Abstract
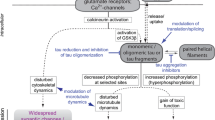
Abnormally hyperphosphorylated tau which is the major protein subunit of paired helical filaments (PHF)/neurofibrillary tangles is the pivotal lesion in Alzheimer disease (AD) and related tauopathies. The cosegregation of tau mutations with disease in inherited cases of frontotemporal dementia has confirmed that abnormalities in this protein can be a primary cause of neurodegeneration. Unlike normal tau that promotes assembly and maintains the structure of microtubules, the abnormally hyperphosphorylated protein sequesters normal tau, MAP1 and MAP2 and consequently disassembles microtubules. The abnormal hyperphosphorylation also promotes the self assembly of tau into tangles of PHF. The hyperphosphorylation of tau in AD is probably due to a protein phosphorylation/dephosphorylation imbalance produced by a decrease in the activity of protein phosphatase (PP)-2A and increase in the activities of tau kinases which are directly or indirectly regulated by PP-2A. Two of the most promising pharmacologic therapeutic approaches to AD are (1) the development of drugs that can inhibit the sequestration of normal MAPs by the abnormally hyperphosphorylated tau, and (2) the development of drugs that can reverse the abnormal hyperphosphorylation of tau by correcting the protein phosphorylation/dephosphorylation imbalance.
Similar content being viewed by others
References
Alafuzoff I., Iqbal K., Friden H., Adolfson R., and Winblad B. (1987) Histopathological criteria for progressive dementia disorders: clinical-pathological correlation and classification by multivariate data analysis. Acta Neuropathol. (Berlin) 74, 209–225.
Alonso A del C., Zaidi T., Grundke-Iqbal I., and Iqbal K. (1994) Role of abnormally phosphorylated tau in the breakdown of microtubules in Alzheimer disease. Proc. Natl. Acad. Sci. USA 91, 5562–5566.
Alonso A del C., Grundke-Iqbal I., and Iqbal K. (1996) Alzheimer’s disease hyperphosphorylated tau sequesters normal tau into tangles of filaments and disassembles microtubules. Nature Med. 2, 783–787.
Alonso A del C., Grundke-Iqbal I., Barra H. S., Iqbal K. (1997) Abnormal phosphorylation of tau and the mechanism of Alzheimer neurofibrillary degeneration: sequestration of MAP1 and MAP2 and the disassembly of microtubules by the abnormal tau. Proc. Natl. Acad. Sci. USA 94, 298–303.
Alonso A del C., Zaidi T., Novak M., Grundke-Iqbal I., Iqbal K. (2001a) Hyperphosphorylation induces self-assembly of tau into tangles of paired helical filaments/straight filaments. Proc. Natl. Acad. Sci. USA 98, 6923–6928.
Alonso A del C., Zaidi T., Wu Q., et al. (2001b) Interaction of tau isoforms with Alzheimer’s disease abnormally hyperphosphorylated tau and in vitro phosphorylation into the disease-like protein. J. Biol. Chem. 276, 37967–37973.
Arrigada P. A., Growdon J. H., Hedley-White E. T., and Hyman B. T. (1992) Neurofibrillary tangles but not senile plaques parallel duration and severity of Alzheimer’s disease. Neurology 42, 631–639.
Bancher C., Brunner C., Lassmann H., Budka H., Jellinger K., Wiche G., et al. (1989) Accumulation of abnormally phosphorylated tau precedes the formation of neurofibrillary tangles in Alzheimer’s disease. Brain Res. 477, 90–99.
Bennecib M., Gong C.-X., Grundke-Iqbal I., and Iqbal K. (2001) Inhibition of PP-2A upregulates CaMKII in rat forebrain and induces hyperphosphorylation of tau at Ser 262/356. FEBS Lett. 490, 15–22.
Cohen P., Alemany S., Hemmings B. A., Resink T. J., Stralfors P., and Lim Tung H. Y. (1988) Protein Phosphatase-1 and Protein Phosphatase-2A from rabbit skeletal muscle. Methods Enzymol. 159, 390–408.
