Abstract
Sorafenib acquired drug resistance during the treatment of hepatocellular carcinoma (HCC) reduces the efficacy of the drug. The immune escape effect induced by PD-L1 is largely associated with drug resistance of HCC. However, the regulated mechanism of PD-L1 is unclear. This research aimed to clarify the control mechanism of PD-L1. c-Met was found abnormally highly expressed in Huh-7SR with high PD-L1 expression. In addition, c-Met, as the upstream target molecule of PD-L1, promoted the proliferation and migration of HCC in vitro and in vivo. We also found that c-Met activated the MAPK signaling pathway and the downstream NF-κBp65 transcription factor, which interacts with the proximal region of the PD-L1 promoter to promote PD-L1 expression. In conclusion, c-Met regulates the transcription of PD-L1 through the MAPK/NF-κBp65 pathway, thereby promoting the progress of HCC. The role of c-Met and PD-L1 in HCC needs to be further studied, but it is a potential target for the treatment of HCC.
Graphical abstract

Key messages
-
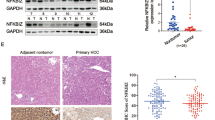
In the study, it was found that c-Met is also abnormally highly expressed in Huh-7SR with high PD-L1 expression and can promote the development of HCC in vitro and in vivo. PD-L1 and c-Met expression levels are positively correlated.
-
In the follow-up mechanism study, we found that c-Met activated the MAPK signaling pathway and subsequently activated the downstream NF-κBp65 transcription factor, which interacts with the proximal region of the PD-L1 promoter to promote PD-L1 expression.
-
Our study found that c-Met regulates the transcription of PD-L1 through the MAPK/NF-κBp65 pathway, thereby promoting the progress of HCC.






Similar content being viewed by others
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Chen Z, Xie H, Hu M, Huang T, Hu Y, Sang N, Zhao Y (2020) Recent progress in treatment of hepatocellular carcinoma. Am J Cancer Res 10:2993–3036
Gentile D, Donadon M, Lleo A, Aghemo A, Roncalli M, di Tommaso L, Torzilli G (2020) Surgical treatment of hepatocholangiocarcinoma: a systematic review. Liver Cancer 9:15–27. https://doi.org/10.1159/000503719
Garten A, Grohmann T, Kluckova K, Lavery GG, Kiess W, Penke M (2019) Sorafenib-induced apoptosis in hepatocellular carcinoma is reversed by SIRT1. Int J Mol Sci 20. https://doi.org/10.3390/ijms20164048.
Zhang Y, Xie C, Li A, Liu X, Xing Y, Shen J, Huo Z, Zhou S, Liu X, Xie Y et al (2019) PKI-587 enhances chemosensitivity of oxaliplatin in hepatocellular carcinoma through suppressing DNA damage repair pathway (NHEJ and HR) and PI3K/AKT/mTOR pathway. Am J Transl Res 11:5134–5149
Xu J, Ji L, Liang Y, Wan Z, Zheng W, Song X, Gorshkov K, Sun Q, Lin H, Zheng X et al (2020) CircRNA-SORE mediates sorafenib resistance in hepatocellular carcinoma by stabilizing YBX1. Signal Transduct Target Ther 5:298. https://doi.org/10.1038/s41392-020-00375-5
Liu X, Xie C, Li A, Zhang Y, Liu X, Zhou S, Shen J, Huo Z, Cao W, Ma Y et al (2019) BEZ235 enhances chemosensitivity of paclitaxel in hepatocellular carcinoma through inhibiting the PI3K/Akt/mTOR pathway. Am J Transl Res 11:7255–7271
Liu J, Liu Y, Meng L, Liu K, Ji B (2017) Targeting the PD-L1/DNMT1 axis in acquired resistance to sorafenib in human hepatocellular carcinoma. Oncol Rep 38:899–907. https://doi.org/10.3892/or.2017.5722
