Abstract
Sewage sludge valorization must be the favored solution of management for a residue that does not present the desired improvement in terms of wastewater treatment. The objective of this research work was to study the dye fixation capacity of the adsorbents produced from sewage sludge. The dried sewage sludge (DSS) was used as an adsorbent to remove a cationic dye, methylene blue (MB), from an aqueous solution in a batch system, as a function of contact time, pH, temperature and initial concentration. The proposed adsorbent was characterized using several techniques such as scanning electron microscopy (SEM), Brunauer–Emmett–Teller (BET) analysis, X-ray diffraction (XRD) and Fourier Transform Infrared Spectroscopy (FTIR). The adsorption equilibrium of MB was described using Langmuir, Freundlich and Sips equations. The equilibrium is perfectly adapted to the Langmuir model with a maximum adsorption capacity of 44.39 mg g−1 on a single layer. In order to study the adsorption mechanisms, first- and second-order kinetic models were used. The adsorbent produced from the sludge was capable of absorbing MB from the solution, with the expression of the second-order rate being preferred to that of the first order. In addition, the thermodynamic parameters were evaluated, demonstrating that the adsorption process is spontaneous and endothermic. Thus, this study convinced that the DDS proved to be an alternative, attractive, effective, economic, and environmentally friendly adsorbent for MB dye removal from aqueous solution.
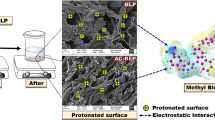
Graphic abstract
Schematic illustration of the prepared dried sewage sludge adsorbent for the removal of methylene blue from aquatic samples










Similar content being viewed by others

References
Chanaka Udayanga WD, Veksha A, Giannis A, Lisak G, Chang VWC, Lim TT (2018) Fate and distribution of heavy metals during thermal processing of sewage sludge. Fuel 226:721–744
Dubis B, Jankowski KJ, Załuski D, Sokólski M (2020) The effect of sewage sludge fertilization on the biomass yield of giant miscanthus and the energy balance of the production process. Energy 206
Werle S, Wilk RK (2010) A review of methods for the thermal utilization of sewage sludge: the polish perspective. Renew Energy 35(9):1914–1919
Werle S, Dudziak M (2014) Gaseous fuels production from dried sewage sludge via air gasification. Waste Manag Res 32(7):601–607
Schnell M, Horst T, Quicker P (2020) Thermal treatment of sewage sludge in Germany: a review. J Environ Manage 263:110367.
Joo SH, Dello Monaco F, Antmann E, Chorath P (2015) Sustainable approaches for minimizing biosolids production and maximizing reuse options in sludge management: a review. J Environ Manage 158:133–145
Agabo-García C, Pérez M, Rodríguez-Morgado B, Parrado J, Solera R (2019) Biomethane production improvement by enzymatic pre-treatments and enhancers of sewage sludge anaerobic digestion. Fuel 255:115713
Rasapoor M, Young B, Brar R, Sarmah A, Zhuang WQ, Baroutian S (2019) Recognizing the challenges of anaerobic digestion: critical steps toward improving biogas generation. Fuel 261:116497
Lu D, Wu D, Qian T, Jiang J, Cao S, Zhou Y (2020) Liquid and solids separation for target resource recovery from thermal hydrolyzed sludge. Water Res 171:115476
Collivignarelli MC, Canato M, Abbà A, Carnevale Miino M (2019) Biosolids: what are the different types of reuse? J Clean Prod 238.
https://secretariatministre@environnement.gov.ma, “Ref, [11),” no. January. pp. 2–3, 1976.
