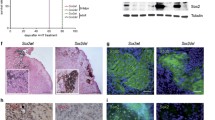
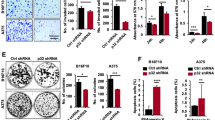
Abstract
Due to the development of resistance to previously effective therapies, there is a constant need for novel treatment modalities for metastatic melanoma. Nischarin (NISCH) is a druggable scaffolding protein reported as a tumor suppressor and a positive prognostic marker in breast and ovarian cancers through regulation of cancer cell survival, motility and invasion. The aim of this study was to examine the expression and potential role of nischarin in melanoma. We found that nischarin expression was decreased in melanoma tissues compared to the uninvolved skin, and this was attributed to the presence of microdeletions and hyper-methylation of the NISCH promoter in the tumor tissue. In addition to the previously reported cytoplasmic and membranous localization, we observed nischarin in the nuclei in melanoma patients’ tissues. NISCH expression in primary melanoma had favorable prognostic value for female patients, but, unexpectedly, high NISCH expression predicted worse prognosis for males. Gene set enrichment analysis suggested significant sex-related disparities in predicted association of NISCH with several signaling pathways, as well as with different tumor immune infiltrate composition in male and female patients. Taken together, our results imply that nischarin may have a role in melanoma progression, but that fine-tuning of the pathways it regulates is sex-dependent.
Key messages
-
Nischarin is a tumor suppressor whose role has not been investigated in melanoma.
-
Nischarin expression was downregulated in melanoma tissue compared to the normal skin.
-
Nischarin had the opposite prognostic value in male and female melanoma patients.
-
Nischarin association with signaling pathways differed in females and males. Our findings challenge the current view of nischarin as a universal tumor suppressor.







Similar content being viewed by others
Availability of data and materials
Abbreviations
- ACTB:
-
Beta actin
- CNA:
-
Copy-number alteration
- DFCI:
-
Dana-Farber Cancer Institute
- FDA:
-
Food and Drug Administration
- FDR:
-
False discovery rate
- FFPE:
-
Formalin-fixed and paraffin-embedded
- GEO:
-
Gene Expression Omnibus
- GSEA:
-
Gene set enrichment analysis
- H&E :
-
Hematoxylin and eosin
- HPA:
-
Human Protein Atlas
- IORS:
-
Institute for Oncology and Radiology of Serbia
- IR1:
-
Imidazoline receptor 1
- IRAS:
-
Imidazoline receptor antisera selected
- LRR:
-
Leucine-rich repeat
- LOH:
-
Loss of heterozygosity
- MSP:
-
Methylation specific PCR
- NCBI:
-
National Center for Biotechnology Information
- NER:
-
Nucleotide excision repair
- NES:
-
Normalized enrichment score
- NISCH:
-
Nischarin
- SKCM:
-
Skin cutaneous melanoma
- TCA:
-
Tricarboxylic acid
- TCGA:
-
The Cancer Genome Atlas
References
Siegel RL, Miller KD, Fuchs HE, Jemal A (2021) Cancer Statistics, 2021. CA Cancer J Clin 71:7–33. https://doi.org/10.3322/caac.21654
Sung H, Ferlay J, Siegel RL et al (2021) Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 71:209–249. https://doi.org/10.3322/caac.21660
Ugurel S, Ro J, Ascierto PA et al (2020) Survival of patients with advanced metastatic melanoma : The impact of MAP kinase pathway inhibition and immune checkpoint inhibition - Update 2019. Eur J Cancer 130:126–138. https://doi.org/10.1016/j.ejca.2020.02.021
Mukherjee N, Schwan JV, Fujita M et al (2015) Alternative Treatments for Melanoma: Targeting BCL-2 Family Members to De-Bulk and Kill Cancer Stem Cells. J Invest Dermatol 135:2155–2161. https://doi.org/10.1038/jid.2015.145
