Abstract
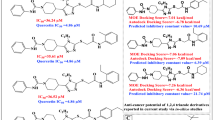
Kinesin Eg5 plays an essential role in the early stages of mitosis, and it is an interesting drug target for the design of potent inhibitors. In this work, combined molecular modeling studies of molecular docking, receptor-guided QSAR methodology, and molecular dynamics (MD) simulation were performed on a series of novel S-trityl-l-cysteine (STLC) analogues as Eg5 inhibitors to understand the structural features and key residues which are involved in the inhibition. Molecular docking study was used to find the actual conformations of STLC analogues in the binding site of Eg5. Multiple linear regression (MLR), artificial neural network (ANN), and support vector machine (SVM) models were developed by the conformation which was obtained by performing docking studies. The satisfactory result of the SVM model (R2 = 0.962, SE = 0.210, RMSE = 0.190, and Q2LOO = 0.930) demonstrated the superiority of this model over other models. Also, the satisfactory agreement between experiment and predicted inhibitory values suggested that the SVM model represents good correlation and predictive power. Molecular docking was used to study the functionalities of active molecular interaction between inhibitors and Eg5. Moreover, molecular dynamics (MD) simulation was performed on the best inhibitor-Eg5 complex to investigate the stability of docked conformation and to study the binding interactions in detail. The MD simulation result showed four hydrogen bond interactions with Eg5 residues including Gly117, Glu116, Gly117, and Glu118. The outcome of this study can be used as a guideline to better interpret the protein-ligand interaction and also can assist in the designing and development of more potent Eg5 inhibitors.










Similar content being viewed by others
References
Slangy A, Lane HA, d'Hérin P, Harper M, Kress M, Niggt EA (1995) Phosphorylation by p34cdc2 regulates spindle association of human Eg5, a kinesin-related motor essential for bipolar spindle formation in vivo. Cell 83(7):1159–1169
Sawin KE, LeGuellec K, Philippe M, Mitchison TJ (1992) Mitotic spindle organization by a plus-end-directed microtubule motor. Nature 359(6395):540–543
Weil D, Garcon L, Harper M, Dumenil D, Dautry F, Kress M (2002) Targeting the kinesin Eg5 to monitor siRNA transfection in mammalian cells. Biotechniques 33(6):1244–1248
Mayer TU, Kapoor TM, Haggarty SJ, King RW, Schreiber SL, Mitchison TJ (1999) Small molecule inhibitor of mitotic spindle bipolarity identified in a phenotype-based screen. Science 286(5441):971–974
Cox CD, Breslin MJ, Mariano BJ, Coleman PJ, Buser CA, Walsh ES, Hamilton K, Huber HE, Kohl NE, Torrent M (2005) Kinesin spindle protein (KSP) inhibitors. Part 1: the discovery of 3, 5-diaryl-4, 5-dihydropyrazoles as potent and selective inhibitors of the mitotic kinesin KSP. Bioorg Med Chem Lett 15(8):2041–2045
Cox CD, Torrent M, Breslin MJ, Mariano BJ, Whitman DB, Coleman PJ, Buser CA, Walsh ES, Hamilton K, Schaber MD (2006) Kinesin spindle protein (KSP) inhibitors. Part 4: structure-based design of 5-alkylamino-3, 5-diaryl-4, 5-dihydropyrazoles as potent, water-soluble inhibitors of the mitotic kinesin KSP. Bioorg Med Chem Lett 16(12):3175–3179
Fraley ME, Garbaccio RM, Arrington KL, Hoffman WF, Tasber ES, Coleman PJ, Buser CA, Walsh ES, Hamilton K, Fernandes C (2006) Kinesin spindle protein (KSP) inhibitors. Part 2: the design, synthesis, and characterization of 2, 4-diaryl-2, 5-dihydropyrrole inhibitors of the mitotic kinesin KSP. Bioorg Med Chem Lett 16(7):1775–1779
Roecker AJ, Coleman PJ, Mercer SP, Schreier JD, Buser CA, Walsh ES, Hamilton K, Lobell RB, Tao W, Diehl RE (2007) Kinesin spindle protein (KSP) inhibitors. Part 8: design and synthesis of 1, 4-diaryl-4, 5-dihydropyrazoles as potent inhibitors of the mitotic kinesin KSP. Bioorg Med Chem Lett 17(20):5677–5682
