Abstract
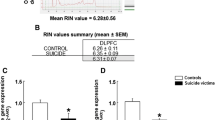
Recent studies have associated alterations of neuronal plasticity in specific brain areas with suicidal behavior. The Notch signaling pathway plays a relevant role in the control of stem cell maintenance, cell migration, and neuronal plasticity. In the present study, the gene expression of the four Notch receptors (NOTCH1–4), the five canonical ligands (DLL1, DLL3, DLL4, JAGGED1, and JAGGED2), the two non-canonical ligands (DLK1 and DLK2), and the transcription factors (HES1, HEY1, and HEY2) were measured in the dorsolateral prefrontal cortex (DLPFC) and amygdala (AMY) of suicide victims (S; n = 13 males, with no clinical psychiatric history and non-treated with anxiolytic or antidepressant drugs) and their corresponding controls (C; n = 13 males) by real-time PCR. The results revealed a reduction of NOTCH2 and NOTCH1, NOTCH3, and NOTCH4 gene expression in the DLPFC and AMY of S compared with C, respectively. DLL1 levels were increased in the DLPFC and decreased in the AMY, whereas DLL4, JAGGED1, and JAGGED2 were significantly decreased in the regions analyzed. DLK1 was reduced in the AMY, whereas no changes were observed in the DLPFC and in DLK2 expression levels in any of the regions analyzed. HES1 was significantly reduced in both brain regions from S, whereas there were no significant changes in HEY1 and HEY2. This study provides evidence suggesting that the Notch signaling pathway could be a potential key target in the treatment of suicidal behaviors.





Similar content being viewed by others
References
WHO. Suicide. Introduction. http://www.who.int/mental_health/media/en/382.pdf. 2012
Dwivedi Y, Rizavi HS, Conley RR, Roberts RC, Tamminga CA, Pandey GN (2003) Altered gene expression of brain-derived neurotrophic factor and receptor tyrosine kinase B in postmortem brain of suicide subjects. Arch Gen Psychiatry 60(8):804–815
Karege F, Vaudan G, Schwald M, Perroud N, La Harpe R (2005) Neurotrophin levels in postmortem brains of suicide victims and the effects of antemortem diagnosis and psychotropic drugs. Brain Res Mol Brain Res 136(1–2):29–37
Dwivedi Y, Mondal AC, Rizavi HS, Conley RR (2005) Suicide brain is associated with decreased expression of neurotrophins. Biol Psychiatry 58(4):315–324
Kim YK, Lee HP, Won SD, Park EY, Lee HY, Lee BH et al (2007) Low plasma BDNF is associated with suicidal behavior in major depression. Prog Neuropsychopharmacol Biol Psychiatry 31(1):78–85
Sarchiapone M, Carli V, Roy A, Iacoviello L, Cuomo C, Latella MC et al (2008) Association of polymorphism (Val66Met) of brain-derived neurotrophic factor with suicide attempts in depressed patients. Neuropsychobiology 57(3):139–145
Perroud N, Courtet P, Vincze I, Jaussent I, Jollant F, Bellivier F et al (2008) Interaction between BDNF Val66Met and childhood trauma on adult's violent suicide attempt. Genes Brain Behav 7(3):314–322
Pregelj P, Nedic G, Paska AV, Zupanc T, Nikolac M, Balazic J et al (2011) The association between brain-derived neurotrophic factor polymorphism (BDNF Val66Met) and suicide. J Affect Disord 128(3):287–290
Keller S, Sarchiapone M, Zarrilli F, Videtic A, Ferraro A, Carli V et al (2010) Increased BDNF promoter methylation in the Wernicke area of suicide subjects. Arch Gen Psychiatry 67(3):258–267
Soloff PH, Pruitt P, Sharma M, Radwan J, White R, Diwadkar VA (2012) Structural brain abnormalities and suicidal behavior in borderline personality disorder. J Psychiatr Res 46(4):516–525
Desmyter S, van Heeringen C, Audenaert K (2011) Structural and functional neuroimaging studies of the suicidal brain. Prog Neuropsychopharmacol Biol Psychiatry 35(4):796–808
Hwang JP, Lee TW, Tsai SJ, Chen TJ, Yang CH, Lirng JF et al (2010) Cortical and subcortical abnormalities in late-onset depression with history of suicide attempts investigated with MRI and voxel-based morphometry. J Geriatr Psychiatry Neurol 23(3):171–184
Imayoshi I, Kageyama R (2011) The role of Notch signaling in adult neurogenesis. Mol Neurobiol 44(1):7–12
Kageyama R, Ohtsuka T (1999) The Notch-Hes pathway in mammalian neural development. Cell Res 9(3):179–188
D'Souza B, Meloty-Kapella L, Weinmaster G (2010) Canonical and non-canonical Notch ligands. Curr Top Dev Biol 92:73–129
Kopan R (2012) Notch signaling. Cold Spring Harb Perspect Biol 4(10)
Borggrefe T, Liefke R (2012) Fine-tuning of the intracellular canonical Notch signaling pathway. Cell Cycle 11(2):264–276
Kageyama R, Ohtsuka T, Kobayashi T (2008) Roles of Hes genes in neural development. Develop Growth Differ 50(Suppl 1):S97–S103
Fischer A, Gessler M (2007) Delta-Notch–and then? Protein interactions and proposed modes of repression by Hes and Hey bHLH factors. Nucleic Acids Res 35(14):4583–4596