Kandolf Sekulovic L, Peris K, Hauschild A et al (2017) More than 5000 patients with metastatic melanoma in Europe per year do not have access to recommended first-line innovative treatments. Eur J Cancer 75:313–322. https://doi.org/10.1016/j.ejca.2017.01.012
Piletz JE, Ivanov TR, Sharp JD et al (2000) Imidazoline receptor antisera-selected (IRAS) cDNA: Cloning and characterization. DNA Cell Biol 19:319–329. https://doi.org/10.1089/10445490050043290
Alahari SK, Lee JW, Juliano RL (2000) Nischarin, a novel protein that interacts with the integrin α5 subunit and inhibits cell migration. J Cell Biol 151:1141–1154. https://doi.org/10.1083/jcb.151.6.1141
Bousquet P, Hudson A, García-Sevilla JA, Li JX (2020) Imidazoline receptor system: The past, the present, and the future. Pharmacol Rev 72:50–79. https://doi.org/10.1124/pr.118.016311
Uhlén M, Fagerberg L, Hallström BM et al (2015) Proteomics. Tissue-based map of the human proteome. Science 347:1260419. https://doi.org/10.1126/science.1260419
Li J, He X, Dong R et al (2015) Frequent loss of NISCH promotes tumor proliferation and invasion in ovarian cancer via inhibiting the FAK signal pathway. Mol Cancer Ther 14:1202–1212. https://doi.org/10.1158/1535-7163.mct-14-0911
Maziveyi M, Alahari SK (2015) Breast cancer tumor suppressors: A special emphasis on novel protein nischarin. Cancer Res 75:4252–4259. https://doi.org/10.1158/0008-5472.CAN-15-1395
Loginov VI, Bazov IV, Khodyrev DS et al (2008) Human chromosome 3P regions of putative tumor-suppressor genes in renal, breast, and ovarian carcinomas. Russ J Genet 44:209–214. https://doi.org/10.1134/s1022795408020142
Ding Y, Zhang R, Zhang K et al (2013) Nischarin Is Differentially Expressed in Rat Brain and Regulates Neuronal Migration. PLoS ONE 8:3–9. https://doi.org/10.1371/journal.pone.0054563
Baranwal S, Wang Y, Rathinam R et al (2011) Molecular characterization of the tumor-suppressive function of nischarin in breast cancer. J Natl Cancer Inst 103:1513–1528. https://doi.org/10.1093/jnci/djr350
Maziveyi M, Dong S, Baranwal S, Alahari SK (2018) Nischarin regulates focal adhesion and Invadopodia formation in breast cancer cells. Mol Cancer 17:1–11. https://doi.org/10.1186/s12943-018-0764-6
Dong S, Baranwal S, Garcia A et al (2017) Nischarin inhibition alters energy metabolism by activating AMP-activated protein kinase. J Biol Chem 292:16833–16846. https://doi.org/10.1074/jbc.M117.784256
Talantov D, Mazumder A, Yu JX et al (2005) Novel Genes Associated with Malignant Melanoma but not Benign Melanocytic Lesions. Clin Cancer Res 11:7234–7242. https://doi.org/10.1158/1078-0432.CCR-05-0683
Kabbarah O, Nogueira C, Feng B et al (2010) Integrative genome comparison of primary and metastatic melanomas. PLoS One 5:e10770. https://doi.org/10.1371/journal.pone.0010770
Liu D, Schilling B, Liu D et al (2019) Integrative molecular and clinical modeling of clinical outcomes to PD1 blockade in patients with metastatic melanoma. Nat Med 25:1916–1927. https://doi.org/10.1038/s41591-019-0654-5
Wouters J, Vizoso M, Martinez-Cardus A et al (2017) Comprehensive DNA methylation study identifies novel progression-related and prognostic markers for cutaneous melanoma. BMC Med 15:101. https://doi.org/10.1186/s12916-017-0851-3
Özdemir BC, Dotto GP (2019) Sex Hormones and Anticancer Immunity. Clin Cancer Res 25:4603–4610. https://doi.org/10.1158/1078-0432.CCR-19-0137
Ye Y, Jing Y, Li L et al (2020) Sex-associated molecular differences for cancer immunotherapy. Nat Commun 11:1–8. https://doi.org/10.1038/s41467-020-15679-x
Selitsky SR, Mose LE, Smith CC et al (2019) Prognostic value of B cells in cutaneous melanoma. Genome Med 11:1–11. https://doi.org/10.1186/s13073-019-0647-5
