Abstract
Introduction
Cystoisospora belli (C. belli) is the only pathogenic species of the Cystoisospora genus responsible for severe diarrhea in immunocompromised patients. Most common microscopic method of diagnosis is less sensitive due to intermittent shedding of oocysts. We developed a new single-run polymerase chain reaction (PCR)–based diagnostic assay for C. belli.
Methods
A new single-run PCR-based diagnostic assay was standardized for the detection of C. belli. Diagnostic reproducibility and repeatability of the PCR assay were evaluated. A cross-sectional analytical study was done on a total of 354 stool samples collected from 331 immunocompromised patients with diarrhea. All the stool samples were tested for the presence of oocysts of C. belli and were also tested by our new PCR assay for C. belli. Three of the representative PCR products were confirmed by sequencing. Fisher’s exact test was used to compare the two proportions.
Results
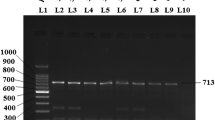
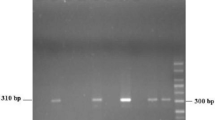
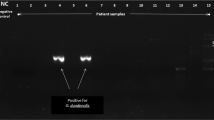
Microscopy detected C. belli in 11/354 (3.1%) of stool samples, and the new PCR-based assay detected C. belli in 16/354 (4.5%). The new single-run PCR-based assay detected C. belli in all the stool samples which were tested positive by microscopy and additionally detected C. belli in five stool samples. The developed PCR assay detected statistically significant proportion of C. belli (p < 0.001) as compared to microscopy. The 795 base pair PCR product from one microscopy positive stool sample and two microscopy negative stool samples were confirmed by sequencing.
Conclusion
Our newly developed single-run PCR-based detection assay for C. belli is robust and reproducible. It may be used for molecular diagnosis of cystoisosporiasis especially in transplant, pediatrics, and human immunodeficiency virus (HIV) positive patients.



Similar content being viewed by others
References
Marcos LA, Gotuzzo E. Intestinal protozoan infections in the immunocompromised host. Curr Opin Infect Dis. 2013;26:295–301.
Wang ZD, Liu Q, Liu HH, et al. Prevalence of Cryptosporidium, microsporidia and Isospora infection in HIV-infected people: a global systematic review and meta-analysis. Parasit Vectors. 2018;11:28.
Legua P, Seas C. Cystoisospora and Cyclospora. Curr Opin Infect Dis. 2013;26:479–83.
Dubey J, Almeria S. Cystoisospora belli infections in humans: the past 100 years. Parasitology. 2019;146:1490–527.
Garcia LS, Arrowood M, Kokoskin E, et al. Laboratory diagnosis of parasites from the gastrointestinal tract. Clin Microbiol Rev. 2018;31:e00025–17.
Mohanty I, Narasimham M, Padhi S, et al. Prevalence of isosporiasis in relation to CD4 cell counts among HIV-infected patients with diarrhea in Odisha, India. Adv Biomed Res. 2013;2:61.
Certad G, Arenas-Pinto A, Pocaterra L, et al. Isosporiasis in Venezuelan adults infected with human immunodeficiency virus: clinical characterization. Am J Trop Med Hyg. 2003;69:217–22.
Iyer RN, Rao JR, Venkatalakshmi A, Nahdi FB. Clinical and microbiology profile and outcome of diarrhea by Coccidian parasites in immunocompetent children. Pediatr Infect Dis J. 2015;34:937–9.
Yadav P, Khalil S, Mirdha BR. Molecular appraisal of intestinal parasitic infection in transplant recipients. Indian J Med Res. 2016;144:258–63.
Ahmed NH, Chowdhary A. Comparison of different methods of detection of enteric pathogenic protozoa. Indian J Med Microbiol. 2013;31:154–60.
Dryden MW, Payne PA, Ridley R, Smith V. Comparison of common fecal flotation techniques for the recovery of parasite eggs and oocysts. Vet Ther. 2005;6:15–28.
