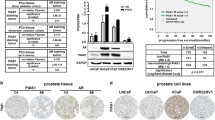
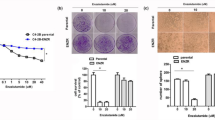
Abstract
Given the poor outcome of patients with hormone-resistant (HR) prostate cancer, new strategies are needed to improve the current therapeutic regimens and/or develop novel treatments. We therefore aimed to provide a better understanding of the molecular mechanisms involved in the aggressive tumor behavior of HR and develop more rational anti-tumor therapies. Three HR prostate cancer cell lines (androgen receptor (AR)-positive LNCaP-HR and 22RV1-HR and AR-negative PC-3) were used. Changes in tumor behavior, treatment response, and related signaling in HR were investigated in vitro and in vivo. The results revealed that constitutional activation of STAT3 and overexpressions of DNMT1 were important in the transition of HR prostate cancer. Furthermore, DNMT1 expression was required for the maintenance of STAT3 activation. When DNMT1 activity in HR was blocked, aggressive tumor behavior and treatment resistance could be overcome, which was seen in both in vitro and in vivo experiments. The underlying changes associated with inhibited DNMT1 included less epithelial–mesenchymal changes, less invasion ability, slower tumor growth, and impaired DNA repair ability, which are independent of AR and p53 status. In conclusion, altered DNMT1 expression associated with activated STAT3 may be crucial in the transition of HR. Targeting DNMT1 could be a promising strategy for the treatment of HR prostate, as evidenced by inhibited tumor growth and enhanced radiosensitivity. These findings provide evidence for therapeutically targeting DNMT1 in HR prostate cancer.






Similar content being viewed by others
References
Craft N, Chhor C, Tran C et al (1999) Evidence for clonal outgrowth of androgen-independent prostate cancer cells from androgen-dependent tumors through a two-step process. Cancer Res 59:5030–5036
Tilley WD, Buchanan G, Hickey TE et al (1996) Mutations in the androgen receptor gene are associated with progression of human prostate cancer to androgen independence. Clin Cancer Res 2:277–285
Scher HI, Buchanan G, Gerald W et al (2004) Targeting the androgen receptor: improving outcomes for castration-resistant prostate cancer. Endocr Relat Cancer 11:459–476
Herman JG, Baylin SB (2003) Gene silencing in cancer in association with promoter hypermethylation. N Engl J Med 349:2042–2054
Robertson KD (2002) DNA methylation and chromatin—unraveling the tangled web. Oncogene 21:5361–5379
Baylin SB, Herman JG (2000) DNA hypermethylation in tumorigenesis: epigenetics joins genetics. Trends Genet 16:168–174
Kim WY, Sharpless NE (2006) The regulation of INK4/ARF in cancer and aging. Cell 127:265–275
Wu CT, Chen WC, Liao SK, Hsu CL, Lee KD, Chen MF (2007) The radiation response of hormone-resistant prostate cancer induced by long-term hormone therapy. Endocr Relat Cancer 14:633–643
Chen CD, Welsbie DS, Tran C et al (2004) Molecular determinants of resistance to antiandrogen therapy. Nat Med 10:33–39
Edwards J, Bartlett JM (2005) The androgen receptor and signal-transduction pathways in hormone-refractory prostate cancer. Part 2: androgen-receptor cofactors and bypass pathways. BJU Int 95:1327–1335
Edwards J, Bartlett JM (2005) The androgen receptor and signal-transduction pathways in hormone-refractory prostate cancer. Part 1: modifications to the androgen receptor. BJU Int 95:1320–1326
Kinkade CW, Castillo-Martin M, Puzio-Kuter A et al (2008) Targeting AKT/mTOR and ERK MAPK signaling inhibits hormone-refractory prostate cancer in a preclinical mouse model. J Clin Invest 118:3051–3064
Culig Z, Steiner H, Bartsch G et al (2005) Mechanisms of endocrine therapy-responsive and -unresponsive prostate tumours. Endocr Relat Cancer 12:229–244
Wallner L, Dai J, Escara-Wilke J et al (2006) Inhibition of interleukin-6 with CNTO328, an anti-interleukin-6 monoclonal antibody, inhibits conversion of androgen-dependent prostate cancer to an androgen-independent phenotype in orchiectomized mice. Cancer Res 66:3087–3095
Gravdal K, Halvorsen OJ, Haukaas SA et al (2007) A switch from E-cadherin to N-cadherin expression indicates epithelial to mesenchymal transition and is of strong and independent importance for the progress of prostate cancer. Clin Cancer Res 13:7003–7011
Thiery JP (2002) Epithelial-mesenchymal transitions in tumour progression. Nat Rev Cancer 2:442–454