Okpechi SC, Yousefi H, Nguyen K et al (2022) Role of Nischarin in the pathology of diseases: a special emphasis on breast cancer. Oncogene 41:1079–1086. https://doi.org/10.1038/s41388-021-02150-4
Botti G, Cerrone M, Scognamiglio G et al (2013) Microenvironment and tumor progression of melanoma: New therapeutic prospectives. J Immunotoxicol 10:235–252. https://doi.org/10.3109/1547691X.2012.723767
Sullivan RJ, Flaherty KT (2013) Resistance to BRAF-targeted therapy in melanoma. Eur J Cancer 49:1297–1304. https://doi.org/10.1016/j.ejca.2012.11.019
Ding Y, Li Y, Lu L et al (2015) Inhibition of Nischarin Expression Promotes Neurite Outgrowth through Regulation of PAK Activity. PLoS One 10:e0144948. https://doi.org/10.1371/journal.pone.0144948
Yaar M, Park HY (2012) Melanocytes: A window into the nervous system. J Invest Dermatol 132:835–845. https://doi.org/10.1038/jid.2011.386
Hoque MO, Kim MS, Ostrow KL et al (2008) Genome-wide promoter analysis uncovers portions of the cancer methylome. Cancer Res 68:2661–2670. https://doi.org/10.1158/0008-5472.CAN-07-5913
La Torre A, Muscarella LA, Parrella P et al (2012) Aberrant genes promoter methylation in neural crest-derived tumors. Int J Biol Markers 27. https://doi.org/10.5301/JBM.2012.9766
Alexandrov LB, Nik-Zainal S, Wedge DC et al (2013) Signatures of mutational processes in human cancer. Nature 500:415–421. https://doi.org/10.1038/nature12477
Gupta S, Artomov M, Goggins W et al (2015) Gender Disparity and Mutation Burden in Metastatic Melanoma. JNCI J Natl Cancer Inst 107:221. https://doi.org/10.1093/JNCI/DJV221
Li CH, Haider S, Shiah YJ et al (2018) Sex Differences in Cancer Driver Genes and Biomarkers. Cancer Res 78:5527–5537. https://doi.org/10.1158/0008-5472.CAN-18-0362
Joosse A, Collette S, Suciu S et al (2012) Superior outcome of women with stage I/II cutaneous melanoma: Pooled analysis of four European organisation for research and treatment of cancer phase III trials. J Clin Oncol 30:2240–2247. https://doi.org/10.1200/JCO.2011.38.0584
Lasithiotakis K, Leiter U, Meier F et al (2008) Age and gender are significant independent predictors of survival in primary cutaneous melanoma. Cancer 112:1795–1804. https://doi.org/10.1002/cncr.23359
de Vries E, Nijsten TEC, Visser O et al (2008) Superior survival of females among 10 538 Dutch melanoma patients is independent of Breslow thickness, histologic type and tumor site. Ann Oncol 19:583–589. https://doi.org/10.1093/annonc/mdm498
Dika E, Patrizi A, Lambertini M (2019) Estrogen Receptors and Melanoma: A Review. Cells 8:1463. https://doi.org/10.3390/CELLS8111463
Dong S, Blüher M, Zhang Y et al (2019) Development of insulin resistance in Nischarin mutant female mice. Int J Obes 43:1046–1057. https://doi.org/10.1038/s41366-018-0241-8
Nguyen TH, Yousefi H, Okpechi SC et al (2022) Nischarin Deletion Reduces Oxidative Metabolism and Overall ATP : A Study Using a Novel NISCH ∆ 5–6 Knockout Mouse Model. Int J Mol Sci 23:1374. https://doi.org/10.3390/IJMS23031374/S1
Ciołczyk-Wierzbicka D, Gil D, Zarzycka M, Laidler P (2020) mTOR inhibitor everolimus reduces invasiveness of melanoma cells. Hum Cell 33:88–97. https://doi.org/10.1007/s13577-019-00270-4
Damsky W, Micevic G, Meeth K et al (2015) mTORC1 activation blocks BrafV600E -induced growth-arrest, but is insufficient for melanoma formation. Cancer Cell 27:41–56. https://doi.org/10.1016/j.ccell.2014.11.014.mTORC1
Lizcano JM, Göransson O, Toth R et al (2004) LKB1 is a master kinase that activates 13 kinases of the AMPK subfamily, including MARK/PAR-1. Embo J 23. https://doi.org/10.1038/sj.emboj.7600110