Cohen P. (1989) The structure and regulation of protein phosphatases. Ann. Rev. Biochem. 58, 453–508.
Dickson D. W., Farlo J., Davies P., Crystal H., Fuld P., and Yen S. H. (1988) Alzheimer’s disease, A double-labeling immunohistochemical study of senile plaques. Am. J. Pathol. 132, 86–101.
Dickson D. W., Crystal H. A., Mattiace L. A., Masur D. M., Blau A. D., Davies P., et al. (1991) Identification of normal and pathological aging in prospectively studied non-demented elderly humans. Neurobiol. Aging 13, 179–189.
Finch C. and Tanzi R. E. (1997) Genetics of aging. Science 278, 407–411.
Gong C.-X., Singh T. J., Grundke-Iqbal I., and Iqbal K. (1993) Phosphoprotein phosphatase activities in Alzheimer disease brain. J. Neurochem. 61, 921–927.
Gong C.-X., Shaikh S., Wang J.-Z., Zaidi T., Grundke-Iqbal I., and Iqbal K. (1995) Phosphatase activity towards abnormally phosphorylated τ: decrease in Alzheimer disease brain. J. Neurochem. 65, 732–738.
Gong C.-X., Lidsky T., Wegiel J., Zuck L., Grundke-Iqbal I., and Iqbal K. (2000) Phosphorylation of microtubule-associated protein tau is regulated by protein phosphatase 2A in mammalian brain. J. Biol. Chem. 275, 5535–5544.
Grundke-Iqbal I., Iqbal K., Quinlan M., Tung Y.-C., Zaidi M. S., and Wisniewski H. M. (1986a) Microtubule-associated protein tau: A component of Alzheimer paired helical filaments. J. Biol. Chem. 261, 6084–6089.
Grundke-Iqbal I., Iqbal K., Tung Y.-C., Quinlan M., Wisniewski H. M., and Binder L. I. (1986b) Abnormal phosphorylation of the microtubule associated protein tau in Alzheimer cytoskeletal pathology. Proc. Natl. Acad. Sci. USA 83, 4913–4917.
Hutton M., et al. (1998) Association of missense and 5′-splice-site mutations in tau with the inherited dementia FTDP-17. Nature 393, 702–705.
Iqbal K., Grundke-Iqbal I., Zaidi T., Merz P. A., Wen G. Y., Shaikh S. S., et al. (1986) Defective brain microtubule assembly in Alzheimer’s disease. Lancet 2, 421–426.
Iqbal K., Grundke-Iqbal I., Smith A. J., George L., Tung Y. C., and Zaidi T. (1989) Identification and localization of a tau peptide to paired helical filaments of Alzheimer disease. Proc. Natl. Acad. Sci. USA 86, 5646–5650.
Iqbal K., Zaidi T., Bancher C., and Grundke-Iqbal I. (1994) Alzheimer paired helical filaments: Restoration of the biological activity by dephosphorylation. FEBS Lett. 349, 104–108.
Iqbal K. and Grundke-Iqbal I. (2000) Metabolic Hypothesis, mechanism and therapeutic targets of Alzheimer neurofibrillary degeneration. Neurosci. News 3, 14–20.
Iqbal K., Alonso A del C., Gondal J. A., Gong C.-X., Haque N., Khatoon S., et al. (2000) Mechanism of neurofibrillary degeneration and pharmacologic therapeutic approach. J. Neural Transm. 59, 213–222.
Katzman R., Terry R. D., DeTeresa R., Brown R., Davies P., Fuld P., et al. (1988) Clinical, pathological and neurochemical changes in dementia: a subgroup with preserved mental status and numerous neocortical plaques. Annals of Neurology 23, 138–144.
Khatoon S., Grundke-Iqbal I., and Iqbal K. (1992) Brain levels of microtubule-associated protein tau are elevated in Alzheimer’s disease: a radioimmuno-slot-blot assay for nanograms of the protein. Journal of Neurochemistry 59, 750–753.