Pacheco FTF, Silva RKNR, Martins AS, et al. Differences in the detection of Cryptosporidium and Isospora (Cystoisospora) oocysts according to the fecal concentration or staining method used in a clinical laboratory. J Parasitol. 2013;99:1002–8.
Jongwutiwes S, Putaporntip C, Charoenkorn M, Iwasaki T, Endo T. Morphologic and molecular characterization of Isospora belli oocysts from patients in Thailand. Am J Trop Med Hyg. 2007;77:107–12.
Verweij JJ, Rune SC. Molecular testing for clinical diagnosis and epidemiological investigations of intestinal parasitic infections. Clin Microbiol Rev. 2014;27:371–418.
World Health Organization document. In: The management and prevention of diarrhoea: practical guidelines. World Health Organization. 1993. https://apps.who.int/iris/handle/10665/37036. Accessed 3 Feb 2021.
Jayalakshmi J, Appalaraju B, Mahadevan K. Evaluation of an enzyme-linked immunoassay for the detection of Cryptosporidium antigen in fecal specimens of HIV/AIDS patients. Indian J Pathol Microbiol. 2008;51:137–8.
Kulkarni SV, Kairon R, Sane SS, et al. Opportunistic parasitic infections in HIV/AIDS patients presenting with diarrhoea by the level of immunesuppression. Indian J Med Res. 2009;130:63–6.
Gupta K, Bala M, Deb M, Muralidhar S, Sharma DK. Prevalence of intestinal parasitic infections in HIV-infected individuals and their relationship with immune status. Indian J Med Microbiol. 2013;31:161–5.
Woon SA, Yang R, Ryan U, Boan P, Prentice D. Chronic Cystoisospora belli infection in an immunocompetent Myanmar refugee - microscopy is not sensitive enough. BMC Infect Dis. 2016;16:221.
Velásquez JN, Osvaldo GA, Di Risio C, et al. Molecular characterization of Cystoisospora belli and unizoite tissue cyst in patients with acquired immunodeficiency syndrome. Parasitology. 2011;138:279–86.
Murphy SC, Hoogestraat DR, Sengupta DJ, Prentice J, Chakrapani A, Cookson BT. Consultations in molecular diagnostics: molecular diagnosis of cystoisosporiasis using extended-range pcr screening. J Mol Diagn. 2011;13:359–62.
Taniuchi M, Verweij JJ, Sethabutr O, et al. Multiplex polymerase chain reaction method to detect Cyclospora, Cystoisospora, and Microsporidia in stool samples. Diagn Microbiol Infect Dis. 2011;71:386–90.
Acknowledgements
The authors thank Sanjay Gandhi Postgraduate Institute of Medical Sciences (SGPGIMS), Lucknow, India, for providing DNA material of C. belli for the study.
Funding
The study was supported by Intramural Research Grant from Jawaharlal Institute of Postgraduate Medical Education and Research (JIPMER), Puducherry, India.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
MK, RG, SP, NR, and RS declare that they have no conflict of interest.
Ethics statement
The authors declare that the study was performed in a manner conforming to the Helsinki declaration of 1975, as revised in 2000 and 2008 concerning human and animal rights, and the authors followed the policy concerning informed consent as shown on Springer.com.
The study was conducted after obtaining proper ethical clearance from the Institutional Ethics Committee (JIP/IEC/2016/26/839).
Disclaimer
The authors are solely responsible for the data and the content of the paper. In no way, the Honorary Editor-in-Chief, Editorial Board Members, the Indian Journal of Gastroenterology or the printer/publishers are responsible for the results/ findings and content of this article.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Katiyar, M., Gulati, R., Pagal, S. et al. Molecular detection of Cystoisospora belli by single-run polymerase chain reaction in stool samples. Indian J Gastroenterol 40, 512–518 (2021). https://doi.org/10.1007/s12664-021-01170-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12664-021-01170-y