Abstract
Background
Sepsis is a global fatal disease and leads to severe lung injury due to dysfunction of inflammation response. TRIM27 is closely related to the diseased with dysfunction of inflammation response. The aim of this study was to clarify the role and mechanism of TRIM27 in sepsis-induced lung injury.
Methods
The lipopolysaccharide (LPS)-induced septic mouse model was successfully established. The lung injury was evaluated by lung wet/dry (W/D) ratio and hematoxylin–eosin (H&E) staining. The cell apoptosis was evaluated by TUNEL assay. The inflammatory cytokines were measured by quantitative real time-PCR (qRT-PCR) assay and commercial enzyme-linked immunosorbent assay (ELISA). The oxidative stress was assessed by the contents of superoxide dismutase (SOD) and malondialdehyde (MDA), and the expression of dihydroethidium (DHE).
Results
In this study, we demonstrated that TRIM27 was up-regulated in LPS-induced septic mice. In loss-of-function experiments, knockdown of TRIM27 alleviated sepsis-induced lung injury, inflammation, apoptosis, and oxidative stress. More importantly, knockdown of TRIM27 was observed to reduce p-p65/NOX4 expression via suppressing ubiquitination of PPARγ. In rescue experiments, overexpression of NOX4 abolished the effect of sh-TRIM27 on alleviating sepsis-induced inflammation, apoptosis, and oxidative stress.
Conclusion
These findings highlighted that knockdown of TRIM27 alleviated sepsis-induced inflammation, oxidative stress and apoptosis via suppressing ubiquitination of PPARγ and reducing NOX4 expression, which supports the potential utility of TRIM27 as a therapeutic target in septic lung injury.
Similar content being viewed by others
Introduction
Sepsis is a global fatal disease and leads to a death rate in hospitalized patients, mainly due to excessive inflammation in response to infection [1, 2]. Owing to maladjusted host defenses against infection, sepsis induces organ dysfunction [3]. The lung is a critical organ for sepsis; thus, the occurrence of sepsis accompanies with lung injury. Notably, acute respiratory distress syndrome (ARDS) is an important factor leading to the onset and death of sepsis [4]. Once pathogen-associated molecular patterns of pathogens are recognized by body’s immune system, immune responses are activated; subsequently, inflammatory mediators are released by macrophages and neutrophils through activating the NF-κB signaling pathway [5]. Infiltration of inflammatory mediators into the lung leads to pulmonary edema and alveolar destruction, eventually resulting in hypoxic respiratory insufficiency [6]. Lipopolysaccharide (LPS) can induce acute inflammation by inducing host cells to produce pro-inflammatory cytokines, which is generally accepted to mimic sepsis-induced lung injury in animals [7].
The tripartite motif (TRIM) protein family is composed of one or two B-box motifs, a characteristic RING domain and a coiled-coil region, which is required for the interactions between proteins and participates in multiple physiological and pathological processes [8]. TRIM27 (also known as RFP) was first identified as a gene involved in oncogenic rearrangement of the RET proto-oncogene, involving in cell proliferation, transcriptional inhibition, deactivation of NF-κB, apoptosis, and inflammation response [9]. It is highly expressed in glomerular endothelial cells of patients with lupus nephritis and patients with Crohn [10, 11]. Moreover, TRIM27 promotes metastasis and proliferation of non-small cell lung cancer cells [12]. It has been reported that TRIM27 is up-regulated in psoriasis and increases the production of inflammatory cytokines [13]. Although, TRIM27 is closely related to the diseased with dysfunction of inflammation response, the role and mechanism of TRIM27 in sepsis-induced lung injury remains unclear.
Interestingly, a recent study has demonstrated that TRIM27 mediates peroxisome proliferator-activated receptor γ (PPARγ) ubiquitination and promotes glutamate-induced apoptosis and inflammation [14]. PPARγ belongs to nuclear hormone receptor family, which typically limits the feedback loop of inflammation [15]. PPARγ has been reported to improve hypoxia-induced vascular remodeling by inhibiting upregulation of NOX4 through NF-κB [9]. In this study, through the successful establishment of sepsis-induced lung injury mouse model, the effects of TRIM27 on lung injury, inflammation, oxidative stress, and apoptosis were explored, providing a novel sight for treatment of sepsis-induced lung injury.
Materials and methods
All animal operations were followed by the Guide for the Care and Use of Laboratory Animals and approved by Tianjin Third Central Hospital.
