Abstract
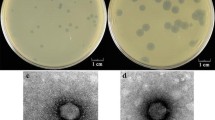
Three phages ofStreptococcus mutans were obtained and partially characterized. The three phages, designated M102, e10, and f1, were found to be strictly lytic, with host ranges restricted to only serotype c, e, and f strains of this species, respectively. Phage sensitivity was not correlated with the presence of plasmids, at least in host strains of serotypes c and e. Each phage produced clear plaques in a number of standard media, even in the presence of sucrose, indicating that the extracellular glucan polysaccharides (mutan) produced by the hosts from this substrate do not prevent phage adsorption and growth. The phages were similar in size and morphology, having icosahedral heads and long (283–287 nm), flexible, noncontractile tails. The genome of each phage was found to consist of linear, double-stranded DNA, 31–35 kb in length, with a base composition of 37–38% G+C. Restricting phage DNAs with four enzymes produced fragment patterns unique to each phage, but common bands between M102 and e10 and between e10 and f1 were produced byBamHI. Labeled e10 and M102 DNAs hybridized strongly with all three phage DNAs, indicating that they share some common sequences. The three phages appear to be more similar than expected and probably evolved from a common ancestor.
Similar content being viewed by others
Literature Cited
Ackerman HW, DuBow MS (1987) Viruses of prokaryotes, vol 1. General properties of bacteriophages. Boca Raton, Florida: CRC Press
Armau E, Bousque JL, Boue D, Tiraby G (1988) Isolation of lytic bacteriophages forStreptococcus mutans andStreptococcus sobrinus [abstr]. J Dent Res 67:121
Caufield PW, Shah G, Hollingshead SK, Parrot M, Lavoie MC (1990) Evidence that mutacin II production is not mediated by a 5.6-kb plasmid inStreptococcus mutans. Plasmid 24:110–118
Delisle AL, Donkersloot JA, Kolenbrander PE, Tylenda CA (1988) Use of lytic bacteriophage forActinomyces viscosus T14V as a probe for cell surface components mediating intergeneric coaggregation. Infect Immun 56:54–59
Greer S, Hsiang BW, Musil G, Zinner DD (1971) Viruses of cariogenic streptococci. J Dent Res 50:1594–1604
Halhoul N, Colvin JR (1975) Virus-like particles in association with a microorganism from human gingival plaque. Arch Oral Biol 20:833–836
Higuchi M, Rhee GH, Araya S, Higuchi M (1977) Bacteriophage deoxyribonucleic acid-induced mutation ofStreptococcus mutans. Infect Immun 15:938–944
Klein JP, Frank RM (1973) Mise en evidence de virus dans les bacterie cariogenes de la plaque dentaire. J Biol Buccale 1:79–85
Loesche WJ (1986) Role ofStreptococcus mutans in human dental decay. Microbiol Rev 50:353–380
Meyers CE, Walters EL, Green LB (1958) Isolation of a bacteriophage specific for aLactobacillus caseii from human oral material. J Dent Res 37:175–178
Sambrook J, Fritsch EF, Maniatis T (1989) Molecular cloning. A laboratory manual, 2nd ed. New York: Cold Spring Harbor Laboratory Press
Tylenda CA, Enriquez E, Kolenbrander PE, Delisle AL (1985) Simultaneous loss of bacteriophage receptor and coaggregation mediator activities inActinomyces viscosus MG-1. Infect Immun 48:228–233
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Delisle, A.L., Rostkowski, C.A. Lytic bacteriophages ofStreptococcus mutans . Current Microbiology 27, 163–167 (1993). https://doi.org/10.1007/BF01576015
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF01576015