Zhang Q, Wang HY, Woetmann A, Raghunath PN, Odum N, Wasik MA (2006) STAT3 induces transcription of the DNA methyltransferase 1 gene (DNMT1) in malignant T lymphocytes. Blood 108:1058–1064
Zhang Q, Wang HY, Marzec M, Raghunath PN, Nagasawa T, Wasik MA (2005) STAT3- and DNA methyltransferase 1-mediated epigenetic silencing of SHP-1 tyrosine phosphatase tumor suppressor gene in malignant T lymphocytes. Proc Natl Acad Sci USA 102:6948–6953
Hodge DR, Cho E, Copeland TD, Guszczynski T, Yang E, Seth AK, Farrar WL (2007) IL-6 enhances the nuclear translocation of DNA cytosine-5-methyltransferase 1 (DNMT1) via phosphorylation of the nuclear localization sequence by the AKT kinase. Cancer Genomics Proteomics 4:387–398
Niu Y, Altuwaijri S, Lai KP, Wu CT, Ricke WA, Messing EM, Yao J, Yeh S, Chang C (2008) Androgen receptor is a tumor suppressor and proliferator in prostate cancer. Proc Natl Acad Sci USA 105:12182–12187
Lee JM, Dedhar S, Kalluri R, Thompson EW (2006) The epithelial-mesenchymal transition: new insights in signaling, development, and disease. J Cell Biol 172:973–981
Chu JH, Yu S, Hayward SW, Chan FL (2009) Development of a three-dimensional culture model of prostatic epithelial cells and its use for the study of epithelial–mesenchymal transition and inhibition of PI3K pathway in prostate cancer. Prostate 69:428–442
Scher HI, Sawyers CL (2005) Biology of progressive, castration-resistant prostate cancer: directed therapies targeting the androgen-receptor signaling axis. J Clin Oncol 23:8253–8261
Levy DE, Darnell JE Jr (2002) Stats: transcriptional control and biological impact. Nat Rev Mol Cell Biol 3:651–662
Culig Z, Steiner H, Bartsch G, Hobisch A (2005) Interleukin-6 regulation of prostate cancer cell growth. J Cell Biochem 95:497–505
Jones PA, Baylin SB (2002) The fundamental role of epigenetic events in cancer. Nat Rev Genet 3:415–428
Morey SR, Smiraglia DJ, James SR, Yu J, Moser MT, Foster BA, Karpf AR (2006) DNA methylation pathway alterations in an autochthonous murine model of prostate cancer. Cancer Res 66:11659–11667
Morey Kinney SR, Smiraglia DJ, James SR, Moser MT, Foster BA, Karpf AR (2008) Stage-specific alterations of DNA methyltransferase expression, DNA hypermethylation, and DNA hypomethylation during prostate cancer progression in the transgenic adenocarcinoma of mouse prostate model. Mol Cancer Res 6:1365–1374
Lin J, Tang H, Jin X, Jia G, Hsieh JT (2002) p53 regulates Stat3 phosphorylation abd DNA binding activity in human prostate cancer cells expressing constitutively active Stat3. Oncogene 21:3082–3088
Hu H, Lee HJ, Jiang C, Zhang J, Wang L, Zhao Y, Xiang Q, Lee EO, Kim SH, Lu J (2008) Penta-1, 2, 3, 4, 6-O-galloyl-beta-d-glucose induces p53 and inhibits STAT3 in prostate cancer cells in vitro and suppresses prostate xenograft tumor growth in vivo. Mol Cancer Ther 7:2681–2691, prostate cancer. Cancer 112:1660–1671
Sonpavde G, Hutson TE, Berry WR (2006) Hormone refractory prostate cancer: management and advances. Cancer Treat Rev 32:90–100
Uzzo RG, Haas NB, Crispen PL, Kolenko VM (2008) Mechanisms of apoptosis resistance and treatment strategies to overcome them in hormone-refractory prostate cancer. Cancer 112:1660–1671
Garner E, Raj K (2008) Protective mechanisms of p53-p21-pRb proteins against DNA damage-induced cell death. Cell Cycle 7:277–282
Brown JM, Attardi LD (2005) The role of apoptosis in cancer development and treatment response. Nat Rev Cancer 5:231–237
Olive PL, Banath JP (2004) Phosphorylation of histone H2AX as a measure of radiosensitivity. Int J Radiat Oncol Biol Phys 58:331–335
Mortusewicz O, Schermelleh L, Walter J, Cardoso MC, Leonhardt H (2005) Recruitment of DNA methyltransferase I to DNA repair sites. Proc Natl Acad Sci USA 102:8905–8909
Acknowledgements
The grant was support by National Science Council, Taiwan (Grant 97-2314-B-182A-079-MY3).
Competing interests
There is no conflict of interest that could be perceived as prejudicing the impartiality of the research reported.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(PDF 834 kb)
Rights and permissions
About this article
Cite this article
Chen, MF., Chen, WC., Chang, YJ. et al. Role of DNA methyltransferase 1 in hormone-resistant prostate cancer. J Mol Med 88, 953–962 (2010). https://doi.org/10.1007/s00109-010-0640-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00109-010-0640-3