Sanchez-Solana B, Nueda ML, Ruvira MD, Ruiz-Hidalgo MJ, Monsalve EM, Rivero S et al (2011) The EGF-like proteins DLK1 and DLK2 function as inhibitory non-canonical ligands of NOTCH1 receptor that modulate each other's activities. Biochim Biophys Acta 1813(6):1153–1164
Arendt T (2003) Synaptic plasticity and cell cycle activation in neurons are alternative effector pathways: the 'Dr. Jekyll and Mr. Hyde concept' of Alzheimer's disease or the yin and yang of neuroplasticity. Prog Neurobiol 71(2–3):83–248
Ren X, Rizavi HS, Khan MA, Dwivedi Y, Pandey GN (2012) Altered Wnt signalling in the teenage suicide brain: focus on glycogen synthase kinase-3beta and beta-catenin. Int J Neuropsychopharmacol 1–11
Shayevitz C, Cohen OS, Faraone SV, Glatt SJ (2012) A re-review of the association between the NOTCH4 locus and schizophrenia. Am J Med Genet B Neuropsychiatr Genet 159B(5):477–483
Vermeulen J, De Preter K, Lefever S, Nuytens J, De Vloed F, Derveaux S (2012) Measurable impact of RNA quality on gene expression results from quantitative PCR. Nucleic Acids Res 39(9):e63
Klempan TA, Ernst C, Deleva V, Labonte B, Turecki G (2009) Characterization of QKI gene expression, genetics, and epigenetics in suicide victims with major depressive disorder. Biol Psychiatry 66(9):824–831
Sakamoto M, Hirata H, Ohtsuka T, Bessho Y, Kageyama R (2003) The basic helix-loop-helix genes Hesr1/Hey1 and Hesr2/Hey2 regulate maintenance of neural precursor cells in the brain. J Biol Chem 278(45):44808–44815
Dwivedi Y (2012) Brain-Derived Neurotrophic Factor in Suicide Pathophysiology. In: Dwivedi Y (ed). The Neurobiological Basis of Suicide: Boca Raton (FL)
Torres-Platas SG, Hercher C, Davoli MA, Maussion G, Labonte B, Turecki G et al (2011) Astrocytic hypertrophy in anterior cingulate white matter of depressed suicides. Neuropsychopharmacology: Off Publ Am Coll Neuropsychopharmacol 36(13):2650–2658
Dwivedi Y (2012) The Neurobiological Basis of Suicide. Chapter 8. Brain-Derived Neurotrophic Factor in Suicide Pathophysiology. CRC Press, Boca Raton
Snyder JL, Kearns CA, Appel B (2012) Fbxw7 regulates Notch to control specification of neural precursors for oligodendrocyte fate. Neural Develop 7:15
Alberi L, Liu S, Wang Y, Badie R, Smith-Hicks C, Wu J et al (2011) Activity-induced Notch signaling in neurons requires Arc/Arg3.1 and is essential for synaptic plasticity in hippocampal networks. Neuron 69(3):437–444
Ferron SR, Charalambous M, Radford E, McEwen K, Wildner H, Hind E et al (2011) Postnatal loss of Dlk1 imprinting in stem cells and niche astrocytes regulates neurogenesis. Nature 475(7356):381–385
Gomez-Duran EL, Martin-Fumado C, Hurtado-Ruiz G (2012) Clinical and epidemiological aspects of suicide in patients with schizophrenia. Actas Esp Psiquiatr 40(6):333–345
Joutel A, Corpechot C, Ducros A, Vahedi K, Chabriat H, Mouton P et al (1996) Notch3 mutations in CADASIL, a hereditary adult-onset condition causing stroke and dementia. Nature 383(6602):707–710
Arboleda-Velasquez JF, Manent J, Lee JH, Tikka S, Ospina C, Vanderburg CR et al (2011) Hypomorphic Notch 3 alleles link Notch signaling to ischemic cerebral small-vessel disease. Proc Natl Acad Sci U S A 108(21):E128–E135
Meng H, Zhang X, Yu G, Lee SJ, Chen YE, Prudovsky I et al (2012) Biochemical characterization and cellular effects of CADASIL mutants of NOTCH3. PloS one 7(9):e44964
Alasaari JS, Lagus M, Ollila HM, Toivola A, Kivimaki M, Vahtera J et al (2012) Environmental stress affects DNA methylation of a CpG rich promoter region of serotonin transporter gene in a nurse cohort. PloS one 7(9):e45813
Piazzi G, Fini L, Selgrad M, Garcia M, Daoud Y, Wex T et al (2011) Epigenetic regulation of Delta-Like1 controls Notch1 activation in gastric cancer. Oncotarget 2(12):1291–1301
Acknowledgements
This research was supported by grants from Ministry of Science and Innovation (SAF 2008–01106, RETICS RD12/0028/0019, and FEDER) and Ministry of Health (PNSD 2007/061). FN is a postdoctoral fellow of the RETICS RD12/0028/0019. We thank Analía Rico for their excellent technical assistance.
Conflict of interest
All authors state that they have no biochemical financial interest or potential conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Monsalve, E.M., García-Gutiérrez, M.S., Navarrete, F. et al. Abnormal Expression Pattern of Notch Receptors, Ligands, and Downstream Effectors in the Dorsolateral Prefrontal Cortex and Amygdala of Suicidal Victims. Mol Neurobiol 49, 957–965 (2014). https://doi.org/10.1007/s12035-013-8570-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12035-013-8570-z