Abstract
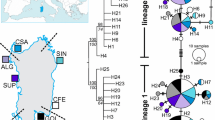
Of the more than 300 octopus species, Octopus minor is one of the most popular and economically important species in Eastern Asia, including Korea, along with O. vulgaris, O. ocellatus, and O. aegina. We developed 19 microsatellite markers from Octopus minor and eight polymorphic markers were developed to analyze the genetic diversity and relationships among four octopus populations from Korea and three from China. The number of alleles per locus varied from 10 to 49, and allelic richness per locus ranged from 2 to 16.4 across all populations. The average allele number among the populations was 11.1, with a minimum of 8.3 and a maximum of 13.6. The mean allelic richness was 8.7 in all populations. The Hardy–Weinberg equilibrium (HWE) test revealed significant deviation in 19 of the 56 single-locus sites, and null alleles were presumed in five of eight loci. The pairwise F ST values between populations from Korea and China differed significantly in all pairwise comparisons. The genetic distances between the China and Korea samples ranged from 0.161 to 0.454. The genetic distances among the populations from Korea ranged from 0.033 to 0.090, with an average of 0.062; those among populations from China ranged from 0.191 to 0.316, with an average of 0.254. The populations from Korea and China formed clearly separated into clusters via an unweighted pair group method with arithmetic mean dendrogram. Furthermore, a population from muddy flats on the western coast of the Korean Peninsula and one from a rocky area on Jeju Island formed clearly separated subclusters. An assignment test based on the allele distribution discriminated between the Korean and Chinese origins with 96.9 % accuracy.


Similar content being viewed by others
References
Norman MD, Hochberg FG (2005) The current state of Octopus taxonomy. Phuket Mar Biol Cent Res Bull 66:127–154
Roper CFE, Sweeney MJ, Nauen CE (1984) Cephalopods of the World: an annotated and illustrated catalogue of species of interest to fisheries. FAO Fish Synop 3(125):277
Kim DH, An HC, Lee KH, Hwang JW (2008) Optimal economic fishing efforts in Korean common octopus Octopus minor trap fishery. Fish Sci 74(6):1215–1221
Norman MD, Sweeney MJ (1997) The shallow-water octopuses (Cephalopoda: Octopodidae) of the Philippines. Invertebr Taxon 11(1):89–140
Thorpe JP, Solé-Cava AM, Watts PC (2000) Exploited marine invertebrates: genetics and fisheries. Hydrobiologia 420(1):165–184
Cheng R, Zheng X, Lin X, Yang J, Li Q (2011) Determination of the complete mitochondrial DNA sequence of Octopus minor. Mol Biol Rep. doi:10.1007/s11033-011-1118-2
Strugnell JM, Allcock AL, Watts PC (2009) A panel of microsatellite loci from two species of octopus, Pareledone turqueti (Joubin, 1905) and Pareledone charcoti (Joubin, 1905). Mol Ecol Resour 9(4):1239–1242
Greatorex EC, Jones CS, Murphy J, Key LN, Emery AM, Boyle PR (2000) Microsatellite markers for investigating population structure in Octopus vulgaris (Mollusca: Cephalopoda). Mol Ecol 9(5):641–642
Zuo Z, Zheng X, Yuan Y, Li Q (2011) Development and characterization of 12 polymorphicmicrosatellite loci in Octopus minor (Sasaki, 1920). Conserv Genet Resour 3:489–491
Kang JH, Yu KH, Kim SK, Park JY, Kim BS, An CM (2010) Species identification and genetic structure of Octopus minor from Korea and China on the basis of partial sequences of mitochondrial cytochrome oxidase I. Korean J Malacol 26(4):285–290
Hamilton MB, Pincus EL, Di Fiore A, Fleischer RC (1999) Universal linker and ligation procedures for construction of genomic DNA libraries enriched for microsatellites. Biotechniques 27(3):500–507
Asahida T, Kobayashi T, Saitoh K, Nakayama I (1996) Tissue preservation and total DNA extraction from fish at ambient temperature using buffers containing high concentration of urea. Fish Sci 62:727–730
Marshall TC, Slate J, Kruuk LEB, Pemberton JM (1998) Statistical confidence for likelihood-based paternity inference in natural population. Mol Ecol 7:639–655
Rice WR (1989) Analyzing tables of statistical tests. Evolution 43:223–225
Van Oosterhout C, Hutchinson WF, Wills DPM, Shipley P (2004) MICRO-CHECKER: software for identifying and correcting genotyping errors in microsatellite data. Mol Ecol Notes 4:535–538
Goudet J (2001) FSTAT, a program to estimate and test gene diversities and fixation indices (Version 2.9.3). http://www.unil.ch/izea/software/fstat.html
Weir BD, Cockerham CC (1984) Estimating F-statistics for the analysis of population structure. Evolution 38:1358–1370
Felsenstein J (1989) PHYLIP-phylogeny inference package (version 3.2). Cladistics 5:164–166
Excoffier L, Lischer HEL (2010) Arlequin suite ver 3.5: a new series of programs to perform population genetics analyses under Linux and Windows. Mol Ecol Resour 10:564–567
Piry S, Alapetite A, Cornuet JM, Paetkau D, Baudouin L, Estoup A (2004) GeneClass2: a software for genetic assignment and first-generation migrant detection. J Hered 95:536–539