Brier S, Lemaire D, DeBonis S, Forest E, Kozielski F (2004) Identification of the protein binding region of S-trityl-L-cysteine, a new potent inhibitor of the mitotic kinesin Eg5. Biochemistry 43(41):13072–13082
Skoufias DA, DeBonis S, Saoudi Y, Lebeau L, Crevel I, Cross R, Wade RH, Hackney D, Kozielski F (2006) S-trityl-L-cysteine is a reversible, tight binding inhibitor of the human kinesin Eg5 that specifically blocks mitotic progression. J Biol Chem 281(26):17559–17569
Kaan HYK, Weiss J, Menger D, Ulaganathan V, Tkocz K, Laggner C, Popowycz F, Bt J, Kozielski F (2011) Structure−activity relationship and multidrug resistance study of new S-trityl-L-cysteine derivatives as inhibitors of Eg5. J Med Chem 54(6):1576–1586
Wang F, Good JA, Rath O, Kaan HYK, Sutcliffe OB, Mackay SP, Kozielski F (2012) Triphenylbutanamines: kinesin spindle protein inhibitors with in vivo antitumor activity. J Med Chem 55(4):1511–1525
Gao C, Lowndes NF, Eriksson LA (2017) Analysis of biphenyl-type inhibitors targeting the Eg5 α4/α6 allosteric pocket
Ambure P, Kar S, Roy K (2014) Pharmacophore mapping-based virtual screening followed by molecular docking studies in search of potential acetylcholinesterase inhibitors as anti-Alzheimer's agents. Biosystems 116:10–20
Balasubramanian PK, Balupuri A, Cho SJ (2016) Molecular modeling studies on series of Btk inhibitors using docking, structure-based 3D-QSAR and molecular dynamics simulation: a combined approach. Arch Pharm Res 39(3):328–339
Amini Z, Fatemi MH, Gharaghani S (2016) Hybrid docking-QSAR studies of DPP-IV inhibition activities of a series of aminomethyl-piperidones. Comput Biol Chem 64:335–345
Fatemi MH, Heidari A, Gharaghani S (2015) QSAR prediction of HIV-1 protease inhibitory activities using docking derived molecular descriptors. J Theor Biol 369:13–22
Balasubramanian PK, Balupuri A, Kang H-Y, Cho SJ (2017) Receptor-guided 3D-QSAR studies, molecular dynamics simulation and free energy calculations of Btk kinase inhibitors. BMC Syst Biol 11(2):6
Zhong H, Huang W, He G, Peng C, Wu F, Ouyang L (2013) Molecular dynamics simulation of tryptophan hydroxylase-1: binding modes and free energy analysis to phenylalanine derivative inhibitors. Int J Mol Sci 14(5):9947–9962
Jiang C, Chen Y, Wang X, You Q (2007) Docking studies on kinesin spindle protein inhibitors: an important cooperative ‘minor binding pocket’which increases the binding affinity significantly. J Mol Model 13(9):987–992
Luo X, Shu M, Wang Y, Liu J, Yang W, Lin Z (2012) 3D-QSAR studies of dihydropyrazole and dihydropyrrole derivatives as inhibitors of human mitotic kinesin Eg5 based on molecular docking. Molecules 17(2):2015–2029
Release H (2002) 7.5 for windows, molecular modeling system, Hypercube. Inc http://www hyper com
Morris GM, Huey R, Lindstrom W, Sanner MF, Belew RK, Goodsell DS, Olson AJ (2009) AutoDock4 and AutoDockTools4: automated docking with selective receptor flexibility. J Comput Chem 30(16):2785–2791
Morris GM, Goodsell DS, Halliday RS, Huey R, Hart WE, Belew RK, Olson AJ (1998) Automated docking using a Lamarckian genetic algorithm and an empirical binding free energy function. J Comput Chem 19(14):1639–1662
Morris GM, Huey R, Olson AJ (2008) Using autodock for ligand-receptor docking. Curr Protoc Bioinformatics:8.14. 11–18.14. 40
Durrant JD, McCammon JA (2011) BINANA: a novel algorithm for ligand-binding characterization. J Mol Graph Model 29(6):888–893
Katritzky AR, Lobanov VS, Karelson M, Murugan R, Grendze MP, Toomey J (1996) Comprehensive descriptors for structural and statistical analysis. 1: correlations between structure and physical properties of substituted pyridines. Rev Roum Chim 41(11–12):851–867
Garg A, Tewari R, Raghava GP (2010) K i DoQ: using docking based energy scores to develop ligand based model for predicting antibacterials. BMC bioinformatics 11(1):125