Mice model
A total of 80 male C57BL/6 mice (8-week-old, Vital River, China) were used in this study. All mice were received adaptive feeding for 1 week. All the mice were divided into eight groups, including sham, LPS, LPS+adeno-associated virus (AAV)-sh-NC, LPS+AAV-shTRIM27#1, LPS+AAV-shTRIM27#2, LPS+AAV-sh-NC+AAV-empty, LPS+AAV-sh-TRIM27#1+AAV-empty, and LPS+AAV-sh-TRIM27#1+AAV-NOX4 (n = 10 per group) at random. The mice were received intratracheal injection with 5 mg/kg LPS. Correspondingly, sham mice were received intratracheal injection with equal dose of phosphate buffer saline (PBS, Sigma). After injection for 24 h, the mice were sacrificed, and lungs were removed for further study. Three weeks before LPS injection, mice were received intratracheal administration with 6 × 1010 vector genomes (vg) of AAV-sh-TRIM27#1 (dissolved in 50 μl of PBS), AAV-sh-TRIM27#2, or AVV-NOX4 after anesthesia. Meanwhile, AAV-sh-NC or AVV-empty was served as negative control, respectively. The AAV vectors were purchased from Hanbio Company (China).
Lung wet/dry (W/D) ratio
The wet weight of lung tissues was measured, the dry weight of lung tissues was measured after desiccated at 80 °C for 48 h, and the wet/dry (W/D) weight ratio was calculated.
Histological examination and lung injury score
The lung tissues were washed with PBS, fixed, paraffin-embedded, and cut into 4-μm sections. Thereafter, the sections were deparaffinized with xylene, rehydrated in ethanol, boiled in retrieval buffer for 5 min, and stained with hematoxylin–eosin (H&E). To assess the lung injury of mice, the system comprises contained four categories (alveolar edema, alveolar hemorrhage, interstitial thickening, and neutrophil infiltration), and each category was graded from 0 (normal) to 4 (severe): 0—no injury, 1—injury to 25% of the field, 2—injury to 25–50% of the field, 3—injury to 50–75% of the field, and 4—diffuse injury.
Biological detection
Inflammatory cytokines including IL-1β, TNF-α, IL-6, and MPO, were measured by the ELISA using commercial ELISA kits (R&D Systems), respectively.
TUNEL staining assay
The cell apoptosis assay was performed by the TUNEL Kit (Roche, USA). The paraffin sections were de-waxed, dehydration, and incubated with protease K for 15 min at 37 ℃. Thereafter, the sections were blocked with TUNEL reaction solution in the dark for 15 min at 37 ℃, followed by POD incubation for 30 min at 37 ℃. Then, the sections were added with DAB for 10 min and then re-stained with hematoxylin. By captured under a fluorescence microscope (Leica, Germany), the cell apoptosis rate was calculated.
Immunofluorescence
The sections were stained with PPARγ antibody (1/400; Abcam, UK), p-p65 antibody (1/400; Abcam, UK), NOX4 antibody (1/400; Abcam, UK), DHE antibody (1/400; Abcam, UK), or TRIM27 antibody (1/400; Abcam, UK) at 4 °C, followed by Goat Anti-Rabbit IgG H&L (Alexa Fluor® 555) (1/1000; Abcam, UK) or Goat Anti-Rabbit IgG H&L (Alexa Fluor® 488) (1/1000; Abcam, UK) for 2 h at room temperature. Subsequently, the sections were stained with a DAPI solution for 8 min at room temperature, and the images were acquired under a fluorescent microscope (Leica, Germany).
Measurement of superoxide dismutase (SOD) and malondialdehyde (MDA) and dihydroethidium (DHE)
The SOD and MDA contents in lung tissues were measured by tauperoxide dismutase (T-SOD) assay kit (Nanjing Jiancheng, China) and cell malondialdehyde assay kit (Nanjing Jiancheng, China), according to the manufacturer’s instructions, respectively.
For DHE intensity measurement, the lung sections were incubated with 2 μmol/L DHE (Sigma-Aldrich, USA) in the dark for 30 min. After washing three times with PBS, the fluorescent images were captured under a fluorescent microscope (Leica, Germany) and the relative DHE intensity was calculated.
Ubiquitination analysis
The TC-1 cells (mouse alveolar epithelial cells) were purchased from the Science Cell Laboratory and cultured in DMEM (Thermo Fisher Scientific, USA) supplemented with 10% FBS (Gibco, USA) at 37 ℃ in 5% CO2. The TC-1 cells transfected with sh-TRIM27#1 or not were pre-treated by 100 ng/mL LPS for 6 h, lysed with RIPA buffer and then reacted with PPARγ antibody. Subsequently, the immunoprecipitated complexes were subjected western blot analysis using Ubiquitin (Ub) antibody (Abcam; ab7780).
