Abstract
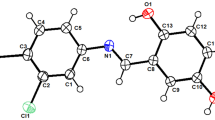
A series of novel 2-phenyl-3-(4-(5-substitutedphenylisoxazol-3-yl)phenyl)quinazolin-4(3H)-one 5a–5o were designed and synthesized from anthranilic acid. All the synthesized compounds were characterized by FT-IR, 1H NMR, mass spectroscopy, and bases of elemental analysis. Tail-flick technique, carrageenan-induced foot paw edema test, and agar streak dilution test were performed for screening analgesic, anti-inflammatory and in vitro antimicrobial activity, respectively. Moreover, all compounds were examined for its ulcerogenicity. Results of biological studies revealed that all title compounds exhibited mild to good analgesic, anti-inflammatory, and antimicrobial activity with low to moderate ulcer index. The relationship between the functional group variation and the biological activity of the evaluated compounds was discussed. Out of fifteen title compounds, 2-phenyl-3-(4-(5-(4-(trifluoromethyl)phenyl)isoxazol-3-yl)phenyl) quinazolin-4(3H)-one 5f was found to be the most active compound.


Similar content being viewed by others
References
Abdel Gawad NM, Georgey HH, Youssef RM, El-Sayed NA (2010) Synthesis and antitumor activity of some 2,3-disubstituted quinazolin-4(3H)-ones and 4,6-disubstituted-1,2,3,4-tetrahydroquinazolin-2H-ones. Eur J Med Chem 45:6058–6067
Alagarsamy V, Rajasolomon V, Vanikavitha G, Paluchamy V, Ravichandran M, Arnoldsujin A, Thangathirupathi A, Amuthalakshmi S, Revathi R (2002) Synthesis, analgesic, anti-inflammatory and antibacterial activities of some novel 2-phenyl-3-substituted quinazolin-4(3H)ones. Biol Pharm Bull 25:1432–1435
Alagarsamy V, Rajasolomon V, Meena R, Ramseshu KV (2005) Synthesis, analgesic, anti-inflammatory and antibacterial activities of some novel 2-butyl-3-substituted quinazolin-4(3H)-ones. Biol Pharm Bull 28:1091–1094
Bartroli J, Turmo E, Alguero M, Bonsompte E, Vericat ML, Conte L, Ramis J, Merlos M, Garsia-Rafanell J, Forn J (1998) New azole antifungals. 3. Synthesis and antifungal activity of 3-substituted 4(3H)-quinazolinones. J Med Chem 41:1869–1882
Beuck M (1999) Non-steroidal anti-inflammatory drugs: a new generation of cyclooxygenase inhibitors. Angew Chem Int Ed 38:631–633
Cheeseright TJ, Holm M, Lehmann F, Luik S, Gottert M, Melville JL, Laufer S (2009) Novel lead structures for p38 MAP kinase via field screen virtual screening. J Med Chem 52:4200–4209
Cheristine O, Matthias H (2004) Tryptanthrin content in isatis tinctoria leaves-A comparative study of selected strains and post harvest treatments. Planta Med 70:642–645
Chopra I, Schofield C, Everett M, Oneill A, Miller K, Wilcox M, Frere JM, Dawson M, Czaplewski L, Urleb U, Courvalin P (2008) Treatment of health care associated infections caused by gram negative bacteria: a consensus statement. Lancet Infect Dis 8:133–139
D’Amour FE, Smith DL (1941) A method for determining loss of pain sensation. J Pharmacol Exp Ther 72:74–79
Devasia RA, Jones TF, Ward J, Stafford L, Hardin H, Bopp C, Beatty M, Mintz E, Schaffner W (2006) Endemically acquired food borne outbreak of enterotoxin producing Eschericia coli serotype O169:H49. Am J Med 119:e7–e10
Dinakaran M, Selvam P, Declercq E, Sridhar SK (2003) Synthesis, antiviral and cytotoxic activity of 6-bromo-2,3-disubstituted-4(3H)-quinazolinones. Biol Pharm Bull 26:1278–1282
Fankhauser S, Laube W, Marti HR, Schultheis HR, Vogtlin J, Graf-Fenried BV (1981) Antipyretic activity of fluproquazone in man. Arzneimittelforschung 31:934–935
Ganguly AK, Bhatnagar OP (1973) Effect of bilateral adrenalectomy on production of restraint ulcers in the stomach of albino rats. Can J Physiol Pharmacol 51:748–750
Goyal RK, Chakrabarti A, Sanyal AK (1985) The effect of biological variables on the anti ulcerogenic effect of vegetable plantain banana. Planta Med 29:85–88