Xu GL, Ni CF, Liang HS, Xu YH, Wang WS, Shen J, Li MM, Zhu XL (2020) Upregulation of PD-L1 expression promotes epithelial-to-mesenchymal transition in sorafenib-resistant hepatocellular carcinoma cells. Gastroenterol Rep (Oxf) 8:390–398. https://doi.org/10.1093/gastro/goaa049
Liang SC, Latchman YE, Buhlmann JE, Tomczak MF, Horwitz BH, Freeman GJ, Sharpe AH (2003) Regulation of PD-1, PD-L1, and PD-L2 expression during normal and autoimmune responses. Eur J Immunol 33:2706–2716. https://doi.org/10.1002/eji.200324228
Wei F, Zhang T, Deng SC, Wei JC, Yang P, Wang Q, Chen ZP, Li WL, Chen HC, Hu H et al (2019) PD-L1 promotes colorectal cancer stem cell expansion by activating HMGA1-dependent signaling pathways. Cancer Lett 450:1–13. https://doi.org/10.1016/j.canlet.2019.02.022
Erber R, Hartmann A (2020) Understanding PD-L1 testing in breast cancer: a practical approach. Breast Care (Basel) 15:481–490. https://doi.org/10.1159/000510812
Cordonnier M, Nardin C, Chanteloup G, Derangere V, Algros MP, Arnould L, Garrido C, Aubin F, Gobbo J (2020) Tracking the evolution of circulating exosomal-PD-L1 to monitor melanoma patients. J Extracell Vesicles 9:1710899. https://doi.org/10.1080/20013078.2019.1710899
Qu QX, Xie F, Huang Q, Zhang XG (2017) Membranous and cytoplasmic expression of PD-L1 in ovarian cancer cells. Cell Physiol Biochem 43:1893–1906. https://doi.org/10.1159/000484109
Wang X, Liu Y (2020) PD-L1 expression in tumor infiltrated lymphocytes predicts survival in triple-negative breast cancer. Pathol Res Pract 216:152802. https://doi.org/10.1016/j.prp.2019.152802
David JM, Dominguez C, McCampbell KK, Gulley JL, Schlom J, Palena C (2017) A novel bifunctional anti-PD-L1/TGF-beta Trap fusion protein (M7824) efficiently reverts mesenchymalization of human lung cancer cells. Oncoimmunology 6:e1349589. https://doi.org/10.1080/2162402X.2017.1349589
Chun HW, Hong R (2019) Significance of PD-L1 clones and C-MET expression in hepatocellular carcinoma. Oncol Lett 17:5487–5498. https://doi.org/10.3892/ol.2019.10222
Peng S, Wang R, Zhang X, Ma Y, Zhong L, Li K, Nishiyama A, Arai S, Yano S, Wang W (2019) EGFR-TKI resistance promotes immune escape in lung cancer via increased PD-L1 expression. Mol Cancer 18:165. https://doi.org/10.1186/s12943-019-1073-4
Wang H, Rao B, Lou J, Li J, Liu Z, Li A, Cui G, Ren Z, Yu Z (2020) The function of the HGF/c-Met axis in hepatocellular carcinoma. Front Cell Dev Biol 8:55. https://doi.org/10.3389/fcell.2020.00055
Marona P, Gorka J, Kotlinowski J, Majka M, Jura J, Miekus K (2019) c-Met as a key factor responsible for sustaining undifferentiated phenotype and therapy resistance in renal carcinomas. Cells 8. https://doi.org/10.3390/cells8030272.
Safaie Qamsari E, Safaei Ghaderi S, Zarei B, Dorostkar R, Bagheri S, Jadidi-Niaragh F, Somi MH, Yousefi M (2017) The c-Met receptor: implication for targeted therapies in colorectal cancer. Tumour Biol 39:1010428317699118. https://doi.org/10.1177/1010428317699118
Mitra S, Bal A, Kashyap D, Kumar S, Shrivastav S, Das A, Singh G (2020) Tumour angiogenesis and c-Met pathway activation — implications in breast cancer. APMIS 128:316–325. https://doi.org/10.1111/apm.13031
Lim L, Wu CC, Hsu YT, Sun FJ, Chang CL (2019) Clinical significance of c-Met and phospho-c-Met (Tyr1234/1235) in ovarian cancer. Taiwan J Obstet Gynecol 58:105–110. https://doi.org/10.1016/j.tjog.2018.11.020
