Abstract
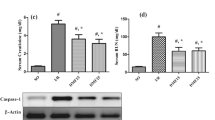
Chronic kidney disease (CKD) is a major public health problem worldwide and is associated with spatial learning deficits. The aim of the present study was to evaluate the protective effects of hydrogen sulfide (H2S) on CKD-mediated behavioral deficits with emphasis to the role of nitric oxide (NO) in these effects. Fifty rats were randomly allocated to five experimental groups including: sham, Five-sixth (5/6) nephrectomy (Nx), 5/6Nx + NaHS, 5/6Nx + NaHS+L-nitroarginine methyl ester (L-NAME), and 5/6Nx + NaHS+aminoguanidine (AMG). Twelve weeks after 5/6Nx, we evaluated proteinuria, creatinine clearance (CrCl), oxidative/antioxidant status, and hippocampus neuro-inflammation and NO synthase genes in all groups. Furthermore, training trials of all animals were conducted in the Morris water maze (MWM) task one day before animal euthanizing. As predicted, 5/6Nx induced several injuries, including enhancement of proteinuria and reduction of CCr, oxidant/antioxidant imbalance and up-regulation of TNF-α and IL-1β gene expressions in the hippocampus tissues. As predicted, 5/6Nx resulted in learning and memory impairments, and increased escape latency during acquisition trials in the MWM task. Interestingly, NaHS (H2S donor) improved behavioral deficits, renal dysfunction, accelerated anti-oxidant/anti-inflammatory responses and increased eNOS and decreased iNOS. Moreover, these effects of NaHS were prevented by L-NAME but not AMG co-administration. In conclusion, H2S ameliorates CKD-mediated brain dysfunctions, through interaction with NO signaling in the hippocampus.





Similar content being viewed by others
References
Askari H, Seifi B, Kadkhodaee M (2016) Evaluation of renal-hepatic functional indices and blood pressure based on the progress of time in a rat model of chronic kidney disease. Nephrourol Mon 8:e37840. https://doi.org/10.5812/numonthly.37840
Askari H, Rajani SF, Poorebrahim M, Haghi-Aminjan H, Raeis-Abdollahi E, Abdollahi M (2018) A glance at the therapeutic potential of irisin against diseases involving inflammation, oxidative stress, and apoptosis: an introductory review. Pharmacol Res 129:44–55. https://doi.org/10.1016/j.phrs.2018.01.012
Atef Y, El-Fayoumi HM, Abdel-Mottaleb Y, Mahmoud MF (2017) Effect of cardamonin on hepatic ischemia reperfusion induced in rats: Role of nitric oxide. Eur J Pharmacol 815:446–453. https://doi.org/10.1016/j.ejphar.2017.09.037
Baeeri M, Momtaz S, Navaei-Nigjeh M, Niaz K, Rahimifard M, Ghasemi-Niri SF, Sanadgol N, Hodjat M, Sharifzadeh M, Abdollahi M (2017) Molecular evidence on the protective effect of ellagic acid on phosalone-induced senescence in rat embryonic fibroblast cells. Food Chem Toxicol 100:8–23. https://doi.org/10.1016/j.fct.2016.12.008
Baylis C (2012) Nitric oxide synthase derangements and hypertension in kidney disease. Curr Opin Nephrol Hypertens 21:1–6. https://doi.org/10.1097/MNH.0b013e32834d54ca
Cao X, Bian JS (2016) The role of hydrogen sulfide in renal system. Front Pharmacol 7:385. https://doi.org/10.3389/fphar.2016.00385
Chillon JM, Massy ZA, Stengel B (2015) Neurological complications in chronic kidney disease patients. Nephrol Dial Transplant 31(10):1606–1614. https://doi.org/10.1093/ndt/gfv315
D'Hooge R, De Deyn PP (2001) Applications of the Morris water maze in the study of learning and memory. Brain Res Brain Res Rev 36:60–90