Khatoon S., Grundke-Iqbal I., and Iqbal K. (1994) Levels of normal and abnormally phosphorylated tau in different cellular and regional compartments of Alzheimer disease and control brains. FEBS Lett. 351, 80–84.
Köpke E., Tung Y.-C., Shaikh S., Alonso A del C., Iqbal K., and Grundke-Iqbal I. (1993) Microtubule associated protein tau: abnormal phosphorylation of non-paired helical filament pool in Alzheimer disease. J. Biol. Chem. 268, 24374–24384.
Lee V. M.-Y., Balin B. J., Otvos L., Jr. and Trojanowski J. Q. (1991) A68: A major subunit of paired helical filaments and derivatized forms of normal tau. Science 251, 675–678.
Li M., Makkinje A., and Damuni Z. (1996a) Molecular identification of I1 PP2A, a novel potent heat-stable inhibitor protein of protein phosphatase 2A. Biochemistry 35, 6998–7002
Li M., Makkinje A., and Damuni Z. (1996b) The Myeloid Leukemia-associated protein SET is a potent inhibitor of Protein Phosphatase 2A. J. Biol. Chem. 271, 11059–11062.
Mah V. H., Eskin T. A., Kazee A. M., Lapham L., and Higgins G. A. (1992) In situ hybridization of calcium/calmodulin dependent protein kinase II and tau mRNAs; species differences and relative preservation in Alzheimer’s disease. Brain Res. Mol. Brain Res. 12, 85–94.
Morsch R., Simon W., and Coleman P. D. (1999) Neurons may live for decades with neurofibrillary tangles. J. Neuropathol. Exp. Neurol. 58, 188–197.
Oliver C. J. and Shenolikar S. (1998) Physiologic importance of protein phosphatase inhibitors. Frontiers Biosci. 3, 961–972.
Poorkaj P., Bird T. D., Wijsman E., Nemens E., Garruto R. M., Anderson L., et al. (1998) Tau is a candidate gene for chromosome 17 frontotemporal dementia. Ann. Neurol. 43, 815–825.
Spillantini M. G., Murrell J. R., Goedert M., Farlow M. R., Klug A., and Ghetti B. (1998) Mutation in the tau gene in familial multiple system tauopathy with presenile dementia. Proc. Natl. Acad. Sci. USA 95, 7737–7741.
Ulitzur N., Rancano C., and Pfeffer S. R. (1997) Biochemical characterization of mapmodulin, a protein that binds microtubule-associated proteins. J. Biol. Chem. 272, 30577–30582.
von Lindern M., van Baal S., Wiegant J., Raap A., Hagemeijer A., and Grosveld G. (1992) can, a putative oncogene associated with Myeloid Leukemogenesis, may be activated by fusion of its 3′ half to different genes: characterization of the set gene. Mol. Cell. Biol. 12, 3346–3355.
Wang J.-Z., Gong C.-X., Zaidi T., Grundke-Iqbal I., and Iqbal K. (1995) Dephosphorylation of Alzheimer paired helical filaments by protein phosphatase-2A and -2B. J. Biol. Chem. 270, 4854–4860.
Wang J.-Z., Grundke-Iqbal I., and Iqbal K. (1996) Restoration of biological activity of Alzheimer abnormally phosphorylated by dephosphorylation with protein phosphatase-2A,-2B and -1. Mol. Brain Res. 38, 200–208.
Walaas S. I. and Greengard P. (1991) Protein phosphorylation and neuronal function. Pharmacol. Rev. 43, 299–349.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Iqbal, K., Alonso, A.d.C., El-Akkad, E. et al. Significance and mechanism of alzheimer neurofibrillary degeneration and therapeutic targets to inhibit this lesion. J Mol Neurosci 19, 95–99 (2002). https://doi.org/10.1007/s12031-002-0017-3
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s12031-002-0017-3