Fan G, Zhang S, Gao Y, Greer PA, Tonks NK (2016) HGF-independent regulation of MET and GAB1 by nonreceptor tyrosine kinase FER potentiates metastasis in ovarian cancer. Genes Dev 30:1542–1557. https://doi.org/10.1101/gad.284166.116
Usatyuk PV, Fu P, Mohan V, Epshtein Y, Jacobson JR, Gomez-Cambronero J, Wary KK, Bindokas V, Dudek SM, Salgia R et al (2014) Role of c-Met/phosphatidylinositol 3-kinase (PI3k)/Akt signaling in hepatocyte growth factor (HGF)-mediated lamellipodia formation, reactive oxygen species (ROS) generation, and motility of lung endothelial cells. J Biol Chem 289:13476–13491. https://doi.org/10.1074/jbc.M113.527556
Carr BI, Wang Z, Wang M, Cavallini A, D’Alessandro R, Refolo MG (2011) c-Met-Akt pathway-mediated enhancement of inhibitory c-Raf phosphorylation is involved in vitamin K1 and sorafenib synergy on HCC growth inhibition. Cancer Biol Ther 12:531–538. https://doi.org/10.4161/cbt.12.6.16053
Qian L, Su H, Wang G, Li B, Shen G, Gao Q (2020) Anti-tumor activity of bufalin by inhibiting c-MET mediated MEK/ERK and PI3K/AKT signaling pathways in gallbladder cancer. J Cancer 11:3114–3123. https://doi.org/10.7150/jca.38393
Santoro A, Simonelli M, Rodriguez-Lope C, Zucali P, Camacho LH, Granito A, Senzer N, Rimassa L, Abbadessa G, Schwartz B et al (2013) A Phase-1b study of tivantinib (ARQ 197) in adult patients with hepatocellular carcinoma and cirrhosis. Br J Cancer 108:21–24. https://doi.org/10.1038/bjc.2012.556
Deng S, Solinas A, Calvisi DF (2021) Cabozantinib for HCC treatment, from clinical back to experimental models. Front Oncol 11:756672. https://doi.org/10.3389/fonc.2021.756672
Chen W, Wu S, Huang Y, Zhang T, Dong H, Zheng X, Chen T, Gong X, Liu G, Zhao X (2021) A c-Met inhibitor suppresses osteosarcoma progression via the ERK1/2 pathway in human osteosarcoma cells. Onco Targets Ther 14:4791–4804. https://doi.org/10.2147/OTT.S317122
Jiang X, Wang J, Deng X, Xiong F, Ge J, Xiang B, Wu X, Ma J, Zhou M, Li X et al (2019) Role of the tumor microenvironment in PD-L1/PD-1-mediated tumor immune escape. Mol Cancer 18:10. https://doi.org/10.1186/s12943-018-0928-4
Hamanishi J, Mandai M, Matsumura N, Abiko K, Baba T, Konishi I (2016) PD-1/PD-L1 blockade in cancer treatment: perspectives and issues. Int J Clin Oncol 21:462–473. https://doi.org/10.1007/s10147-016-0959-z
Dermani FK, Samadi P, Rahmani G, Kohlan AK, Najafi R (2019) PD-1/PD-L1 immune checkpoint: potential target for cancer therapy. J Cell Physiol 234:1313–1325. https://doi.org/10.1002/jcp.27172
Ma Y, Xu R, Liu X, Zhang Y, Song L, Cai S, Zhou S, Xie Y, Li A, Cao W et al (2021) LY3214996 relieves acquired resistance to sorafenib in hepatocellular carcinoma cells. Int J Med Sci 18:1456–1464. https://doi.org/10.7150/ijms.51256
Li D, Sun FF, Wang D, Wang T, Peng JJ, Feng JQ, Li H, Wang C, Zhou DJ, Luo H et al (2020) Programmed death ligand-1 (PD-L1) regulated by NRF-2/microRNA-1 regulatory axis enhances drug resistance and promotes tumorigenic properties in sorafenib-resistant hepatoma cells. Oncol Res 28:467–481. https://doi.org/10.3727/096504020X15925659763817
Shrestha R, Prithviraj P, Bridle KR, Crawford DHG, Jayachandran A (2021) Combined inhibition of TGF-beta1-induced EMT and PD-L1 silencing re-sensitizes hepatocellular carcinoma to sorafenib treatment. J Clin Med 10.https://doi.org/10.3390/jcm10091889.