Drew DA, Weiner DE (2014) Cognitive impairment in CKD: keep vascular disease in mind: commentary for kidney international on ‘cognitive impairment in chronic kidney disease: prevalence, severity and association with HbA1c and fibrinogen’. Kidney Int 85:505–507. https://doi.org/10.1038/ki.2013.437
Farhad AR, Razavi SM, Rozati AR, Shekarchizade N, Manshaei M (2017) Selective nitric oxide synthase inhibitor promotes bone healing. Dent Res J 14:306–313
Gabbai FB (2001) Effects of nitric oxide synthase blockers on renal function. Nephrol Dial Transplant 16:10–13. https://doi.org/10.1093/ndt/16.suppl_1.10
Gironacci MM, Vicario A, Cerezo G, Silva MG (2018) The depressor axis of the renin-angiotensin system and brain disorders: a translational approach. Clin Sci (Lond) 132(10):1021–1038
Godinho J, de Oliveira RMW, de Sa-Nakanishi AB, Bacarin CC, Huzita CH, Longhini R, Mello JCP, Nakamura CV, Previdelli IS, Dal Molin Ribeiro MH, Milani H (2018) Ethyl-acetate fraction of Trichilia catigua restores long-term retrograde memory and reduces oxidative stress and inflammation after global cerebral ischemia in rats. Behav Brain Res 337:173–182. https://doi.org/10.1016/j.bbr.2017.08.050
Gong QH, Wang Q, Pan LL, Liu XH, Huang H, Zhu YZ (2010) Hydrogen sulfide attenuates lipopolysaccharide-induced cognitive impairment: a pro-inflammatory pathway in rats. Pharmacol Biochem Behav 96:52–58. https://doi.org/10.1016/j.pbb.2010.04.006
Gonzalez P, Machado I, Vilcaes A, Caruso C, Roth GA, Schioth H, Lasaga M, Scimonelli T (2013) Molecular mechanisms involved in interleukin 1-beta (IL-1beta)-induced memory impairment. Modulation by alpha-melanocyte-stimulating hormone (alpha-MSH). Brain Behav Immun 34:141–150. https://doi.org/10.1016/j.bbi.2013.08.007
Haghi-Aminjan H, Asghari MH, Farhood B, Rahimifard M, Hashemi Goradel N, Abdollahi M (2017a) The role of melatonin on chemotherapy-induced reproductive toxicity. J Pharm Pharmacol 70:291–306. https://doi.org/10.1111/jphp.12855
Haghi-Aminjan H, Asghari MH, Goharbari MH, Abdollahi M (2017b) A systematic review on potential mechanisms of minocycline in kidney diseases. Pharmacol Rep 69:602–609
Haruyama N, Fujisaki K, Yamato M, Eriguchi M, Noguchi H, Torisu K, Tsuruya K, Kitazono T (2014) Improvement in spatial memory dysfunction by telmisartan through reduction of brain angiotensin II and oxidative stress in experimental uremic mice. Life Sci 113:55–59. https://doi.org/10.1016/j.lfs.2014.07.032
Hu M, Zou W, Wang CY, Chen X, Tan HY, Zeng HY, Zhang P, Gu HF, Tang XQ (2016) Hydrogen sulfide protects against chronic unpredictable mild stress-induced oxidative stress in hippocampus by upregulation of BDNF-TrkB pathway. Oxid Med Cell Longev:2153745. https://doi.org/10.1155/2016/2153745
Ito H, Antoku S, Mori T, Nakagawa Y, Mizoguchi K, Matsumoto S, Omoto T, Shinozaki M, Nishio S, Abe M, Mifune M, Togane M (2017) Association between chronic kidney disease and the cognitive function in subjects without overt dementia. Clin Nephrol. https://doi.org/10.5414/cn109188
Kimura Y, Goto Y, Kimura H (2010) Hydrogen sulfide increases glutathione production and suppresses oxidative stress in mitochondria. Antioxid Redox Signal 12:1–13. https://doi.org/10.1089/ars.2008.2282
Kovalcikova A, Vavrincová-Yaghi D, Vavrinec P, Tothova L, Boor P, Sebekova K, Celec P (2018) Oxidative stress in the brain caused by acute kidney injury. Metab Brain Dis 33(3):961–967. https://doi.org/10.1007/s11011-018-0204-8