Quantitative real time-PCR (qRT-PCR)
The total RNA was isolated from lung tissues with TRIzol reagent (Invitrogen, USA) and then were converted to cDNA with SuperScript IV (Invitrogen, USA) and amplified using SYBR Green Master Mix (Takara, Japan). The relative mRNA levels were normalized to GAPDH and calculated using 2−△△Ct formula. The primers were as follows: GAPDH-forward: 5′-TCATTGACCTCAACTACAGGT-3′ and GAPDH-reverse: 5′-CTAAGCAGTTGGTGGTGCAG-3′; IL-6-forward: 5′-CCGGAGAGGAGACTTCACAG-3′ and IL-6-reverse: 5′-TGGTCTTGGTCCTTAGCCAC-3′; Il-1β-forward: 5′-GGAGAAGCTGTGGCAGCTA-3′ and Il-1β-reverse: 5′-GCTGATGTACCAGTTGGGGA-3′; TNF-α-forward: 5′-GACCCTCACACTCAGATCAT-3′ and TNF-α-reverse: 5′-TTGAAGAGAACCTGGGAGTA-3′.
Western blot
Proteins were extracted from tissues with RIPA lysis and separated by SDS-PAGE as previously described [16]. The primary antibodies were listed as below: TRIM27 (1/800; Abcam, UK), GAPDH (1/800; Abcam, UK), Bax (1/800; Abcam, UK), Bcl-2 (1/800; Abcam, UK), cleaved-caspase3 (1/800; Abcam, UK), caspase3 (1/800; Abcam, UK), cleaved-caspase9 (1/800; Abcam, UK), caspase9 (1/800; Abcam, UK), PPARγ (1/800; Abcam, UK), p-p65 (1/800; Abcam, UK), p65 (1/800; Abcam, UK), and NOX4 (1/800; Abcam, UK).
Statistical analysis
The data in this study were shown as mean ± SD, repeated three times and analyzed by GraphPad Prism7.0 (USA). For survival analysis of mice, the Kaplan–Meier method with log-rank tests was performed to plot survival curves. The comparison among three groups was performed by ANOVA with Tukey’s post hoc tests. The p < 0.05 was defined as statistically significant.
Results
TRIM27 was highly expressed in LPS-induced septic mice
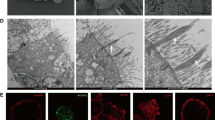
Analyzed by Kaplan–Meier survival curves, mice with LPS-induced sepsis presented a lower survival rate than sham mice (Fig. 1A). Moreover, the LPS-induced septic mice significant alveolar wall thickening with profound inflammatory cell infiltration, while the sham mice displayed normally (Fig. 1B). The LPS-induced septic mice showed a higher lung injury score than sham mice (Fig. 1C). Furthermore, the W/D ratio of lung tissues in LPS-induced septic mice was higher than that of in sham mice, indicating that LPS induced pulmonary edema in LPS-induced septic mice (Fig. 1D). Besides, cytokines, including IL-6, TNF-α, and IL-1β, in BALF were also strongly increased in LPS-induced septic mice (Fig. 1E). These data indicated the successful establishment of LPS-induced septic mouse model. Interestingly, the IF staining and western blot assays confirmed that TRIM27 was highly expressed in lung tissues of LPS-induced septic mice (Fig. 1F and G). In summary, these results concluded that TRIM27 was up-regulated in LPS-induced septic mice.
TRIM27 was highly expressed in LPS-induced septic mice. The mice were received intratracheal injection of 5 mg/kg LPS or equal PBS. A The survival of sham mice and LPS-induced septic mice was recorded over 7 days. B HE staining of mouse lung tissues at 24 h after intratracheal infection. C Increased lung injury score in LPS-treated mice. D Pulmonary edema formation was measured via determination of the wet-to-dry (W/D) lung weight ratio at 24 h after intratracheal infection. E The concentrations of TNF-α, IL-6, and IL-1β in BALF were determined using ELISA at 24 h after intratracheal infection. F The expression of TRIM27 in mice was determined by western blot. G The representative images of lung sections at 24 h after intratracheal infection for detection of TRIM27 (red) and DAPI (blue) signals. ***p < 0.001 versus the matched group
Knockdown of TRIM27 alleviated sepsis-induced lung injury
To explore the role of TRIM27 in sepsis, the sh-TRIM27 was performed to knock down the expression of TRIM27 in LPS-induced septic mice. The results of western blot assay showed that sh-TRIM27#1 and sh-TRIM27#2 efficiently reduced the expression of TRIM27 in LPS-induced septic mice (Fig. 2A). Moreover, sh-TRIM27 significantly promoted the survival of mice with LPS-induced sepsis (Fig. 2B). Furthermore, the knockdown of TRIM27 notably alleviated LPS-induced alveolar wall thickening and profound inflammatory cell infiltration in LPS-induced septic mice (Fig. 2C). Besides, the lung injury score and lung W/D ratios were markedly suppressed by sh-TRIM25 (Fig. 2D and E). These findings suggested that knockdown of TRIM27 alleviated sepsis-induced lung injury.