Gupta RR, Kumar M, Gupta V (1999) Heterocyclic Chemistry II Five-Membered Heterocycles. Springer links publishers, New york, pp 455–472
Gureje O, Von Knoff M, Simon GE, Gater R (1998) Persistent pain and well being: a world health organization study in primary care. J Am Med Assoc 280:147–151
Hawkey PM, Lewis DA (1994) Medical bacteriology-a practical approach. Oxford university press, United Kingdom, pp 181–194
Jantova S, Stankovsk S, Spirakova K (2004) In vitro antibacterial activity of ten series of substituted quinazolines. Biologica Bratislava 59:741–752
Kulkarni SK (1980) Heat and other physiological stress-induced analgesia: catecholamine mediated and naloxone reversible response. Life Sci 27:185–188
Kumar P, Shrivastava B, Pandeya SN, Stables JP (2011) Design, synthesis and potential 6 Hz psychomotor seizure test activity of some novel 2-(substituted)-3-{[substituted]amino}-quinazolin-4(3H)-one. Eur J Med Chem 46:1006–1018
Mohamed MS, Kamel MM, Kassem EMM, Abotaleb N, Abd El-moez SI, Ahmed MF (2010) Novel 6,8-dibromo-4(3H)-quinazolinone derivatives of antibacterial and antifungal activities. Eur J Med Chem 45:3311–3319
Olfert ED, Cross BM, McWilliam AA (1993) Canadian council of animal care guide to the care and use of experimental animals, vol 1, 2nd edn. Canadian Council on Animal Care, Ottawa
O’Neill LA (2006) Targeting signal transduction as a strategy to treat inflammatory diseases. Nat Rev Drug Discov 5:549–563
Panda SS, Chowdary PVR, Jayashree BS (2009) Synthesis, anti-inflammatory and antibacterial activity of novel indolyl-isoxazoles. Indian J Pharm Sci 71:684–687
Pattan SR, Krishna Reddy VV, Manvi FV, Desai BG, Bhat AR (2006) Synthesis of N-3[4(4-chlorophenylthiazol-2-yl)-2-aminomethyl]quinazolin-4(3H)-one and their derivatives for antitubercular activity. Indian J Chem 45B:1778–1781
Shay M, Komarov SA, Fels D, Meranze D, Grunstein H, Siplet H (1945) A simple method for the uniform production of gastric ulceration in the rats. Gastroenterology 5:43–61
Shukla JS, Shukla R (1989) Synthesis of 4-(5-substitutedarylidine-4-thiazolidinone-2-thione)-6,8-substituted quinazolines as potential antihelmintic agents. J Indian Chem Soc 66:209–210
Spellberg B, Guidos R, Gilbert D, Bradley J, Boucher H, Scheld WT, Bartlett J, Edwards J (2008) The epidemic of antibiotic resistant infections: a call to action for the medical community from the Infectious Diseases Society of America. Clin Infect Dis 46:155–164
Van Ryn J (1971) Inhibition of prostaglandin synthesis as a mechanism of action for aspirin like drugs. Nat New Biol 231:232–235
Van Ryn J, Botting RM (1995) New insights into the mode of action of anti-inflammatory drugs. Inflamm Res 44:1–10
Van Ryn J, Trummlitz G, Pairet M (2000) COX-2 selectivity and inflammatory processes. Curr Med Chem 7:1145–1161
Vanryzin RJ, Trpold JH (1980) The toxicology profile of the anti-inflammatory drug proquazone in animals. Drug Chem Toxicol 3:361–379
Wheathly D (1982) Analgesic properties of fluproquazone. Rheumatol Rehabil 21:98–100
Winter CA, Risley EA, Nuss GW (1962) Carrageenin induced edema in hind paw of the rat as an assay for anti-inflammatory drugs. Exp Biol Med 111:544–547
Acknowledgments
The authors gratefully acknowledge the Central Instrumentation Facility, IIT Chennai, India for the spectral analysis of the compounds used in this study. The authors are also wish to thank the management of Bapatla College of Pharmacy for providing infrastructure facilities to carry out this research work.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Saravanan, G., Alagarsamy, V. & Kumar, P.D. Synthesis and pharmacological investigations of novel 2-phenylquinazolin-4(3H)-one derivatives. Med Chem Res 24, 408–422 (2015). https://doi.org/10.1007/s00044-014-1134-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00044-014-1134-6