Abstract
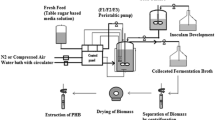
Polyhydroxyalkanoates are biodegradable polymer materials that accumulate in numerous bacteria. The polyhydroxybutyrate is the most common type of polyhydroxyalkanoates, which potentially serves as precursor for bioplastic production. The most extensively studied polyhydroxybutyrate producing bacteria is Cupriavidus necator due to its capability to accumulate large amounts of this biopolymer in simple culture medium. Accumulation of polyhydroxyalkanoates granules in the cytoplasm of C. necator significantly depended on pH, aeration, carbon sources, nitrogen sources, and minerals in the culture medium. In the present study, the effect of both nutritional and physical variables on polyhydroxybutyrate production was investigated in order to optimize these conditions. At first, on the basis of one-factor-at-a-time experiments, fructose and ammonium chloride were found to be the most suitable sources of carbon and nitrogen for biopolymer production. Then the most significant factors affecting granules accumulation were recognized as fructose, agitation speed, KH2PO4, and initial pH using the Plackett–Burman and central composite design. ANOVA analysis showed significant interaction between fructose and agitation speed. After optimization of the medium, compositions for polyhydroxybutyrate production were determined as follows: fructose 35 g/L, KH2PO4 1.75 g/L, MgSO4·7H2O 1.2 g/L, citric acid 1.7 g/L, trace element 10 mL/L, initial pH = 7, and agitation speed 175 rpm. Under this optimal culture conditions, the maximum yield of PHB was 7.48 g/L. The present strategies included in this study could be used for PHB production by this bacterium. These results are the highest values of PHB ever obtained from batch culture of C. necator reported so far.




Similar content being viewed by others
References
Anderson-Cook CM, Borror CM, Montgomery DC (2009) Response surface design evaluation and comparison. J Stat Plan Inference 139:629–641
Arun A, Murrugappan RM, Ravindran AVD, Veeramaniknandan V, Balaji S (2006) Utilization of various industrial wastes for the production of poly-b-hydroxy butyrate (PHB) by Alcaligenes eutrophus. Afr J Biotechnol 5(17):1524–1527
Beaulieu M, Beaulieu Y, Mélinard J, Pandian S, Goulet J (1995) Influence of ammonium salts and cane molasses on growth of Alcaligenes eutrophus and production of polyhydroxybutyrate. Appl Environ Microbiol 61:165–169
Bertrand JL, Ramsay BA, Ramsay JA, Chavarria C (1990) Biosynthesis of poly-β-hydroxyalkanoates from pentoses by Pseudomonas pseudoflava. Appl Environ Microbiol 56(10):3133–3138
Burdon KL (1946) Fatty materials in bacteria and fungi revealed by staining dried, fixed slide preparations. J Bacteriol 52:665–678
Cavaillé L, Grousseau E, Pocquet M, Lepeuple AS, Uribelarrea JL, Hernandez-Raquet G, Paul E (2013) Polyhydroxybutyrate production by direct use of waste activated sludge in phosphorus-limited fed-batch culture. Bioresour Technol 149:301–309
Cavalheiro JMBT, de Almeida MCMD, Grandfils C, da Fonseca MMR (2009) Poly (3-hydroxybutyrate) production by Cupriavidus necator using waste glycerol. Process Biochem 44:509–515
Chen GQ (2005) Polyhydroxyalkanoates. In: Smith RFL (ed) Biodegradable polymers for industrial applications. CRC Press, Boca Raton
Chen GQ, Qiong W (2005) The application of polyhydroxyalkanoates as tissue engineering materials. Biomaterials 26:6565–6578