Knockdown of TRIM27 alleviated sepsis-induced lung injury. Before received intratracheal injection of 5 mg/kg LPS or equal PBS, the mice were pre-treated with sh-NC, sh-TRIM27#1 or sh-TRIM27#2 to decline the endogenous TRIM27 expression. A The expression of TRIM27 was detected by western blot. B The survival of mice was recorded over 7 days. C HE staining showing that knockdown of TRIM27 reduced lung injury in LPS-induced septic mice. The lung injury score (D) and W/D lung weight ratio E were measured to evaluate the sepsis-induced lung injury. ***p < 0.001, **p < 0.01 versus the matched group
Knockdown of TRIM27 alleviated sepsis-induced inflammation
Lung MPO activity is an important indicator of neutrophil infiltration [17]. Interestingly, we demonstrated that the expression and concentration of TNF-α, IL-6, IL-1β, and MPO in BALF were promoted in LPS-induced septic mice, which was further declined by knockdown of TRIM27 (Fig. 3A and B). These data indicated that knockdown of TRIM27 alleviated sepsis-induced inflammation.
Knockdown of TRIM27 alleviated sepsis-induced inflammation. (A) The expressions of TNF-α, IL-6, IL-1β, and MPO in BALF were increased in LPS-induced septic mice, and then suppressed by sh-TRIM27. (B) The concentrations of TNF-α, IL-6, IL-1β, and MPO in BALF were measured by commercial kits, respectively. ***p < 0.001, **p < 0.01 versus the matched group
Knockdown of TRIM27 suppressed sepsis-induced cell apoptosis
To further confirm the function of TRIM27 on cell apoptosis, the TUNEL staining assay was performed. Interestingly, LPS injection enhanced the number of TUNEL-positive cells in lung tissues of septic mice, while sh-TRIM27 reduced the number of TUNEL-positive cells (Fig. 4A). As we expected, the western blot results showed that knockdown of TRIM27 reduced Bcl-2, caspase3, and caspase9 expression, and promoted Bax, cleaved-caspase3 and cleaved-caspase 9 expression, indicating that knockdown of TRIM27 suppressed sepsis-induced cell apoptosis.
Knockdown of TRIM27 suppressed sepsis-induced cell apoptosis. A The cell apoptosis of lung tissues was determined by TUNEL staining assay. B The expression of Bax, Bcl-2, cleaved-caspase3, caspase3, cleaved-caspase9, and caspase9 in lung tissues were detected by western blot. ***p < 0.001 versus the matched group
Knockdown of TRIM27 alleviated sepsis-induced oxidative stress
Organ dysfunction secondary to sepsis is closely related to a surge of pro-oxidants and reduced antioxidant activity. The activity of SOD in lung tissues was increased by sh-TRIM27, while the MDA level was decreased by sh-TRIM27 (Fig. 5A). As shown in Fig. 5B, the stained lung tissues showed a strong red fluorescence produced from DHE oxidization in LPS-induced septic mice; nevertheless, knockdown of TRIM27 reduced the red fluorescence. Consistently, the LPS-induced DHE intensity was suppressed by sh-TRIM27 (Fig. 5C). These data revealed that knockdown of TRIM27 alleviated sepsis-induced oxidative stress.
Knockdown of TRIM27 alleviated sepsis-induced oxidative stress. A The SOD and MDA in lung tissues were measured, respectively. B Representative images of DHE fluorescent imaging of lung tissue sections in LPS+sh-NC group, LPS+sh-TRIM27#1 group and LPS+sh-TRIM27#2 group. C The intensity of DHE was calculated. ***p < 0.001, **p < 0.01 versus the matched group
Knockdown of TRIM27 reduced p-p65/NOX4 expression via suppressing ubiquitination of PPARγ
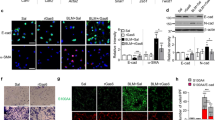
Recent studies have demonstrated that TRIM27 ubiquitinates and degrades PPARγ, and PPARγ regulates NOX4 expression through NF-κB [14, 18]. Fortunately, the ubiquitination analysis showed that TRIM27 knockdown resulted in a significant decrease in ubiquitination of PPARγ in the presence of LPS (Fig. 6A). More importantly, LPS treatment decreased PPARγ expression, and increased the ratio of p-65/p65 and NOX4 expression, while these changes were rescued by sh-TRIM27 (Fig. 6B). More importantly, the IF assay further confirmed that knockdown of TRIM27 promoted PPARγ expression but suppressed p-p65 and NOX4 expression (Fig. 6C). These findings indicated that knockdown of TRIM27 reduced p-p65/NOX4 expression via suppressing ubiquitination of PPARγ.