Han P, Li H, Jiang X, Zhai B, Tan G, Zhao D, Qiao H, Liu B, Jiang H, Sun X (2017) Dual inhibition of Akt and c-Met as a second-line therapy following acquired resistance to sorafenib in hepatocellular carcinoma cells. Mol Oncol 11:320–334. https://doi.org/10.1002/1878-0261.12039
Xiang QF, Zhan MX, Li Y, Liang H, Hu C, Huang YM, Xiao J, He X, Xin YJ, Chen MS et al (2019) Activation of MET promotes resistance to sorafenib in hepatocellular carcinoma cells via the AKT/ERK1/2-EGR1 pathway. Artif Cells Nanomed Biotechnol 47:83–89. https://doi.org/10.1080/21691401.2018.1543195
Cai H, Yan L, Liu N, Xu M, Cai H (2020) IFI16 promotes cervical cancer progression by upregulating PD-L1 in immunomicroenvironment through STING-TBK1-NF-kB pathway. Biomed Pharmacother 123:109790. https://doi.org/10.1016/j.biopha.2019.109790
Antonangeli F, Natalini A, Garassino MC, Sica A, Santoni A, Di Rosa F (2020) Regulation of PD-L1 expression by NF-kappaB in cancer. Front Immunol 11:584626. https://doi.org/10.3389/fimmu.2020.584626
Lu PD, Zhao YH (2020) Targeting NF-kappaB pathway for treating ulcerative colitis: comprehensive regulatory characteristics of Chinese medicines. Chin Med 15:15. https://doi.org/10.1186/s13020-020-0296-z
Kang KF, Wang XW, Chen XW, Kang ZJ, Zhang X, Wilbur RR, Cheng F, Zhou SF (2013) Beclin 1 and nuclear factor-kappaBp65 are upregulated in hepatocellular carcinoma. Oncol Lett 5:1813–1818. https://doi.org/10.3892/ol.2013.1307
Torrealba N, Vera R, Fraile B, Martinez-Onsurbe P, Paniagua R, Royuela M (2020) TGF-beta/PI3K/AKT/mTOR/NF-kB pathway. Clinicopathological features in prostate cancer. Aging Male 23:801–811. https://doi.org/10.1080/13685538.2019.1597840
Wang Y, Ma ZH, Zhong LP, Yu KN, He LQ (2017) Effect of Kangxianling recipe on p38MAPK/NF-KBp65 mediated inflammatory factors in 5/6 nephrectomized mice. Zhongguo Zhong Xi Yi Jie He Za Zhi 37:365–370
Dissanayake K, Toth R, Blakey J, Olsson O, Campbell DG, Prescott AR, MacKintosh C (2011) ERK/p90(RSK)/14-3-3 signalling has an impact on expression of PEA3 Ets transcription factors via the transcriptional repressor capicua. Biochem J 433:515–525. https://doi.org/10.1042/BJ20101562
Yang S, Liu G (2017) Targeting the Ras/Raf/MEK/ERK pathway in hepatocellular carcinoma. Oncol Lett 13:1041–1047. https://doi.org/10.3892/ol.2017.5557
Li A, Zhang R, Zhang Y, Liu X, Wang R, Liu J, Liu X, Xie Y, Cao W, Xu R et al (2019) BEZ235 increases sorafenib inhibition of hepatocellular carcinoma cells by suppressing the PI3K/AKT/mTOR pathway. Am J Transl Res 11:5573–5585
Sawicka K, Pyronneau A, Chao M, Bennett MV, Zukin RS (2016) Elevated ERK/p90 ribosomal S6 kinase activity underlies audiogenic seizure susceptibility in fragile X mice. Proc Natl Acad Sci U S A 113:E6290–E6297. https://doi.org/10.1073/pnas.1610812113
Kim JB, Yang EY, Woo J, Kwon H, Lim W, Moon BI (2020) Sodium selenite enhanced the anti-proliferative effect of MEK-ERK inhibitor in thyroid cancer cells. In Vivo 34:185–190. https://doi.org/10.21873/invivo.11760
Funding
The National Natural Science Fund of China (82071862, 81872017), University Natural Science Research Project of Anhui Province (KJ2018ZD011, KJ2020A0340, KJ2019A0093), Anhui Provincial Science and Technology program (202004j07020053), and Research Foundation of the Institute of Environment-friendly Materials and Occupational Health (Wuhu), Anhui University of Science and Technology (ALW2020YF11) funded this research.
Author information
Authors and Affiliations
Contributions
Ruyue Xu, Xinkuang Liu, Yongfang Ma, Amin Li, Li Song, Jiaojiao Liang, and Jiafeng Gao conceived the study and participated in the data analysis and drafting, and wrote the manuscript. Ruyue Xu participated in the design of the study and carried out the animal and cell experiments. Xiaolong Tang conceived and designed the study. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval
Research experiments conducted in this article with animals were approved by the Ethical Committee and responsible authorities of Anhui University of Science and Technology (No. 20191013–008) following all guidelines, regulations, legal, and ethical standards as required for humans or animals.
Informed consent
Not applicable.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Xu, R., Liu, X., Li, A. et al. c-Met up-regulates the expression of PD-L1 through MAPK/NF-κBp65 pathway. J Mol Med 100, 585–598 (2022). https://doi.org/10.1007/s00109-022-02179-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00109-022-02179-2