Lavoie J, Srinivasan S, Nagarajan R (2011) Ref 12. J Hazard Mater 194:85–91
Belloulid MO, Moussaoui TEL, Saykouk J, Azzouzi FEL, Words KEY (2018) National program of sanitation and wastewater treatment in Morocco: objectives, achievements and challenges programme national d ’ assainissement liquide et d ’ épuration des eaux usées au Maroc: objectifs, réalisations et défis. Environ Water Sci Public Heal Territ Intell 2(1):67–76
Hadi P, Xu M, Ning C, Sze Ki Lin C, McKay G (2015) A critical review on preparation, characterization and utilization of sludge-derived activated carbons for wastewater treatment. Chem Eng J 260:895–906
Naik PK (2017) Water crisis in Africa: myth or reality? Int J Water Resour Dev 33(2):326–339
Choukri F et al (2020) Distinct and combined impacts of climate and land use scenarios on water availability and sediment loads for a water supply reservoir in northern Morocco. Int Soil Water Conserv Res 8(2):141–153
Ahrouch M, Gatica JM, Draoui K, Bellido D, Vidal H (2020) Honeycomb filters as an alternative to powders in the use of clays to remove cadmium from water. Chemosphere 259:127526
Gorguner M, Kavvas ML (2020) Modeling impacts of future climate change on reservoir storages and irrigation water demands in a Mediterranean basin. Sci Total Environ 748:141246
Zhang H, et al. (2020) Production of biochar from waste sludge/leaf for fast and efficient removal of diclofenac, vol. 299. Elsevier B.V.
Li WH, Yue QY, Gao BY, Ma ZH, Li YJ, Zhao HX (2011) Preparation and utilization of sludge-based activated carbon for the adsorption of dyes from aqueous solutions. Chem Eng J 171(1):320–327
Wang M et al (2021) Fabrication of monodispersed plasmonic photocatalysts on activated carbon with the carbon source and reduction property of sewage sludge. Appl Surf Sci 538:148036
Costa JAS, de Jesus RA, Santos DO, Mano JF, Romão LPC, Paranhos CM (2020) Recent progresses in the adsorption of organic, inorganic, and gas compounds by MCM-41-based mesoporous materials. Microporous Mesoporous Mater 291:109698
Sellaoui L, et al. (2020) Insights of the adsorption mechanism of methylene blue on brazilian berries seeds: Experiments, phenomenological modelling and DFT calculations. Chem Eng J 394
Kausar A et al (2019) Preparation and characterization of chitosan/clay composite for direct Rose FRN dye removal from aqueous media: Comparison of linear and non-linear regression methods. J Mater Res Technol 8(1):1161–1174
Tran HN, et al. (2020) Innovative spherical biochar for pharmaceutical removal from water: insight into adsorption mechanism. J Hazard Mater 394: 122255
Yazidi A et al (2019) Adsorption of amoxicillin and tetracycline on activated carbon prepared from durian shell in single and binary systems: Experimental study and modeling analysis. Chem Eng J 379(July):2020
Pang X et al (2020) Preparation and characterization of a novel mountain soursop seeds powder adsorbent and its application for the removal of crystal violet and methylene blue from aqueous solutions. Chem Eng J 391:123617
Chahinez HO, Abdelkader O, Leila Y, Tran HN (2020) One-stage preparation of palm petiole-derived biochar: Characterization and application for adsorption of crystal violet dye in water. Environ Technol Innov 19:100872
Ahsaine HA et al (2018) Photo/electrocatalytic properties of nanocrystalline ZnO and La–Doped ZnO: Combined DFT fundamental semiconducting properties and experimental study. Chem Select 3(27):7778–7791
Anfar Z, El Haouti R, Lhanafi S, Benafqir M, Azougarh Y, El Alem N (2017) Treated digested residue during anaerobic co-digestion of Agri-food organic waste: Methylene blue adsorption, mechanism and CCD-RSM design. J Environ Chem Eng 5(6):5857–5867
Malik PK, Saha SK (2003) Oxidation of direct dyes with hydrogen peroxide using ferrous ion as catalyst. Sep Purif Technol 31(3):241–250
Silva LGM, et al. (2020) Integration of fenton’s reaction based processes and cation exchange processes in textile wastewater treatment as a strategy for water reuse. J Environ Manage 272
