Abstract
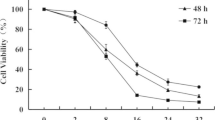
A series of glycyrrhetinic acid, oleanolic acid and ursolic acid derivatives were designed and synthesized. Structures of all novel compounds were determined by 1H NMR, 13C NMR, and HRMS methods. In vitro anti-proliferative effects of all compounds were evaluated against A549, Hela and HepG2 cancer cell lines. The three compounds G2, G3 and G4 showed better anti-proliferative effects than the positive control doxorubicin on the three kinds of cancer cells. The two compounds O1 and U11 showed better anti-proliferative effects than doxorubicin on Hela cells. The four compounds O2, O3, O8 and U8 showed better anti-proliferative effects than doxorubicin on HepG2 cells. Notably, compound G3 demonstrated the strongest growth inhibitory effects with IC50 values of 0.16 ± 0.23 µM and 0.33 ± 0.41 µM on Hela and HepG2 cells, respectively, and compound G2 demonstrated the strongest growth inhibitory effect with IC50 values of 0.80 ± 1.03 µM on A549 cells.









Similar content being viewed by others
References
Yadav D, Mishra NB, Khan F. 3D-QSAR and docking studies on ursolic acid derivatives for anticancer activity based on bladder cell line T24 targeting NF-kB pathway inhibition. J Biomol Stuct Dyn.2019;37:3822–37. https://doi.org/10.1080/07391102.2018.1528888.
International Agency for Research on Cancer. Global burden of cancer attributable to infections in 2018: a worldwide incidence analysis. 2019; 37:3822 https://doi.org/10.1016/S2214-109X(19)30488-7.
Wang HY, Zhu YF, Hao CY, Fan J, Liu Y, Wang YF. Establishment of a drug-resistant human cervical cancer cell line and research on its drug-resistance. J Cancer Res Therapeutics. 2017;15:1221–5. https://doi.org/10.4103/0973-1482.204900.
Jessica R, Anna L, Elena A, Silvia P, Francesca B, Fabrizio C, et al. A potentiated cooperation of carbonic anhydrase IX and histone deacetylase inhibitors against cancer. J Enzyme Inhibition Med Chem. 2020;35:391–7. https://doi.org/10.1080/14756366.2019.1706090.
Feng B, Zhao CH, Li JQ, Yu JW, Zhang Y, Zhang XY. et al. The novel synthetic triterpene methyl 3β-O-[4-(2-Aminoethylamino)-4-oxo-butyryl]olean-12-ene-28-oate inhibits breast tumor cell growth in vitro and in vivo. Chem Pharm Bull. 2020;68:962–70. https://doi.org/10.1248/cpb.c20-00353.
Karim C, Anis R, Stephanie D, Patrick D, Nicolas E, David T, et al. Regiospecific synthesis by copper- and ruthenium-catalyzed azide–alkyne 1,3-dipolar cycloaddition, anticancer and anti-inflammatory activities of oleanolic acid triazole derivatives. Arab J Chem. 2019;12:3732–42. https://doi.org/10.1016/j.arabjc.2015.12.013.
Tohmé MJ, Giménez MC, Peralta A, Colombo MI, Delgui LR. Ursolic acid: a novel antiviral compound inhibiting rotavirus infection in vitro. Int J Antimicrobial Agents. 2019;54:601–9. https://doi.org/10.1016/j.ijantimicag.2019.07.015.
Imran K, Mahfoozur R, Muhammad A, Gaurav G, Shakir S, Obaid A, et al. Anti-diabetic potential of ursolic acid stearoyl glucoside: a new triterpenic gycosidic ester from Lantana camara. Fitoterapia. 2012;83:142–6. https://doi.org/10.1016/j.fitote.2011.10.004.
Liang SB, Li M, Yu XJ, Jin HW, Zhang YM, Zhang LH, et al. Synthesis and structure-activity relationship studies of water-soluble β-cyclodextrin-glycyrrhetinic acid conjugates as potential anti-influenza virus agents. Eur J Med Chem. 2019;166:328–38. https://doi.org/10.1016/j.ejmech.2019.01.074.
Ma CM, Nakamura N, Hattori M. Chemical modification of oleanene type triterpenes and their inhibitory activity against HIV-1 protease dimerization. Chem Pharm Bull. 2000;48:1681–8. https://doi.org/10.1248/cpb.48.1681.
Cui ZL, Liu Z, Zeng JX, Zhang SL, Chen L, Zhang GR, et al. TRIM59 promotes gefitinib resistance in EGFR mutant lung adenocarcinoma cells. Life Sci. 2019;224:23–32. https://doi.org/10.1016/j.lfs.2019.03.041.
Onmaz DE, Abusoglu S, Unlu A, Dagli BM, Bagci M, Tok O, et al. Determination of serum imatinib and its’ metabolite in patients chronic myeloid leukemia. Clin Chim Acta. 2019;497:120–4. https://doi.org/10.1016/j.cca.2019.07.025.
Tian T, Liu X, Lee ES, Sun J, Feng Z, Zhao L, et al. Synthesis of novel oleanolic acid and ursolic acid in C-28 position derivatives as potential anticancer agents. Arch Pharmacal Res. 2017;40:458 https://doi.org/10.1007/s12272-016-0868-8.
Zhao CH, Zhang CL, Shi JJ, Hou XY, Feng B, Zhao LX. Design synthesis, and biofunctional evaluation of novel pentacyclic triterpenes bearing O-[4-(1-piperazinyl)-4-oxo-butyryl moiety as Anti-proliferative agents. Bioorganic Med Chem Lett. 2015;25:4500–4. https://doi.org/10.1016/j.bmcl.2015.08.076.
Wang L, Xu J, Zhao CH, Zhao LX, Feng B. Anti-proliferative, cell-cycle dysregulation effects of novel asiatic acid derivatives on human non-small cell lung cancer cells. Chem Pharm Bull. 2013;61:1015–23.
Stefan S, René C. Synthesis and anti-tumour activity of glycyrrhetinic acid derivatives. Bioorganic Med Chem. 2010;18:7458–74. https://doi.org/10.1016/j.bmc.2010.08.054.
Immo S, René C. An Improved scalable synthesis of α- and β-Amyrin. Molecules. 2018;23:1552–60. https://doi.org/10.3390/molecules23071552.
Tu HY, Huang AM, Wei BL, Gan KH, Hour TC, Yang SC, et al. Ursolic acid derivatives induce cell cycle arrest and apoptosis in NTUB1 cells associated with reactive oxygen species. Bioorganic Med Chem. 2009;17:7265–74. https://doi.org/10.1016/j.bmc.2009.08.046.
Jin M, Sun J, Li R, Diao S, Zhang C, Cui J, et al. Two new quinones from the roots of Juglans mandshurica. Arch Pharmacal Res. 2016;39:1237–41. https://doi.org/10.1007/s12272-016-0781-1.
Acknowledgements
This work was supported by the National Natural Science Foundation of China (Nos. 81760627).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Wang, Q., Jin, M., Liu, Y. et al. Synthesis, characterization and in vitro anti-proliferative effects of pentacyclic triterpenoids. Med Chem Res 30, 2055–2068 (2021). https://doi.org/10.1007/s00044-021-02795-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00044-021-02795-6