Colak F, Atar N, Olgun A (2009) Biosorption of acidic dyes from aqueous solution by Paenibacillus macerans: kinetic, thermodynamic and equilibrium studies. Chem Eng J 150:122–130
Colak F, Atar N, Yazıcıoglua D, Olgun A (2011) Biosorption of lead from aqueous solutions by Bacillus strains possessing heavy-metal resistance. Chem Eng J 173:422–428
De Almeida A, Giordano AM, Nikel PI, Pettinari MJ (2010) Effects of aeration on the synthesis of poly(3-hydroxybutyrate) from glycerol and glucose in recombinant Escherichia coli. Appl Environ Microbiol 76:2036–2040
Elibol M (2004) Optimization of medium composition for actinorhodin production by Streptomyces coelicolor A3 (2) with response surface methodology. Process Biochem 39:1057–1062
Flieger M, Kantorova M, Prell A, Rezanka T, Votruba J (2003) Biodegradable plastics from renewable sources. Folia Microbiol 48(2):27–44
Frey DD, Engelhardt F, Greitzer EM (2003) A role for ‘‘one-factor-at-a-time’’ experimentation in parameter design. Res Eng Des 14:65–74
Galgani F, Leaute JP, Moguedet P, Souplet A, Verin Y, Carpentier A, Goraguer H, Latrouite D, Andral B, Cadiou Y, Mahe JC, Poulard JC, Nerisson P (2000) Litter on the sea floor along European coasts. Mar Pollut Bull 40:516–527
Galindo E, Peña C, Núñez C, Segura D, Espin G (2007) Molecular and bioengineering strategies to improve alginate and polyhydroxyalkanoate production by Azotobacter vinelandii. Microb Cell Fact 6:1–16
Gregory MR (2009) Environmental implications of plastic debris in marine settings-entanglement, ingestion, smothering, hangers-on, hitch-hiking and alien invasions. Philos Trans R Soc B 364:2013–2025
Gupta VK, Atar N, Yola ML, Darcan C, Idil O, Ustundag Z, Suhas (2013) Biosynthesis of silver nanoparticles using chitosan immobilized Bacillus cereus: nanocatalytic studies. J Mol Liq 188:81–88
Ivanov V, Stabnikov V, Ahmed Z, Dobrenko S, Saliuk A (2014) Production and applications of crude polyhydroxyalkanoate containing bioplastic from the organic fraction of municipal solid waste. Int J Environ Sci Technol 12:725–738
Jung YM, Park JS, Lee YH (2000) Metabolic transformation of cloned phbCAB genes for the investigation of the regulatory mechanism of polyhydroxyalkanoate biosynthesis. Enz Microb Technol 26:201–208
Khanna S, Srivastava AK (2005) Statistical media optimization studies for growth and PHB production by Ralstonia eutropha. Process Biochem 4:2173–2183
Khosravi Darani K, Vasheghani Farahani E, Shoja Alsadati SA (2004) Application of the Taguchi design for production of poly(b-hydroxybutyrate) by Ralstonia eutropha. Iran J Chem Chem Eng 23:131–136
Kim BS, Lee SC, Lee SY, Chang HN, Chang YK, Woo SI (1994a) Production of poly (3-hydroxybutyric- co-3-hydroxyvaleric acid) by fed-batch culture of Alcaligenes eutrophus with substrate control using online glucose analyzer. Enz Microb Technol 16:556–561
Kim BS, Lee SC, Lee SY, Chang HN, Chang YK, Woo SI (1994b) Production of poly (3-hydroxybutyric acid) by fed-batch culture of Alcaligenes eutrophus with glucose concentration control. Biotechnol Bioeng 43:892–898
Koutinas AA, Xu Y, Wang R, Webb C (2007) Polyhydroxybutyrate production from a novel feedstock derived from a wheat-based biorefinery. Enz Microb Technol 40:1035–1044
Liu JG, Xing JM, Chang TS, Ma ZY, Liu HZ (2005) Optimization of nutritional conditions for nattokinase production by Bacillus natto NLSSE using statistical experimental methods. Process Biochem 40:2757–2762