Ciardelli G, Corsi L, Marcucci M (2001) Membrane separation for wastewater reuse in the textile industry. Resour Conserv Recycl 31(2):189–197
Alventosa-Delara E, Barredo-Damas S, Zuriaga-Agustí E, Alcaina-Miranda MI, Iborra-Clar MI (2014) Ultrafiltration ceramic membrane performance during the treatment of model solutions containing dye and salt. Sep Purif Technol 129:96–105
Ambaye TG, Hagos K (2020) Photocatalytic and biological oxidation treatment of real textile wastewater. Nanotechnol Environ Eng 5
Zbair M, Ait Ahsaine H, Anfar Z (2018) Porous carbon by microwave assisted pyrolysis: an effective and low-cost adsorbent for sulfamethoxazole adsorption and optimization using response surface methodology. J Clean Prod 202:571–581
Badri A, Jabli M, López ML, Ben Amara M (2019) Synthesis, electrical and dye adsorption properties of KZnFe(PO4)2 zeolite type structure. Inorg Chem Commun 110: 107609
Mohammed MA, Shitu A, Ibrahim A (2014) Removal of methylene blue using low cost adsorbent: a review. Res J Chem Sci 4(1):91–102
Ahrouch M, Gatica JM, Draoui K, Vidal H (2019) Adding value to natural clays as low-cost adsorbents of methylene blue in polluted water through honeycomb monoliths manufacture. SN Appl Sci 1:2019
Qi Y, Hoadley AFA, Chaffee AL, Garnier G (2011) Characterisation of lignite as an industrial adsorbent. Fuel 90(4):1567–1574
Yin Q, Liu M, Ren H (2019) Biochar produced from the co-pyrolysis of sewage sludge and walnut shell for ammonium and phosphate adsorption from water. J Environ Manage 249:109410
Otero M, Rozada F, Calvo LF, García AI, Morán A (2003) Kinetic and equilibrium modelling of the methylene blue removal from solution by adsorbent materials produced from sewage sludges. Biochem Eng J 15(1):59–68
Dhaouadi H, M’Henni F (2009) Vat dye sorption onto crude dehydrated sewage sludge. J Hazard Mater 164(2–3):448–458
Fan S et al (2016) Biochar prepared from co-pyrolysis of municipal sewage sludge and tea waste for the adsorption of methylene blue from aqueous solutions: kinetics, isotherm, thermodynamic and mechanism. J Mol Liq 220:432–441
Li H, Liu L, Cui J, Cui J, Wang F, Zhang F (2020) High-efficiency adsorption and regeneration of methylene blue and aniline onto activated carbon from waste edible fungus residue and its possible mechanism. RSC Adv 10(24):14262–14273
Sawasdee S, Jankerd H, Watcharabundit P (2017) Adsorption of dyestuff in household-scale dyeing onto rice husk. Energy Procedia 138:1159–1164
Mouni L, et al. (2018) Removal of methylene blue from aqueous solutions by adsorption on kaolin: kinetic and equilibrium studies. Appl Clay Sci 153:38–45
Cheng J et al (2020) Highly efficient removal of methylene blue dye from an aqueous solution using cellulose acetate nanofibrous membranes modified by polydopamine. ACS Omega 5(10):5389–5400
Abdallah M, Hijazi A, Hamieh M, Alameh M, Toufaily J, Rammal H (2016) étude de l’adsorption du Bleu de Méthylène sur un biomatériau à base de l’eucalyptus selon la taille des particules. J Mater Environ Sci 7(11):4036–4048
Barka N, Abdennouri M, Makhfouk MEL (2011) Removal of methylene blue and eriochrome black T from aqueous solutions by biosorption on Scolymus hispanicus L.: Kinetics, equilibrium and thermodynamics. J Taiwan Inst Chem Eng 42(2):320–326
Rida K, Bouraoui S, Hadnine S (2013) Adsorption of methylene blue from aqueous solution by kaolin and zeolite. Appl Clay Sci 83–84:99–105
Malekbala MR, Khan MA, Hosseini S, Abdullah LC, Choong TSY (2015) Adsorption/desorption of cationic dye on surfactant modified mesoporous carbon coated monolith: equilibrium, kinetic and thermodynamic studies. J Ind Eng Chem 21:369–377
Djilani C et al (2015) Adsorption of dyes on activated carbon prepared from apricot stones and commercial activated carbon. J Taiwan Inst Chem Eng 53:112–121
Elmoubarki R et al (2015) Adsorption of textile dyes on raw and decanted Moroccan clays: kinetics, equilibrium and thermodynamics. Water Resour Ind 9:16–29