Knockdown of TRIM27 reduced p-p65/NOX4 expression via suppressing ubiquitination of PPARγ. A PPARγ was immunoblotted in TC-1 cells transfected with sh-TRIM27#1 or not in the absence or presence of LPS. B The expression of PPARγ, NOX4, p-p65, and p65 were detected by western blot. C The distributions of PPARγ, NOX4, and p-p65 were determined by IF assay. ***p < 0.001 versus the matched group
Knockdown of TRIM27 suppressed inflammation, apoptosis, and oxidative stress via reducing NOX4 expression
To further investigate the mechanism of TRIM27 in sepsis, the mice were pre-treated with AAV-NOX4. As shown in Fig. 7A, overexpression of NOX4 promoted the sh-TRIM27-induced reduction of IL-6, TNF-α, IL-1β, and MPO concentrations in BALF of LPS-induced septic mice. More importantly, the sh-TRIM27 increased Bcl-2, caspase3, and caspase9 expression, but decreased Bax, cleaved-caspase3, and cleaved-caspase9 expression in lung tissues, which were reversed by overexpression of NOX4 (Fig. 7B). Besides, the sh-TRIM27 induced the accumulation of SOD accompanied with a reduction of MDA, which was rescued by overexpression of TRIM27 (Fig. 7C). More importantly, the IF assay confirmed that sh-TRIM27 suppressed DHE expression, which was further enhanced by AAV-NOX4 (Fig. 7D). These findings revealed that knockdown of TRIM27 suppressed inflammation, apoptosis, and oxidative stress via reducing NOX4 expression.
Knockdown of TRIM27 suppressed inflammation, apoptosis, and oxidative stress via reducing NOX4 expression. The mice were pre-treated with sh-TRIM27 and AAV-NOX4. A The concentrations of TNF-α, IL-6, IL-1β, and MPO in BALF were measured by commercial kits. B The expression of Bax, Bcl-2, cleaved-caspase3, caspase3, cleaved-caspase9, and caspase9 in lung tissues were detected by western blot. C The SOD and MDA in lung tissues were measured. D The distribution of DHE was determined by IF staining assay. ***p < 0.001, **p < 0.01 versus the matched group
Discussion
In this study, the LPS-induced septic mouse model was successfully established. Notably, we demonstrated that TRIM27 was up-regulated in LPS-induced septic mice. In loss-of-function experiments, knockdown of TRIM27 alleviated sepsis-induced lung injury, inflammation, oxidative stress, and apoptosis. More importantly, knockdown of TRIM27 was observed to reduce p-p65/NOX4 expression via suppressing ubiquitination of PPARγ. In rescue experiments, overexpression of NOX4 abolished the effect of sh-TRIM27 on alleviating sepsis-induced inflammation, apoptosis, and oxidative stress. These findings highlighted that knockdown of TRIM27 alleviated sepsis-induced inflammation, apoptosis, and oxidative stress via suppressing ubiquitination of PPARγ and reducing NOX4 expression, supporting the potential utility of TRIM27 as a therapeutic target in sepsis-induced lung injury.
Inflammatory imbalance is the most important basis for the onset of sepsis throughout the whole process of sepsis. It has been observed that several inflammatory mediators tend to be elevated during sepsis, especially TNF-α, IL-1β, and IL-6, which have been reported to increase in sepsis patients [19]. Notably, sepsis-induced lung injury results to excessive inflammatory factors TNF-α, IL-6, and IL-1β release in blood and lung tissue of ALI mice, leading to the initiation and modulation of inflammatory response [20]. It has been reported that deficiency of TRIM27 exerts anti‐inflammatory function by impairing inflammatory cytokine expression in colitis [9]. In the current study, LPS-induced excessive of inflammatory cytokines were reflected by elevation of TNF‐α, IL-1β, and IL‐6 contents. Notably, sh-TRIM27 significantly suppressed the TNF‐α, IL-1β, and IL‐6 contents in both lung LPS-treated tissues, suggesting that its protective effect on sepsis-induced lung injury was closely associated with anti‐inflammatory properties. Nevertheless, TRIM27 can reduce the inflammatory response and ameliorate liver damage, suggesting that the function of TRIM27 is diverse in multiple diseases [21].
Accumulating evidence puts forward that inflammation cascade can induce mass necrosis and apoptosis in sepsis-induced lung injury [22]. Apoptosis is a programmed cell death process and participated in the regulation of maintaining the homeostasis of cellular differentiation and death, which may result in sepsis-induced lung injury through the activation of caspases [23, 24]. Caspase-3 belongs to in the caspase family and is a key protease to cleave the cytoskeleton and nuclear proteins, thereby, it is often used as a hallmark of apoptosis [25]. Hao et al. [14] have reported that overexpression expression of TRIM27 significantly promotes cleaved-caspase-3 expression and induced cell apoptosis. Our study indicated that sh-TRIM27 decreased the expression of cleaved-caspase-3 and suppressed sepsis-induced cell apoptosis in lung tissues, which corresponds with Hao’s report.
Oxidative stress is one of the main pathological basis of sepsis-induced organ dysfunction, as well as inflammation and apoptosis [26]. It has been clarified that Gly–Pro–Ala (GPA) peptide can attenuate sepsis-induced lung injury via suppressing oxidative stress [27]. Besides, inhibition of ERRα aggravates sepsis-induced lung injury through provoking oxidative stress and inflammation [28]. These studies indicated that increased oxidative stress plays an essential role in sepsis-induced lung injury. SOD is the primary enzymatic antioxidant enzyme in lung, which is an important indicator to assess lung damage [29]. In this study, LPS reduced SOD production and promoted oxidative stress in the mouse lung tissues, which was substantially abolished by TRIM27 knockdown. Our results may at least in part explain that TRIM27 knockout had alleviated oxidative stress in sepsis-induced lung injury. This might be the first time exploring the suppressive effect of sh-TRIM27 on oxidative stress. Nevertheless, the mechanism of how sh-TRIM27 modulates sepsis-induced inflammation, apoptosis, and oxidative stress remains unclear.
It has been reported that TRIM27 inhibits PPARγ expression via ubiquitination of PPARγ [14]. Similarly, we found that knockdown of TRIM27 enhanced PPARγ expression via reducing ubiquitination of PPARγ. Mechanically, PPARγ can bind to NF-κB p65 and suppress NF-κB. Besides, NF-κB subunits p65 stimulates NOX4 promoter activity [18]. More importantly, targeting NOX4 alleviates sepsis-induced lung injury and inflammatory responses [30, 31]. We speculate that knockdown of TRIM27 suppressed the ubiquitination of PPARγ and promoted PPARγ expression, and then inhibited the phosphorylation of p65 and NF-κB, eventually leading to the reduction of NOX4 expression and the attenuation of sepsis-induced inflammation, oxidative stress, and apoptosis. Nevertheless, there are still many limitations in the current study. First, we have yet to show whether PPARγ/NOX4 mechanism is the only mechanism of TRIM27’s therapeutic work. Also, other therapeutic targets of TRIM27 are not known yet.
In conclusion, we explored the possible effects and molecular mechanisms of TRIM27 knockout in alleviating sepsis-induced lung injury, inflammation, apoptosis, and oxidative stress via suppressing ubiquitination of PPARγ and reducing NOX4 expression, suggesting that knockdown of TRIM27 has positive significance for prevention and treatment of sepsis-induced lung injury.
Availability of data and materials
All data generated or analyzed during this study are included in this published article.
References
Wang L, Tang C, He S, Chen Y, Xie C. Combined suPAR and qSOFA for the prediction of 28-day mortality in sepsis patients. Signa Vitae. 2022;18(3):119–27. https://doi.org/10.22514/sv.2021.143.
Jiao Y, Zhang T, Zhang C, Ji H, Tong X, Xia R, et al. Exosomal miR-30d-5p of neutrophils induces M1 macrophage polarization and primes macrophage pyroptosis in sepsis-related acute lung injury. Crit Care. 2021;25(1):356. https://doi.org/10.1186/s13054-021-03775-3.
Hwang JS, Kim KH, Park J, Kim SM, Cho H, Lee Y, et al. Glucosamine improves survival in a mouse model of sepsis and attenuates sepsis-induced lung injury and inflammation. J Biol Chem. 2019;294(2):608–22. https://doi.org/10.1074/jbc.RA118.004638.
Angus DC, van der Poll T. Severe sepsis and septic shock. N Engl J Med. 2013;369(9):840–51. https://doi.org/10.1056/NEJMra1208623.
Wang YM, Ji R, Chen WW, Huang SW, Zheng YJ, Yang ZT, et al. Paclitaxel alleviated sepsis-induced acute lung injury by activating MUC1 and suppressing TLR-4/NF-kappaB pathway. Drug Des Devel Ther. 2019;13:3391–404. https://doi.org/10.2147/DDDT.S222296.