Md Din MF, Ponraj M, Van Loosdrecht M, Ujang Z, Chelliapan S, Zambare V (2014) Utilization of palm oil mill effluent for polyhydroxyalkanoate production and nutrient removal using statistical design. Int J Environ Sci Technol 11:671–684
Mu WM, Chen C, Li XF, Zhang T, Jiang B (2009) Optimization of culture medium for the production of phenyllactic acid by Lactobacillus sp. SK007. Bioresource Technol 100:1366–1370
Mumtaz T, Khanc MR, Hassana ML (2010) Study of environmental biodegradation of LDPE films in soil using optical and scanning electron microscopy. Micron 41:430–438
Owen HR, Wengerd D, Miller AR (1991) Culture medium pH is influenced by basal medium, carbohydrate source, gelling agent, activated charcoal, and medium storage method. Plant Cell Rep 10:583–586
Page WJ (1992) Production of poly-b-hydroxybutyrate by Azotobacter vinelandii UWD in media containing sugars and complex nitrogen sources. Appl Microbiol Biotechnol 38:117–121
Poirier Y (1999) Production of new polymeric compounds in plants. Plant Biotechnol 10:181–185
Raje P, Srivastava AK (1998) Poly (3-hydroxybutyrate) synthesis in fed-batch culture of Ralstonia eutropha with phosphate limitation under different glucose concentrations. Bioresour Technol 64:185–192
Repaske R (1962) Nutritional requirements for Hydrogenomonas eutropha. J Bacteriol 83:418–422
Sandhya M, Aravind J, Kanmani P (2013) Production of polyhydroxyalkanoates from Ralstonia eutropha using paddy straw as cheap substrate. Int J Environ Sci Technol 10:47–54
Shang L, Jiang M, Chang HN (2003) Poly (3-hydroxybutyrate) synthesis in fed-batch culture of Ralstonia eutropha with phosphate limitation under different glucose concentrations. Biotechnol Lett 25:1415–1419
Singh P, Parmar N (2011) Isolation and characterization of two novel polyhydroxybutyrate (PHB)-producing bacteria. Afr J Biotech 10:4907–4919
Tripathi AD, Srivastava SK, Singh RP (2013) Statistical optimization of physical process variables for bio-plastic (PHB) production by Alcaligenes sp. Biomass Bioenerg 55:243–250
Wang YW, Wu Q, Chen GQ (2005) Gelatin blending improves the performance of poly(3-hydroxybutyrate-co-3-hydroxyhexanoate) films for biomedical application. Biomacromolecules 6:566–571
Wei YH, Chen WC, Huang CK, Wu HS, Sun YM, Lo CW, Janarthanan OM (2011) Screening and evaluation of polyhydroxybutyrate producing strains from indigenous isolate Cupriavidus taiwanensis strains. Int J Mol Sci 12:252–265
Xu H, Sun LP, Shi YZ, Wu YH, Zhang B, Zhao DQ (2008) Optimization of cultivation conditions for extracellular polysaccharide and mycelium biomass by Morchella esculenta As51620. Biochem Eng J 39:66–73
Zinn M, Weilenmann H, Hany R, Schmid M, Egli T (2003) Tailored synthesis of poly-R-β-hydroxybutyrate-co-hydroxyvalerate (PHB/HV) in Ralstonia euthropha DSM 428. Acta Biotechnol 23:309–316
Acknowledgments
The authors would like to thank the research council of Malek-Ashtar University of Technology for the financial support of this investigation.
Author information
Authors and Affiliations
Corresponding author
Abbreviations
- PHA
-
Polyhydroxyalkanoate
- PHB
-
Polyhydroxybutyrate
- CCD
-
Central composite design
- RSM
-
Response surface methodology
Rights and permissions
About this article
Cite this article
Aramvash, A., Akbari Shahabi, Z., Dashti Aghjeh, S. et al. Statistical physical and nutrient optimization of bioplastic polyhydroxybutyrate production by Cupriavidus necator . Int. J. Environ. Sci. Technol. 12, 2307–2316 (2015). https://doi.org/10.1007/s13762-015-0768-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13762-015-0768-3