Jain P, Baranwal S, Dong S et al (2013) Integrin-binding protein nischarin interacts with tumor suppressor liver kinase B1 (LKB1) to regulate cell migration of breast epithelial cells. J Biol Chem 288:15495–15509. https://doi.org/10.1074/jbc.M112.418103
Haupt S, Caramia F, Klein SL et al (2021) Sex disparities matter in cancer development and therapy. Nat Rev Cancer 21:393. https://doi.org/10.1038/S41568-021-00348-Y
Kusakabe M, Onishi Y, Tada H et al (2019) Mechanism and regulation of DNA damage recognition in nucleotide excision repair. Genes Environ 41:1–6. https://doi.org/10.1186/S41021-019-0119-6/FIGURES/3
Ji Y, Hao H, Reynolds K et al (2019) Wnt Signaling in Neural Crest Ontogenesis and Oncogenesis. Cells 8:1173. https://doi.org/10.3390/CELLS8101173
Chien AJ, Moore EC, Lonsdorf AS et al (2009) Activated Wnt/ß-catenin signaling in melanoma is associated with decreased proliferation in patient tumors and a murine melanoma model. Proc Natl Acad Sci USA 106:1193–1198. https://doi.org/10.1073/PNAS.0811902106
Chien AJ, Haydu LE, Biechele TL et al (2014) Targeted BRAF Inhibition Impacts Survival in Melanoma Patients with High Levels of Wnt/β-Catenin Signaling. PLoS One 9:e94748. https://doi.org/10.1371/JOURNAL.PONE.0094748
Conrad WH, Swift RD, Biechele TL et al (2012) Regulating the response to targeted MEK inhibition in melanoma. Cell Cycle 11:3724–3730. https://doi.org/10.4161/CC.21645
Spranger S, Bao R, Gajewski TF (2015) Melanoma-intrinsic β-catenin signalling prevents anti-tumour immunity. Nature 523:231–235. https://doi.org/10.1038/NATURE14404
Li X, Xiang Y, Li F et al (2019) WNT/β-catenin signaling pathway regulating T cell-inflammation in the tumor microenvironment. Front Immunol 10:2293. https://doi.org/10.3389/FIMMU.2019.02293/BIBTEX
Acknowledgements
The results shown here are in part based upon data generated by the TCGA Research Network: https://www.cancer.gov/tcga.
Funding
This study was supported by the Ministry of Education, Science and Technological Development of the Republic of Serbia (Agreement No. 451-03-68/2022-14/200043 and 451-03-68/2022-14/200015). JG is supported by Horizon 2020 MSC grant agreement No. 891135.
Author information
Authors and Affiliations
Contributions
Marija Ostojić, Marko Jevrić, Tatjana Srdić-Rajić and Jelena Grahovac contributed to the study conception and design. Material preparation, data collection and analysis were performed by Marija Ostojić, Marko Jevrić, Olivera Mitrović-Ajtić, Kristina Živić, Miljana Tanić, Milena Čavić, and Jelena Grahovac. Tatjana Srdić-Rajić and Jelena Grahovac secured the acquisition of funds for the study. The first draft of the manuscript was written by Marija Ostojić and Jelena Grahovac, and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Ethics approval
The study was approved by the Ethics Committee of the Institute for Oncology and Radiology of Serbia (Approval No5549-01, from 11.12.2017) and was performed in accordance with the Helsinki Declaration of 1975, as revised in 2013.
Consent to participate
Informed consent was obtained from all individual participants included in the study.
Consent to publish
Not applicable.
Competing interests
The authors have no relevant financial or non-financial interests to disclose.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ostojić, M., Jevrić, M., Mitrović-Ajtić, O. et al. Nischarin expression may have differing roles in male and female melanoma patients. J Mol Med 101, 1001–1014 (2023). https://doi.org/10.1007/s00109-023-02339-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00109-023-02339-y