Summary
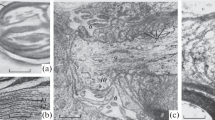
This paper examines the neuropathology of oxygen-glucose deprivation uncomplicated by stagnant conditions. Rabbit vagus nerves were pulled into asmulti-compartment perfusion chamber, stimulated five times per second and deprived of energy by substituting nitrogen and deoxyglucose for oxygen and glucose in the Locke's perfusate. After incubation the compartments were perfused with gluteraldehyde solution, and the nerves were prepared for electron microscopy. Fixation in the compartments ensured precise cross and longitudinal sections which permitted quantitative comparisons. Although the action potentials ceased in 45 min, 1 h of energy deprivation did not significantly affect the ultrastructure. After 2 h of deprivation the axons were smaller and flattened and microtubules appeared packed together. In the smallest axons the microtubules were gone, the neurofilaments were compacted and the few mitochondria had a dense, homogenous appearance. By 4 h the shrinking was extreme, yet 8% were swollen much larger than any of the controls. Longitudinal views showed these balloned areas were greatly expanded regions of the smallest axons. Both tiny and huge regions were devoid of microtubules and the swollen axons contained expanded mitochondria.
Calcium is indirectly implicated in the pathogenesis by the concurrence of mitochondrial alteration as the microtubules disappear coupled with the known role of mitochondria in calcium regulation and the reported effect of high calcium on microtubual dissociation. In is suggested that axons first shrink as osmotially active molecules are used or washed out. After a time without energy the mitochondria can no longer regulate the intracellular calcium, microtubules dissociate, and calcium-activated phospholipases create osmotically active molecules. Finally, high-amplitude, disruptive swelling occurs.
Similar content being viewed by others
References
Brown AW (1977) Structural abnormalities in neurons. J Clin Pathol 30: [Suppl (Roy Coll Pathol) 11]:155–169
Brown AW, Brierley JB (1973) The carliest alterations in rat neurons and astrocytes after anoxia-ischemia. Acta Neuropathol (Berl) 23:9–22
Dahl NA, Shirer HW (1979) Multicompartment nerve chamber for recording electrical potentials. J Electrophysiol Tech 6:26–33
Donoso JA, Illanes J-P, Samson F (1977) Dimethylsulfoxide action on fast axoplasmic transport and ultrastructure of vagal axons. Brain Res 120:28–301
Ellisman MH (1981) Beyond neurofilaments and microtubules. Neurosci Res Prog Bull 19:43–58
Garcia JH, Kalimo H, Kamiyo Y, Trump BF (1977) Cellular events during partial cerebral ischemia. I. Electron microscopy of feline cerebral cortex after middle-cerebral-artery occlusion. Virchows Arch B [Cell Pathol] 25:191–206
Garcia HJ, Lossinsky AS, Kauffman FC, Conger KA (1978) Neuronal ischemic injury: Light microscopy, ultrastructure and biochemistry. Acta Neuropathol (Berl) 43:85–95
Kalimo H, Garcia JH, Kamijyo Y, Tanaka J, Trump BF (1977) The ultrastructure of “brain death”. II. Electron microscopy of feline cortex after complex ischemia. Virchows Arch B [Cell Pathol] 25:207–220
Lee YC, Samson FE, Houston LL, Himes RH (1974) The in vitro polymerization of tubulin from beef brain. J Neurobiol 5:317–330
Levy DE, Brierley JB, Plum F (1975) Ischemic brain damage in the gerbil in the absence of “no-reflow”. J Neurol Neurosurg Psychiat 38:1197–1205
Shay J (1973) Morphometry of an ischemic lesion of cat spinal cord. Am J Pathol 72:397–402
Siesjö BK (1981) Cell damage in the brain: a speculative synthesis. J Cerebr Blood Flow Metab 1:155–185
Sokal RR, Rohlf JF (1969) Biometry. The principles and practice of statistics in biological research. Freeman & Co. San Francisco, CA
Tanaka R, Tanimura K, Ueki K (1977) Ultrastructural and biochemical studies on ouabain-induced oedematous brain. Acta Neuropathol (Berl) 37:95–100
Trump BF, Berezesky IK, Laiho KU, Osornio AR, Mergner WJ, Smith MW (1980) The role of calcium in cell injury: A review. Scan Electron Microsc 2:437–462
Author information
Authors and Affiliations
Additional information
Supported, in part, by a Grant-in-aid from the American Heart Association with funds contributed by the American Heart Association, Kansas Affiliate and by the University of Kansas Biomedical Sciences Support Grant RR0737
Rights and permissions
About this article
Cite this article
Dahl, N.A., Looney, G.A. & Black, W.H. Ultrastructure of non-myelinated neurons during energy deprivation. Acta Neuropathol 57, 111–120 (1982). https://doi.org/10.1007/BF00685378
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00685378