Abstract
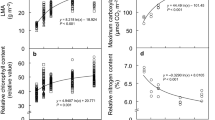
Measurements of CO2 and H2O exchange rate and the calculated leaf conductance of attached leaves were conducted over a range of leaf-to-air vapour pressure difference (VPD) (1.5 to 5.5 kPa) to compare the response of the parasitic mistletoe, Phthirusa pyrifolia, with that of its host, the mandarin orange, Citrus reticulata. Seedlings of the host infected with the parasite were grown in well-watered and adequately fertilized large pots outdoors at the CIAT headquarters, Palmira, Colombia, South America. Observations of leaf anatomy of the parasite and nutrient analysis of young tissues of both the parasite and host were made. The photosynthetic rate of the host decreased linearly with increased VPD, whereas the parasite showed a constant rate. This trend coincided with similar responses in leaf conductance. Due to the insensitivity of the parasite stomata, the transpiration rate increased linearly with VPD as compared with an initial increase and then a decrease in the host transpiration rate. The higher photosynthetic rate and the closure of stomata of the host resulted in high water use efficiency as compared with that of the parasite. The parasite accumulated in its leaves more N, P, K and less Ca and Mg than the host. The significance of the host-parasite differential response to air humidity is discussed in relation to mechanism underlying stomatal sensitivity and in the context of host-parasite association.
Similar content being viewed by others
References
Arbelaez EP (1956) Plantas Utiles de Colombia. Sucesores de Rivadeneyra. Madrid
Atsatt PR (1983) Host-parasite interactions in higher plants. In Lange OL, Nobel PS, Osmond CB and Ziegler H (eds.). Encyclopedia of plant physiology. Vol. 12C pp. 519–535 Springer-Verlag. Berlin
Barlow BA and Wines D (1977) Host-parasite resemblance in Australian mistletoes: the case for cryptic mimicry. Evolution 31: 69–84
Bunce JA (1985) Effect of boundary layer conductance on the response of stomata to humidity. Plant Cell Environ 8: 55–57
Dobbins DR and Kuijt J (1974) Anatomy and fine structure of the mistletoe haustorium (Phthirus pyrifolia). I. Development of the young haustorium. Amer J Bot 61: 535–543
Dobbins DR and Kuijt J (1974) Anatomy and fine structure of the mistletoe haustorium (Phthirusa pyrifolia). II. Penetration attempts and formation of the gland. Amer J Bot 544–550
Ehleringer JR, Schulze E-D, Ziegler H, Lange OL, Farquhar GD and Cown IR (1985) Xylem-tapping mistletoes: water or nutrient parasite? Science 227: 1479–1481
El-Sharkawy MA, Cock JH, and Hernández AP (1985) Stomatal response to air humidity and its relation to stomatal density in a wide range of warm climate species. Photosynthesis Research (in press)
El-Sharkawy MA and Cock JH (1984) Water use efficiency of cassava. I. Effects of air humidity and water stress on stomatal conductance and gas exchange. Crop Sci 24: 497–502
El-Sharkawy MA, Cock JH and Held AK (1984) Water use efficiency of cassava. II. Differing sensitivity of stomata to air humidity in cassava and other warm-climate species. Crop Sci 24: 503–507
Farquhar GD and Sharkey TD (1982) Stomatal conductance and photosynthesis. Ann Rev Plant Physiology. 33: 317–345
Gaastra P (1959) Photosynthesis of crop plants as influenced by light, CO2, temperature and stomatal diffusion resistance. Meded Landbouwhogesch Wageningen 59: 1–68
Jarvis PG (1980) Stomatal response to water stress in conifers. In Turner NC and Kramer PJ (eds.) Adaptation of plants to water and high temperature stress pp. 105–122. John Wiley & Sons. New York.
Korner CH, Scheel JA and Bauer H (1979) Maximum leaf diffusive conductance in vascular plants. Photosynthetica 13: 45–82
Kuijt J (1977) Haustoria of phanerogamic parasites. Ann Rev Phytopathol 17: 91–118
Kuijt J and Toth R (1976) Ultrastructure of Angiosperm haustoria—A review. Ann Bot 40: 1121–1130
Lange OL, Losch R, Schulze E-D and Kappen L (1971) Responses of stomata to changes in humidity. Planta, 100: 76–86
Ludlow MM (1980) Adaptive significance of stomatal responses to water stress. In Turner NC and Kramer PJ (eds.) Adaptation of plants to water and high temperature stress pp. 123–138. John Wiley & Sons. New York
Maier-Maercker U (1983) The role of peristomatal transpiration in the mechanism of stomatal movement. Plant Cell Environ 6: 369–380
Mansfield TA and Davies WJ (1981) Stomata and Stomatal mechanisms. In Paleg and Aspinal D (eds) The physiology and biochemistry of drought resistance in plants. Academic Press, Sydney
Raven JA (1983) Phytophages of xylem and phloem: a comparison of Animal and Plant sap-feeders. Advances in Ecological Research 13: 135–234
Schulze E-D, Turner NC and Glatzel G (1984) Carbon, water and nutrient relations of two mistletoes and their hosts: A hypothesis. Plant Cell Environ 7: 293–299
Schulze E-D and Hall AE (1982) Stomatal responses, water loss and CO2 assimilation rates of plants in contrasting environments. In Lange OL, Nobel PS, Osmond CB and Ziegler H (eds.) Encyclopedia of plant physiology. Vol. 12B. pp 181–230 Springer-Verlag, Berlin
Sheriff DW (1979) Stomatal aperture and the sensing of the environment by guard cells. Plant Cell Environ. 2: 15–22
Ziegler H (1975) Nature of transported substance. In Zimmermann MH and Milburn JA (eds.) Encyclopedia of plant physiology. Vol. 1 pp 59–100 Springer-Verlag. Berlin
Author information
Authors and Affiliations
Additional information
Visiting Scientist, Coordinator, and Research Assistant of the Cassava Physiology Program.
Rights and permissions
About this article
Cite this article
El-Sharkawy, M.A., Cock, J.H. & Del Pilar Hernandez, A. Differential response of stomata to air humidity in the parasitic mistletoe (Phthirusa pyrifolia) and its host, mandarin orange (Citrus resitulata). Photosynth Res 9, 333–343 (1986). https://doi.org/10.1007/BF00029798
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00029798