Koylu EO, Kanit L, Taskiran D, Dagci T, Balkan B, Pogun S (2005) Effects of nitric oxide synthase inhibition on spatial discrimination learning and central DA2 and mACh receptors. Pharmacol Biochem Behav 81:32–40. https://doi.org/10.1016/j.pbb.2005.02.008
Kurella Tamura M, Tam K, Vittinghoff E, Raj D, Sozio SM, Rosas SE, Makos G, Lora C, He J, Go AS, Hsu CY, Yaffe K (2017) Inflammatory markers and risk for cognitive decline in chronic kidney disease: The CRIC study. Kidney Int Rep 2:192–200. https://doi.org/10.1016/j.ekir.2016.10.007
Lanzetti M, da Costa CA, Nesi RT, Barroso MV, Martins V, Victoni T, Lagente V, Pires KM, e Silva PM, Resende AC, Porto LC, Benjamim CF, Valenca SS (2012) Oxidative stress and nitrosative stress are involved in different stages of proteolytic pulmonary emphysema. Free Radic Biol Med 53:1993–2001. https://doi.org/10.1016/j.freeradbiomed.2012.09.015
Li P, Ma LL, Xie RJ, Xie YS, Wei RB, Yin M, Wang JZ, Chen XM (2012) Treatment of 5/6 nephrectomy rats with sulodexide: a novel therapy for chronic renal failure. Acta Pharmacol Sin 33:644–651. https://doi.org/10.1038/aps.2012.2
Liu H, Deng Y, Gao J, Liu Y, Li W, Shi J, Gong Q (2015) Sodium hydrosulfide attenuates beta-amyloid-induced cognitive deficits and neuroinflammation via modulation of MAPK/NF-kappaB pathway in rats. Curr Alzheimer Res 12:673–683
Liu C, Xu X, Gao J, Zhang T, Yang Z (2016) Hydrogen sulfide prevents synaptic plasticity from VD-induced damage via Akt/GSK-3beta pathway and notch signaling pathway in rats. Mol Neurobiol 53:4159–4172. https://doi.org/10.1007/s12035-015-9324-x
Lv Z, Gao J, Wang L, Chen Z, Yuan H, Ma X, Lu J, Lv J, Wu X, Zhang L, Wei L, Xue R, Fu R, Ma L (2016) Uremia-caused changes of ghrelin system in hippocampus may be associated with impaired cognitive function of hippocampus. Int Urol Nephrol 48:807–815. https://doi.org/10.1007/s11255-016-1228-9
Mard SA, Askari H, Neisi N, Veisi A (2014a) Antisecretory effect of hydrogen sulfide on gastric acid secretion and the involvement of nitric oxide. Biomed Res Int 2014:480921. https://doi.org/10.1155/2014/480921
Mard SA, Maleki M, Askari H, Gharib Naseri M (2014b) Inhibition of histamine-stimulated gastric acid secretion by hydrogen sulfide in male rats. Adaptive Med 6:157–160
Mehrabi S, Sanadgol N, Barati M, Shahbazi A, Vahabzadeh G, Barzroudi M, Seifi M, Gholipourmalekabadi M, Golab F (2018) Evaluation of metformin effects in the chronic phase of spontaneous seizures in pilocarpine model of temporal lobe epilepsy. Metab Brain Dis 33(1):107–114. https://doi.org/10.1007/s11011-017-0132-z
Mutlu O, Ulak G, Belzung C (2011) Effects of nitric oxide synthase inhibitors 1-(2-trifluoromethylphenyl)--imidazole (TRIM) and 7-nitroindazole (7-NI) on learning and memory in mice. Fundam Clin Pharmacol 25:368–377. https://doi.org/10.1111/j.1472-8206.2010.00851.x
Najafi S, Payandemehr B, Tabrizian K, Shariatpanahi M, Nassireslami E, Azami K, Mohammadi M, Asadi F, Roghani A, Sharifzadeh M (2013) The role of nitric oxide in the PKA inhibitor induced spatial memory deficits in rat: involvement of choline acetyltransferase. Eur J Pharmacol 714:478–485. https://doi.org/10.1016/j.ejphar.2013.06.039
Nicolle MM, Gonzalez J, Sugaya K, Baskerville KA, Bryan D, Lund K, Gallagher M, McKinney M (2001) Signatures of hippocampal oxidative stress in aged spatial learning-impaired rodents. Neuroscience 107:415–431
Ruebner RL, Laney N, Kim JY, Hartung EA, Hooper SR, Radcliffe J, Furth SL (2016) Neurocognitive dysfunction in children, adolescents, and young adults with CKD. Am J Kidney Dis 67:567–575. https://doi.org/10.1053/j.ajkd.2015.08.025