Pan X, Xu S, Zhou Z, Wang F, Mao L, Li H, et al. Fibroblast growth factor-2 alleviates the capillary leakage and inflammation in sepsis. Mol Med. 2020;26(1):108. https://doi.org/10.1186/s10020-020-00221-y.
Lien E, Means TK, Heine H, Yoshimura A, Kusumoto S, Fukase K, et al. Toll-like receptor 4 imparts ligand-specific recognition of bacterial lipopolysaccharide. J Clin Invest. 2000;105(4):497–504. https://doi.org/10.1172/JCI8541.
Okumura F, Matsunaga Y, Katayama Y, Nakayama KI, Hatakeyama S. TRIM8 modulates STAT3 activity through negative regulation of PIAS3. J Cell Sci. 2010;123(Pt 13):2238–45. https://doi.org/10.1242/jcs.068981.
Zhang HX, Xu ZS, Lin H, Li M, Xia T, Cui K, et al. TRIM27 mediates STAT3 activation at retromer-positive structures to promote colitis and colitis-associated carcinogenesis. Nat Commun. 2018;9(1):3441. https://doi.org/10.1038/s41467-018-05796-z.
Liu J, Xu J, Huang J, Gu C, Liu Q, Zhang W, et al. TRIM27 contributes to glomerular endothelial cell injury in lupus nephritis by mediating the FoxO1 signaling pathway. Lab Invest. 2021;101(8):983–97. https://doi.org/10.1038/s41374-021-00591-9.
Zurek B, Schoultz I, Neerincx A, Napolitano LM, Birkner K, Bennek E, et al. TRIM27 negatively regulates NOD2 by ubiquitination and proteasomal degradation. PLoS ONE. 2012;7(7): e41255. https://doi.org/10.1371/journal.pone.0041255.
Liu S, Tian Y, Zheng Y, Cheng Y, Zhang D, Jiang J, et al. TRIM27 acts as an oncogene and regulates cell proliferation and metastasis in non-small cell lung cancer through SIX3-beta-catenin signaling. Aging (Albany NY). 2020;12(24):25564–80. https://doi.org/10.18632/aging.104163.
Miao X, Xiang Y, Mao W, Chen Y, Li Q, Fan B. TRIM27 promotes IL-6-induced proliferation and inflammation factor production by activating STAT3 signaling in HaCaT cells. Am J Physiol Cell Physiol. 2020;318(2):C272–81. https://doi.org/10.1152/ajpcell.00314.2019.
Hao Y, Bai S, Peng J, Hong R, Ding J, Li Z, et al. TRIM27-mediated ubiquitination of PPARgamma promotes glutamate-induced cell apoptosis and inflammation. Exp Cell Res. 2021;400(1): 112437. https://doi.org/10.1016/j.yexcr.2020.112437.
Yao J, Pan D, Zhao Y, Zhao L, Sun J, Wang Y, et al. Wogonin prevents lipopolysaccharide-induced acute lung injury and inflammation in mice via peroxisome proliferator-activated receptor gamma-mediated attenuation of the nuclear factor-kappaB pathway. Immunology. 2014;143(2):241–57. https://doi.org/10.1111/imm.12305.
Chen HG, Han HZ, Li Y, Yu YH, Xie KL. Hydrogen alleviated organ injury and dysfunction in sepsis: the role of cross-talk between autophagy and endoplasmic reticulum stress: experimental research. Int Immunopharmacol. 2020;78: 106049. https://doi.org/10.1016/j.intimp.2019.106049.
Zhang X, Chang N, Zhang Y, Ye M, Han Z, Li J, et al. Bakuchiol protects against acute lung injury in septic mice. Inflammation. 2017;40(2):351–9. https://doi.org/10.1007/s10753-016-0481-5.
Lu X, Murphy TC, Nanes MS, Hart CM. PPAR{gamma} regulates hypoxia-induced Nox4 expression in human pulmonary artery smooth muscle cells through NF-{kappa}B. Am J Physiol Lung Cell Mol Physiol. 2010;299(4):L559–66. https://doi.org/10.1152/ajplung.00090.2010.
Kurt AN, Aygun AD, Godekmerdan A, Kurt A, Dogan Y, Yilmaz E. Serum IL-1beta, IL-6, IL-8, and TNF-alpha levels in early diagnosis and management of neonatal sepsis. Mediators Inflamm. 2007;2007:31397. https://doi.org/10.1155/2007/31397.