Phillips JC, Braun R, Wang W, Gumbart J, Tajkhorshid E, Villa E, Chipot C, Skeel RD, Kale L, Schulten K (2005) Scalable molecular dynamics with NAMD. J Comput Chem 26(16):1781–1802
Zoete V, Cuendet MA, Grosdidier A, Michielin O (2011) SwissParam: a fast force field generation tool for small organic molecules. J Comput Chem 32(11):2359–2368
Ryckaert J-P, Ciccotti G, Berendsen HJ (1977) Numerical integration of the cartesian equations of motion of a system with constraints: molecular dynamics of n-alkanes. J Comput Phys 23(3):327–341
Darden T, York D, Pedersen L (1993) Particle mesh Ewald: an N· log (N) method for Ewald sums in large systems. J Comput Phys 98(12):10089–10092
Roy K, Chakraborty P, Mitra I, Ojha PK, Kar S, Das RN (2013) Some case studies on application of “rm2” metrics for judging quality of quantitative structure–activity relationship predictions: emphasis on scaling of response data. J Comput Chem 34(12):1071–1082
Golbraikh A, Tropsha A (2002) Beware of q 2! J Mol Graph Model 20(4):269–276
Roy K, Mitra I, Kar S, Ojha PK, Das RN, Kabir H (2012) Comparative studies on some metrics for external validation of QSPR models. J Chem Inf Model 52(2):396–408
Minovski N, Župerl Š, Drgan V, Novič M (2013) Assessment of applicability domain for multivariate counter-propagation artificial neural network predictive models by minimum Euclidean distance space analysis: a case study. Anal Chim Acta 759:28–42
Papa E, Kovarich S, Gramatica P (2009) Development, validation and inspection of the applicability domain of QSPR models for physicochemical properties of polybrominated diphenyl ethers. Mol Inform 28(8):790–796
Tetko IV, Sushko I, Pandey AK, Zhu H, Tropsha A, Papa E, Oberg T, Todeschini R, Fourches D, Varnek A (2008) Critical assessment of QSAR models of environmental toxicity against Tetrahymena pyriformis: focusing on applicability domain and overfitting by variable selection. J Chem Inf Model 48(9):1733–1746
Gramatica P (2007) Principles of QSAR models validation: internal and external. Mol Inform 26(5):694–701
Katritzky AR, Mu L, Lobanov VS, Karelson M (1996) Correlation of boiling points with molecular structure. 1. A training set of 298 diverse organics and a test set of 9 simple inorganics. J Phys Chem 100(24):10400–10407
Papa E, Luini M, Gramatica P (2009) Quantitative structure–activity relationship modelling of oral acute toxicity and cytotoxic activity of fragrance materials in rodents. SAR QSAR Environ Res 20(7–8):767–779
Luan F, Melo A, Borges F, Cordeiro MND (2011) Affinity prediction on a 3 adenosine receptor antagonists: the chemometric approach. Bioorg Med Chem 19(22):6853–6859
Todeschini R, Consonni V (2003) DRAGON software (version 1.11-2001). Milano, Italy
Fornabaio M, Spyrakis F, Mozzarelli A, Cozzini P, Abraham DJ, Kellogg GE (2004) Simple, intuitive calculations of free energy of binding for protein−ligand complexes. 3. The free energy contribution of structural water molecules in HIV-1 protease complexes. J Med Chem 47(18):4507–4516
Ladbury JE (1996) Just add water! The effect of water on the specificity of protein-ligand binding sites and its potential application to drug design. Chem Biol 3(12):973–980
Fatemeh Mousavi MHF (2018) 3D-QSAR modeling of some S-trityl-L-cysteine analogues as inhibitors of mitotic kinesin Eg5 by CoMFA, CoMSIA and H-QSAR methodologies. Lett Drug Des Discov 15(9):979–987
Stanton DT, Jurs PC (1990) Development and use of charged partial surface area structural descriptors in computer-assisted quantitative structure-property relationship studies. Anal Chem 62(21):2323–2329
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Mousavi, S.F., Fatemi, M.H. A combination of molecular docking, receptor-guided QSAR, and molecular dynamics simulation studies of S-trityl-l-cysteine analogues as kinesin Eg5 inhibitors. Struct Chem 30, 115–126 (2019). https://doi.org/10.1007/s11224-018-1178-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11224-018-1178-1