Sanadgol N, Golab F, Mostafaie A, Mehdizadeh M, Abdollahi M, Sharifzadeh M, Ravan H (2016) Ellagic acid ameliorates cuprizone-induced acute CNS inflammation via restriction of microgliosis and down-regulation of CCL2 and CCL3 pro-inflammatory chemokines. Cell Mol Biol (Noisy-le-grand) 62:24–30. https://doi.org/10.14715/cmb/2016.62.12.5
Sanadgol N, Golab F, Askari H, Moradi F, Ajdary M, Mehdizadeh M (2018) Alpha-lipoic acid mitigates toxic-induced demyelination in the corpus callosum by lessening of oxidative stress and stimulation of polydendrocytes proliferation. Metab Brain Dis 33(1):27–37. https://doi.org/10.1007/s11011-017-0099-9
Shefa U, Yeo SG, Kim MS, Song IO, Jung J, Jeong NY, Huh Y (2017) Role of Gasotransmitters in oxidative stresses, neuroinflammation, and neuronal repair Biomed Res Int 2017:1689341 doi:https://doi.org/10.1155/2017/1689341
Shokrzadeh M, Ahmadi A, Chabra A, Naghshvar F, Salehi F, Habibi E, Haghi-Aminjan H (2014) An ethanol extract of Origanum vulgare attenuates cyclophosphamide-induced pulmonary injury and oxidative lung damage in mice. Pharm Biol 52:1229–1236. https://doi.org/10.3109/13880209.2013.879908
Tanada Y, Okuda J, Kato T, Minamino-Muta E, Murata I, Soga T, Shioi T, Kimura T (2017) The metabolic profile of a rat model of chronic kidney disease. Peer J 5:e3352
Tian JS, Zhai QJ, Zhao Y, Chen R, Zhao LD (2017) 2-(2-benzofuranyl)-2-imidazoline (2-BFI) improved the impairments in AD rat models by inhibiting oxidative stress, inflammation and apoptosis. J Integr Neurosci 16:385–400. https://doi.org/10.3233/jin-170032
Tsuruya K (2014) Brain atrophy and cognitive impairment in patients with chronic kidney disease. Nihon Rinsho 72:708–714
Tu DG, Chang YL, Chou CH, Lin YL, Chiang CC, Chang YY, Chen YC (2017) Preventive effects of taurine against d-galactose-induced cognitive dysfunction and brain damage. Food Funct. https://doi.org/10.1039/c7fo01210a
Wang H, Peng RY (2016) Basic roles of key molecules connected with NMDAR signaling pathway on regulating learning and memory and synaptic plasticity. Mil Med Res 3:26. https://doi.org/10.1186/s40779-016-0095-0
Wei J, Zhang J, Wang L, Cha BJ, Jiang S, Liu R (2018) A new low-nephron CKD model with hypertension, progressive decline of renal function, and enhanced inflammation in C57BL/6 mice. Am J Physiol Renal Physiol 314(5). https://doi.org/10.1152/ajprenal.00574.2017
Wu J, Zhao YM, Deng ZK (2017) Tangeretin ameliorates renal failure via regulating oxidative stress, NF-kappaB-TNF-alpha/iNOS signalling and improves memory and cognitive deficits in 5/6 nephrectomized rats. Inflammopharmacology. https://doi.org/10.1007/s10787-017-0394-4
Zhou XY, Zhang F, Ying CJ, Chen J, Chen L, Dong J, Shi Y, Tang M, Hu XT, Pan ZH, Xu NN, Zheng KY, Tang RX, Song YJ (2017) Inhibition of iNOS alleviates cognitive deficits and depression in diabetic mice through downregulating the NO/sGC/cGMP/PKG signal pathway. Behav Brain Res 322(Pt A):70–82. https://doi.org/10.1016/j.bbr.2016.12.046
Author information
Authors and Affiliations
Contributions
Askari H. and Sanadgol N. contributed to the conception, study design and manuscript preparation. Nouri M., Mirfallah R., Tahmasbi N., Abedi M. and Salarian A. did the data collection. Fatima and MF. Abazari contributed to the critical revision of the article. Askari H. and Belaran M. did the analysis and interpretation of data. