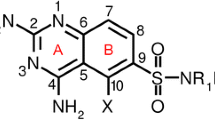
Abstract
Chalcones have shown potential to several pharmacological applications including antimalarial properties. We employed multivariate data analysis to correlate the antimalarial activity with electronic structure descriptors obtained through quantum mechanical calculations. The results show high statistical significance and bring valuable insights in order to design new compounds.




Similar content being viewed by others
References
Aoki T, Akashi T, Ayabe S (2000) Flavonoids of leguminous plants: structure, biological activity, and biosynthesis. J Plant Res 113:475–488. doi:10.1007/PL00013958
Awasthi SK, Mishra N, Kumar B, Sharma M, Bhattacharya A, Mishra LC, Bhasin VK (2009) Potent antimalarial activity of newly synthesized substituted chalcone analogs in vitro. Med Chem Res 18:407–420. doi:10.1007/s00044-008-9137-9
Barone PMVB, Camilo J, Galvão DS (1996) Theoretical approach to identify carcinogenic activity of polycyclic aromatic hydrocarbons. Phys Rev Lett 77:1186. doi:10.1103/PhysRevLett.77.1186
Braga SF, Galvão DS (2003) A structure–activity study of taxol, taxotere, and derivatives using the electronic indices methodology (EIM). J Chem Inf Comput Sci 43:699–706. doi:10.1021/ci025640v
Breman JG, Holloway CN (2007) Malaria surveillance counts. Am J Trop Med Hyg 77(6 Suppl):36–47
Coluci VR, Vendrame R, Braga RS, Galvão DS (2002) Identifying relevant molecular descriptors related to carcinogenic activity of polycyclic aromatic hydrocarbons (PAHs) using pattern recognition methods. J Chem Inf Comput Sci 42:1479–1489. doi:10.1021/ci025577+
Dewar MJS, Zoebisch EG, Healy EF, Stewart JJP (1985) Development and use of quantum mechanical molecular models 76. AM1: a new general purpose quantum mechanical molecular model. J Am Chem Soc 107:3902–3909. doi:10.1021/ja00299a024
Ghiotto RCT, Lavarda FC, Ferreira FJB (2004) Antioxidant activity of flavonols. Int J Quantum Chem 97:949–952. doi:10.1002/qua.10798
Guerin PJ, Olliaro P, Nosten F, Druilhe P, Laxminarayan R, Binka F, Kilama WL, Ford N, White NJ (2002) Malaria: current status of control, diagnosis, treatment, and a proposed agenda for research and development. Lancet Infect Dis 2:564–573. doi:10.1016/S1473-3099(02)00372-9
Hair JF, Tatham RL, Anderson RE, Black W (1998) Multivariate data analysis, 5th edn. Prentice Hall, New Jersey, p 730
Hyde J (2005) Drug-resistant malaria. Trends Parasitol 21:494–498. doi:10.1016/j.pt.2005.08.020
IBM (2001) SPSS for Windows. SPSS Inc., Chicago. www.spss.com. Accessed 22 June 2012
Karelson M, Lobanov VS, Katritzky AR (1996) Quantum-chemical descriptors in QSAR/QSPR studies. Chem Rev 96:1027–1044. doi:10.1021/cr950202r
Lavarda FC (2003) Relation between antioxidant activity and electronic structure of phenols. Int J Quantum Chem 95:219–223. doi:10.1002/qua.10692
Li R, Chen X, Gong B, Dominguez JN, Davidson E, Kurzban G, Miller RE, Nuzum EO, Rosentha PJ (1995) In vitro antimalarial activity of chalcones and their derivatives. J Med Chem 38:5031–5037. doi:10.1021/jm00026a010
Liu M, Wilairat P, Go M-L (2001) Antimalarial alkoxylated and hydroxylated chalones: structure–activity relationship analysis. J Med Chem 44:4443–4452. doi:10.1021/jm0101747
Liu M, Wilairat P, Croft SL, Tan AL-C, Go M-L (2003) Structure–activity relationships of antileishmanial and antimalarial chalcones. Bioorg Med Chem 11:2729–2738. doi:10.1016/S0968-0896(03)00233-5
Motta LF, Gaudio AC, Takahata Y (2006) Quantitative structure–activity relationships of a series of chalcone derivatives (1,3-diphenyl-2-propen-1-one) as anti Plasmodium falciparum agents (anti malaria agents). Internet Electron J Mol Des 5:555–569
Rahman MA (2011) Chalcone: a valuable insight into the recent advances and potential pharmacological activities. Chem Sci J 2011:CSJ–29
Sachs J, Malaney P (2002) The economic and social burden of malaria. Nature 415:680–685. doi:10.1038/415680a
Stewart JJP (1990) MOPAC: a semiempirical molecular orbital program. J Comput Aided Mol Des 4:1–103. doi:10.1007/BF00128336
Vendrame R, Braga RS, Takahata Y, Galvão DS (1999) Structure–activity relationship studies of carcinogenic activity of polycyclic aromatic hydrocarbons using calculated molecular descriptors with principal component analysis and neural network methods. J Chem Inf Comput Sci 39:1094–1104. doi:10.1021/ci990326v
Wongsrichanalai C, Pickard AL, Wernsdorfer WH, Meshnick SR (2002) Epidemiology of drug-resistant malaria. Lancet Infect Dis 2:209–218. doi:10.1016/S1473-3099(02)00239-6
Xue CX, Cui SY, Liu MC, Hu ZD, Fan BT (2004) 3D QSAR studies on antimalarial alkoxylated and hydroxylated chalcones by CoMFA and CoMSIA. Eur J Med Chem 39:745–753. doi:10.1016/S0968-0896(03)00233-5
Acknowledgments
This work was supported by Fundação de Amparo à Pesquisa do Estado de São Paulo (FAPESP, proc. 04/13341-1).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Batagin-Neto, A., Lavarda, F.C. The correlation between electronic structure and antimalarial activity of alkoxylated and hydroxylated chalcones. Med Chem Res 23, 580–586 (2014). https://doi.org/10.1007/s00044-013-0667-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00044-013-0667-4