Abstract
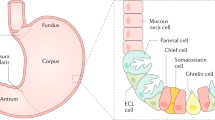
Pernicious anemia is the hematologic manifestation of chronic atrophic gastritis affecting the corpus of the stomach that denudes the gastric mucosa of gastric parietal cells. Asymptomatic autoimmune gastritis, a chronic inflammatory disease of the gastric mucosa, precedes the onset of corpus atrophy by 10–20 years. The gastritis arises from activation of pathologic Th1 CD4 T cells to gastric H/K ATPase that is normally resident on gastric mucosal secretory membranes. The onset of autoimmune gastritis is marked by circulating parietal cell antibody to gastric H/K ATPase. Gastric parietal cells produce two essential biologics: intrinsic factor and HCl acid. Pernicious anemia is a consequence of intrinsic factor loss and neutralizing intrinsic factor antibody that impairs cobalamin absorption. Acid loss leads to iron deficiency anemia that precedes cobalamin-deficient pernicious anemia by 20 years. Laboratory diagnosis rests on parietal cell antibody with or without intrinsic factor antibody, cobalamin-deficient megaloblastic anemia and elevated serum gastrin from loss of acid secretion. Autoimmune gastritis is associated with autoimmune thyroiditis and type 1 diabetes mellitus.

Similar content being viewed by others
References
Toh BH, Alderuccio F. Pernicious anaemia. Autoimmunity. 2004;37(4):357–61.
Toh BH. Diagnosis and classification of autoimmune gastritis. Autoimmun Rev. 2014;13(4–5):459–62.
Toh BH, van Driel IR, Gleeson PA. Pernicious anemia. N Engl J Med. 1997;337(20):1441–8.
Toh BH, et al. Cutting edge issues in autoimmune gastritis. Clin Rev Allergy Immunol. 2012;42(3):269–78.
Alderuccio F, et al. Animal models of human disease: experimental autoimmune gastritis–a model for autoimmune gastritis and pernicious anemia. Clin Immunol. 2002;102(1):48–58.
Biondo M, et al. Local transgenic expression of granulocyte macrophage-colony stimulating factor initiates autoimmunity. J Immunol. 2001;166(3):2090–9.
Hershko C, et al. Variable hematologic presentation of autoimmune gastritis: age-related progression from iron deficiency to cobalamin depletion. Blood. 2006;107(4):1673–9.
Lam-Tse WK, et al. The association between autoimmune thyroiditis, autoimmune gastritis and type 1 diabetes. Pediatr Endocrinol Rev PER. 2003;1(1):22–37.
Tsirogianni A, Pipi E, Soufleros K. Specificity of islet cell autoantibodies and coexistence with other organ specific autoantibodies in type 1 diabetes mellitus. Autoimmun Rev. 2009;8(8):687–91.
Ness-Abramof R, et al. Prevalence and evaluation of B12 deficiency in patients with autoimmune thyroid disease. Am J Med Sci. 2006;332(3):119–22.
Burman P, et al. A study of autoimmune gastritis in the postpartum period and at a 5-year follow-up. Gastroenterology. 1992;103(3):934–42.
Zauli D, et al. Prevalence of autoimmune atrophic gastritis in vitiligo. Digestion. 1986;34(3):169–72.
De Block CE, De Leeuw IH, Van Gaal LF. Autoimmune gastritis in type 1 diabetes: a clinically oriented review. J Clin Endocrinol Metab. 2008;93(2):363–71.
Korstanje A, et al. The serological gastric biopsy: a non-endoscopical diagnostic approach in management of the dyspeptic patient: significance for primary care based on a survey of the literature. Scand J Gastroenterol Suppl. 2002;236:22–6.
Iijima K, et al. Serum biomarker tests are useful in delineating between patients with gastric atrophy and normal, healthy stomach. World J Gastroenterol. 2009;15(7):853–9.
Korstanje A, et al. Comparison between serology and histology in the diagnosis of advanced gastric body atrophy: a study in a Dutch primary community. J Clin Gastroenterol. 2008;42(1):18–22.
Checchi S, et al. Serum ghrelin as a marker of atrophic body gastritis in patients with parietal cell antibodies. J Clin Endocrinol Metab. 2007;92(11):4346–51.
Suzuki H, et al. Plasma ghrelin concentration correlates with the levels of serum pepsinogen I and pepsinogen I/II ratio—a possible novel and non-invasive marker for gastric atrophy. Hepatogastroenterology. 2004;51(59):1249–54.
Chuang JS, et al. Diagnostic ELISA for parietal cell autoantibody using tomato lectin-purified gastric H+/K(+)-ATPase (proton pump). Autoimmunity. 1992;12(1):1–7.
Sugiu K, et al. Evaluation of an ELISA for detection of anti-parietal cell antibody. Hepatogastroenterology. 2006;53(67):11–4.
Toh BH, et al. Parietal cell antibody identified by ELISA is superior to immunofluorescence, rises with age and is associated with intrinsic factor antibody. Autoimmunity. 2012;45(7):527–32.
Lahner E, et al. Reassessment of intrinsic factor and parietal cell autoantibodies in atrophic gastritis with respect to cobalamin deficiency. Am J Gastroenterol. 2009;104(8):2071–9.
Khan S, et al. Limited value of testing for intrinsic factor antibodies with negative gastric parietal cell antibodies in pernicious anaemia. J Clin Pathol. 2009;62(5):439–41.
Tozzoli R, et al. Autoantibodies to parietal cells as predictors of atrophic body gastritis: a five-year prospective study in patients with autoimmune thyroid diseases. Autoimmun Rev. 2010;10(2):80–3.
Alonso N, et al. Serum autoimmune gastritis markers, pepsinogen I and parietal cell antibodies, in patients with type 1 diabetes mellitus: a 5-year prospective study. J Endocrinol Invest. 2011;34(5):340–4.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Toh, BH. Pathophysiology and laboratory diagnosis of pernicious anemia. Immunol Res 65, 326–330 (2017). https://doi.org/10.1007/s12026-016-8841-7
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12026-016-8841-7