Abstract
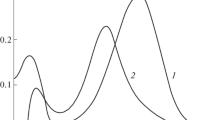
The complex formation of U(VI), Np(VI) and Pu(VI) with chloride ions was studied in HClO4−HCl solutions at ionic strength of 2.0 and [H+]=2.0M by the method of extraction chromatography using dilute HDEHP as the stationary phase.
Similar content being viewed by others
References
A. D. JONES, G. R. CHOPPIN, Actinides Rev., 1 (1969) 311.
H. LAHR, W. KNOCH, Radiochim. Acta, 13 (1970) 1.
I. FIDELIS, S. SIEKIERSKI, J. Inorg. Nucl. Chem., 28 (1966) 185.
I. FIDELIS, Bull. Acad. Polon. Sci., Ser. Sci. Chim., 18 (1970) 681.
S. SIEKIERSKI, I. FIDELIS, J. Inorg. Nucl. Chem., 31 (1972) 2225.
J. NARBUTT, W. SMUŁEK, Nukleonika, 18 (1973) 375.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Bednarczyk, L., Fidelis, I. Determination of stability constants of U(VI), Np(VI) and Pu(VI) with chloride ions by extraction chromatography. J. Radioanal. Chem. 45, 325–330 (1978). https://doi.org/10.1007/BF02519599
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02519599