Zhao H, Chen H, Xiaoyin M, Yang G, Hu Y, Xie K, et al. Autophagy activation improves lung injury and inflammation in sepsis. Inflammation. 2019;42(2):426–39. https://doi.org/10.1007/s10753-018-00952-5.
Chen SY, Zhang HP, Li J, Shi JH, Tang HW, Zhang Y, et al. Tripartite motif-containing 27 attenuates liver ischemia/reperfusion injury by suppressing transforming growth factor beta-activated kinase 1 (TAK1) by TAK1 binding protein 2/3 degradation. Hepatology. 2021;73(2):738–58. https://doi.org/10.1002/hep.31295.
Li K, He Z, Wang X, Pineda M, Chen R, Liu H, et al. Apigenin C-glycosides of Microcos paniculata protects lipopolysaccharide induced apoptosis and inflammation in acute lung injury through TLR4 signaling pathway. Free Radic Biol Med. 2018;124:163–75. https://doi.org/10.1016/j.freeradbiomed.2018.06.009.
Li X, Jamal M, Guo P, Jin Z, Zheng F, Song X, et al. Irisin alleviates pulmonary epithelial barrier dysfunction in sepsis-induced acute lung injury via activation of AMPK/SIRT1 pathways. Biomed Pharmacother. 2019;118: 109363. https://doi.org/10.1016/j.biopha.2019.109363.
Aziz M, Ode Y, Zhou M, Ochani M, Holodick NE, Rothstein TL, et al. B-1a cells protect mice from sepsis-induced acute lung injury. Mol Med. 2018;24(1):26. https://doi.org/10.1186/s10020-018-0029-2.
Xu X, Zhu Q, Niu F, Zhang R, Wang Y, Wang W, et al. A2BAR activation attenuates acute lung injury by inhibiting alveolar epithelial cell apoptosis both in vivo and in vitro. Am J Physiol Cell Physiol. 2018;315(4):C558–70. https://doi.org/10.1152/ajpcell.00294.2017.
van der Slikke EC, An AY, Hancock REW, Bouma HR. Exploring the pathophysiology of post-sepsis syndrome to identify therapeutic opportunities. EBioMedicine. 2020;61: 103044. https://doi.org/10.1016/j.ebiom.2020.103044.
Liu Y, Zhang Y, Feng Q, Liu Q, Xie J, Li H, et al. GPA peptide attenuates sepsis-induced acute lung injury in mice via inhibiting oxidative stress and pyroptosis of alveolar macrophage. Oxid Med Cell Longev. 2021;2021:5589472. https://doi.org/10.1155/2021/5589472.
Xia W, Pan Z, Zhang H, Zhou Q, Liu Y. Inhibition of ERRalpha aggravates sepsis-induced acute lung injury in rats via provoking inflammation and oxidative stress. Oxid Med Cell Longev. 2020;2020:2048632. https://doi.org/10.1155/2020/2048632.
Ward PA. Oxidative stress: acute and progressive lung injury. Ann N Y Acad Sci. 2010;1203:53–9. https://doi.org/10.1111/j.1749-6632.2010.05552.x.
Yuan Y, Wang W, Zhang Y, Hong Q, Huang W, Li L, et al. Apelin-13 attenuates lipopolysaccharide-induced inflammatory responses and acute lung injury by regulating PFKFB3-driven glycolysis induced by NOX4-dependent ROS. J Inflamm Res. 2022;15:2121–39. https://doi.org/10.2147/JIR.S348850.
Jiang J, Huang K, Xu S, Garcia JGN, Wang C, Cai H. Erratum to Targeting NOX4 alleviates sepsis-induced acute lung injury via attenuation of redox-sensitive activation of CaMKII/ERK1/2/MLCK and endothelial cell barrier dysfunction, Redox Biology 36 (2020) 101638. Redox Biol. 2021;48: 102200. https://doi.org/10.1016/j.redox.2021.102200.
Acknowledgements
Not applicable.
Funding
This work was supported by the Science and Technology Project of Tianjin Health Committee (Grant No. ZC20226).
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation and the experiments were performed by MN and YL. Data collection and analysis were performed by DW and JW. The first draft of the manuscript was written by GH and PX, and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Conflicts of interest
The authors state that there are no conflicts of interest to disclose.
Ethics approval
Ethical approval was obtained from the Ethics Committee of Tianjin Third Central Hospital.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ning, M., Liu, Y., Wang, D. et al. Knockdown of TRIM27 alleviated sepsis-induced inflammation, apoptosis, and oxidative stress via suppressing ubiquitination of PPARγ and reducing NOX4 expression. Inflamm. Res. 71, 1315–1325 (2022). https://doi.org/10.1007/s00011-022-01625-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00011-022-01625-8