Conclusion
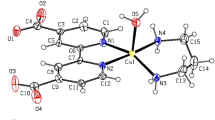
The x-ray structural results confirm what has been ascertained by thermodynamic and spectroscopic data in aqueous solution(7). It is evident that the addition of pyridine to the solution containing the mixed species [Cu(bipy)(pydca)(H2O)] leads to the substitution of a water molecule directly bound to copper(II) ion by a pyridine molecule. This experiment also demonstrated the presence of a water molecule in the equatorial plane of the complex.
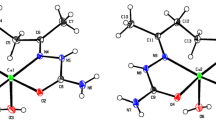
The subsequent diffractometric study on single crystals derived from the copper(II)/bipy/pydca system revealed the existence in the solid state of [Cu2(bipy)2(pydca)2] · 4H2O. Thus the pydca dianion, instead of forming the statistically favoured mixed complex [Cu(bipy)(pydca)], gives rise to crystals containing two different copper(II) environments: [Cu(pydca)2]2− and [Cu(bipy)2]2+, linked by O-carboxylate bridges. The facility with which [Cu(bipy)(pydca)(py)] can be obtained shows that the addition of pyridine prevents the formation of polynuclear species.
Similar content being viewed by others
References
G. Nardin, L. Randaccio, R. P. Bonomo and E. Rizzarelli,J. Chem. Soc., Dalton Trans., 369 (1980).
R. P. Bonomo, E. Rizzarelli, N. Bresciani-Pahor and G. Nardin,J. Chem. Soc., Dalton Trans., 681 (1982).
R. P. Bonomo, E. Rizzarelli, N. Bresciani-Pahor and G. Nardin,Inorg. Chim. Acta, 54, L17 (1981).
R. P. Bonomo, E. Rizzarelli, N. Bresciani-Pahor and G. Nardin,J. Chem. Soc., Dalton Trans., 1797 (1983).
N. Bresciani-Pahor, G. Nardin, R. P. Bonomo and E. Rizzarelli,J. Chem. Soc., Dalton Trans., 2625 (1984).
J. R. Pilbrow and M. E. Winfield,Mol. Phys., 25, 1073 (1973).
R. P. Bonomo, R. Calí, F. Riggi, E. Rizzarelli and S. Sammartano,Inorg. Chem., 18, 3417 (1980).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Bresciani-Pahor, N., Nardin, G., Bonomo, R.P. et al. Structure and spectroscopic characterization of the quaternary system: Copper(II), pyridine-2,6-dicarboxylate, 2,2′-bipyridyl and pyridine. Transition Met Chem 10, 316–318 (1985). https://doi.org/10.1007/BF00619018
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00619018