharmacol 152:75–83. doi:10.1016/j.cbpc.2010.03.003
Williams ME, Sussex IM (1995) Developmental regulation of ribosomal protein L16 genes in Arabidopsis thaliana. Plant J 8:65–76. doi:10.1046/j.1365-313X.1995.08010065.x
Withers GS, Fahrbach SE, Robinson GE (1993) Selective neuroanatomical plasticity and division of labour in the honeybee. Nature 15:238–240. doi:10.1038/364238a0
Woyciechowski M, Moroń D (2009) Life expectancy and onset of foraging in the honeybee (Apis mellifera). Insect Soc 56:193–201. doi:10.1007/s00040-009-0012-6
Zhai H, Shi Y, Jin H, Li Y, Lu Y, Chen X, Wang J, Ding L, Wang X, Fan D (2008) Expression of calcyclin-binding protein/Siah-1 interacting protein in normal and malignant human tissues: an immunohistochemical survey. J Histochem Cytochem 56:765–772. doi:10.1369/jhc.2008.950519
Zhang Y, Wolf GW, Bhat K, Jin A, Allio T, Burkhart WA, Xiong Y (2003) Ribosomal protein L11 negatively regulates oncoprotein MDM2 and mediates a p53-dependent ribosomal-stress checkpoint pathway. Mol Cell Biol 23:8902–8912. doi:10.1128/MCB.23.23.8902-8912.2003
Acknowledgments
This work was financially supported by the earmarked fund for China Agriculture Research System (No. CARS-45) and special fund for Agro-scientific Research in the Public Interest (No. 200903006).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Communicated by: Sven Thatje
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(DOC 20 kb)
ESM 2
(DOC 43 kb)
ESM 3
(DOC 35 kb)
Supplementary Fig. 1
Structural and copy number analysis of the genomic sequences of AccRPL11. Intron-exon structure of the RPL11 gene from Mus musculus (MmRPL11, Genbank ID: NC_000070.5), Nasonia vitripennis (NvRPL11, Genbank ID: NW_001820126.1), Apis mellifera (AmRPL11, Genbank ID: NC_007071.3) and A. cerana cerana (AccRPL11, Genbank ID: JN699055). Exons are indicated with white box and introns are shown by lines. UTRs are displayed with black box in this alignment. Lengths of the exons are marked according to the scale above. (JPEG 31 kb)
Supplementary Fig. 2
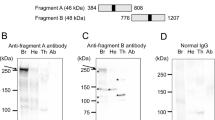
Overexpression and western blot analysis of AccRPL11. (a) Expression and purification of AccRPL11 protein using SDS-PAGE. Lanes 1: and 2: uninduced expression of recombinant AccRPL11 protein; lanes 3 and 4: overexpression of recombinant AccRPL11 protein after IPTG induction; lane 5: purified recombinant AccRPL11 protein and lane M: protein molecular weight marker. AccRPL11 is indicated by an arrowhead. (b) The anti-AccRPL11 specificity was evaluated using immunoblot analysis (left) and ponceau staining (right). The ponceau staining verified that similar amounts of protein were loaded in each lane. Lane 1: induced recombinant AccRPL11 protein; lane 2: purified recombinant AccRPL11 protein; and lane M: pre-stained protein molecular weight marker. (JPEG 12 kb)
Rights and permissions
About this article
Cite this article
Meng, F., Lu, W., Yu, F. et al. Ribosomal protein L11 is related to brain maturation during the adult phase in Apis cerana cerana (Hymenoptera, Apidae). Naturwissenschaften 99, 343–352 (2012). https://doi.org/10.1007/s00114-012-0905-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00114-012-0905-5