Sekino M, Hara M, Taniguchi N (2002) Loss of microsatellite and mitochondrial DNA variation in hatchery trains of Japanese flounder Paralichthys olivaceus. Aquaculture 213:101–112
Kanno M, Suyama Y, Li Q, Kijima A (2006) Microsatellite analysis of Japanese sea cucumber, Stichopus (Apostichopus) japonicus, supports reproductive isolation in color variants. Mar Biotechnol 8(6):672–685
Yu H, Li Q (2007) Genetic variation of wild and hatchery populations of the Pacific oyster Crassostrea gigas assessed by microsatellite markers. J Genet Genomics 34(12):1114–1122
Hess JE, Matala AP, Narum SR (2011) Comparison of SNPs and microsatellites for fine-scale application of genetic stock identification of Chinook salmon in the Columbia River Basin. Mol Ecol Resour Suppl 1:137–149
Strugnell JM, Allcock AL, Watts PC (2009) Microsatellite loci from the endemic Southern Ocean octopus Adelieledone polymorpha (Robson, 1930). Mol Ecol Resour 9(3):1068–1070
Kang JH, Kim YK, Park JY, An CM, Nam MM, Byun SG, Lee BI, Lee JH, Choi TJ (2011) Microsatellite analysis as a tool for discriminating an interfamily hybrid between olive flounder and starry flounder. Genet Mol Res 10(4):2786–2794
Cabranes C, Fernandez-Rueda P, Martínez JL (2007) Genetic structure of Octopus vulgaris around the Iberian Peninsula and Canary Islands as indicated by microsatellite DNA variation. ICES J Mar Sci 65:12–16
Moreira AA, Tomás ARG, Hilsdorf AWS (2011) Evidence for genetic differentiation of Octopus vulgaris (Mollusca, Cephalopoda) fishery populations from the southern coast of Brazil as revealed by microsatellites. J Exp Mar Biol Ecol 407:34–40
Doubleday ZA, Semmens JM, Smolenski AJ, Shaw PW (2009) Microsatellite DNA markers and morphometrics reveal a complex population structure in a merobenthic octopus species (Octopus maorum) in south-east Australia and New Zealand. Mar Biol 156:1183–1192
Juárez OE, Rosas C, Arena L (2010) Heterologous microsatellites reveal moderate genetic structure in the Octopus maya population. Fish Res 106:209–213
Perez-Losada M, Guerra A, Carvalho GR, Sanjuan A, Shaw PW (2002) Extensive population subdivision of the cuttlefish Sepia officinalis (Mollusca: Cephalopoda) around the Iberian Peninsula indicated by microsatellite DNA variation. Heredity 89:417–424
Shaw P, Pierce J, Boyle PR (1999) Subtle population structuring within a highly vagile marine invertebrate, the veined squid Loligo forbesi, demonstrated with microsatellite DNA markers. Mol Ecol 8:407–417
Bohonak AJ (1999) Dispersal, gene flow, and population structure. Q Rev Biol 74(1):21–45
Duran S, Pascual M, Esyoup A, Turon X (2004) Strong population structure in the marine sponge Crambe crambe (Poecilosclerida) as revealed by microsatellite markers. Mol Ecol 13:511–522
Addison JA, Hart MW (2005) Spawning, copulation and inbreeding coefficients in marine invertebrates. Biol Lett 1(4):450–453
Addison JA, Hart MW (2005) Colonization, dispersal, and hybridization influence phylogeography of North Atlantic sea urchins (Strongylocentrotus droebachiensis). Evolution 59(3):532–543
Iglesias J, Otero JJ, Moxica C, Fuentes L, Sanchez FJ (2004) The completed life cycle of the octopus (Octopus vulgaris, Cuvier) under culture conditions: paralarval rearing using Artemia and zoeae, and first data on juvenile growth up to 8 months of age. Aquac Int 12:481–487
Pang IC, Kim KH (1998) Seasonal variation of water mass distributions in the eastern yellow sea and the yellow sea warm current. J Korean Soc Oceanogr 3(3):41–52
Li YL, Kong XY, Yu ZN, Kong J, Ma S, Chen LM (2009) Genetic diversity and historical demography of Chinese shrimp Feneropenaeus chinensis in Yellow Sea and Bohai Sea based on mitochondrial DNA analysis. Afr J Biotechnol 8(7):1193–1202
English LJ, Maguire GB, Ward RD (2000) Genetic variation of wild and hatchery populations of the pacific oyster, Crassostrea gigas (Thunberg) in Australia. Aquaculture 187:283–298
Pan G, Yang J (2010) Analysis of microsatellite DNA markers reveals no genetic differentiation between wild and hatchery populations of Pacific Threadfin in Hawaii. Int J Biol Sci 6(7):827–833
Norris AT, Bradley DG, Cunningham EP (1999) Microsatellite genetic variation between and within farmed and wild Atlantic salmon (Salmonsalar) populations. Aquaculture 180:247–264
Hara M, Sekino M (2007) Genetic differences between hatchery stocks and natural populations in Pacific Abalone (Haliotis discus) estimated using microsatellite DNA markers. Mar Biotech 9:74–81
Li Q, Park C, Endo T, Kijima A (2004) Loss of genetic variation at microsatellite loci in hatchery stocks of the Pacific abalone (Haliotis discus). Aquaculture 235:207–222
Acknowledgments
This work was supported by Grants from the National Fisheries Research and Development Institute (NFRDI) in Korea (RP-2012-BT-006).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kang, JH., Kim, YK., Park, JY. et al. Development of microsatellite markers to genetically differentiate populations of Octopus minor from Korea and China. Mol Biol Rep 39, 8277–8286 (2012). https://doi.org/10.1007/s11033-012-1675-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-012-1675-z