Bentahar Y, Draoui K, Hurel C, Ajouyed O, Khairoun S, Marmier N (2019) Physico-chemical characterization and valorization of swelling and non-swelling Moroccan clays in basic dye removal from aqueous solutions. J African Earth Sci 154:80–88
Nethaji S, Sivasamy A, Mandal AB (2013) Adsorption isotherms, kinetics and mechanism for the adsorption of cationic and anionic dyes onto carbonaceous particles prepared from Juglans regia shell biomass. Int J Environ Sci Technol 10(2):231–242
Chen H, Zhao J, Wu J, Dai G (2011) Isotherm, thermodynamic, kinetics and adsorption mechanism studies of methyl orange by surfactant modified silkworm exuviae. J Hazard Mater 192(1):246–254
Lima EC, Hosseini-Bandegharaei A, Moreno-Piraján JC, Anastopoulos I (2019) A critical review of the estimation of the thermodynamic parameters on adsorption equilibria. Wrong use of equilibrium constant in the Van’t Hoof equation for calculation of thermodynamic parameters of adsorption. J Mol Liq 273:425–434
Lebkiri I et al (2019) Removal of methylene blue dye from aqueous solution using a superabsorbant hydrogel the polyacrylamide: Isotherms and kinetic studies. Mediterr J Chem 9(5):337–346
Xia Yang C, Lei L, Xin Zhou P, Zhang Z, Qiang Lei Z (2015) Preparation and characterization of poly(AA co PVP)/PGS composite and its application for methylene blue adsorption. J Colloid Interface Sci 443: 97–104
Sakin Omer O, Hussein MA, Hussein BHM, Mgaidi A (2018) Adsorption thermodynamics of cationic dyes (methylene blue and crystal violet) to a natural clay mineral from aqueous solution between 293.15 and 323.15 K. Arab J Chem 11(5): 615–623
Li H, Dai M, Dai S, Dong X, Li F (2018) Methylene blue adsorption properties of mechanochemistry modified coal fly ash. Hum Ecol Risk Assess 24(8):2133–2141
Albadarin AB, Collins MN, Naushad M, Shirazian S, Walker G, Mangwandi C (2017) Activated lignin-chitosan extruded blends for efficient adsorption of methylene blue. Chem Eng J 307:264–272
Brindley GW, Thompson TD (1970) Methylene blue absorption by montmorillonites. Determinations of surface areas and exchange capacities with different initial cation saturations (clay-organic studies XIX). Isr J Chem 8(3):409–415
Auta M, Hameed BH (2012) Modified mesoporous clay adsorbent for adsorption isotherm and kinetics of methylene blue. Chem Eng J 198–199:219–227
Han R, Zhang J, Han P, Wang Y, Zhao Z, Tang M (2009) Study of equilibrium, kinetic and thermodynamic parameters about methylene blue adsorption onto natural zeolite. Chem Eng J 145(3):496–504
Sakr F, Alahiane S, Sennaoui A, Dinne M, Bakas I, Assabbane A (2020) Removal of cationic dye (Methylene Blue) from aqueous solution by adsorption on two type of biomaterial of South Morocco. Mater Today Proc 22: 93–96
El-Sayed GO (2011) Removal of methylene blue and crystal violet from aqueous solutions by palm kernel fiber. Desalination 272(1–3):225–232
Alizadeh A, Fakhari M, Safaei Z, Khodeai MM, Repo E, Asadi A (2020) Ionic liquid-decorated Fe3O4@SiO2 nanocomposite coated on talc sheets: An efficient adsorbent for methylene blue in aqueous solution. Inorg Chem Commun 121:108204
Badri A, Alvarez-Serrano I, Luisa López M, Ben Amara M (2020) Sol-gel synthesis, magnetic and methylene blue adsorption properties of lamellar iron monophosphate KMgFe(PO4)2. Inorg Chem Commun 121:108217.
Acknowledgments
The research work was supported by Ministry of Energy, and Mines and the Environment within the Research Project with grant number 018-1212-00019.
Author information
Authors and Affiliations
Contributions
AEHAY and AM have contributed equally to this work.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that there is no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Aoulad El Hadj Ali, Y., Ahrouch, M., Ait Lahcen, A. et al. Dried sewage sludge as an efficient adsorbent for pollutants: cationic methylene blue removal case study. Nanotechnol. Environ. Eng. 6, 17 (2021). https://doi.org/10.1007/s41204-021-00111-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s41204-021-00111-6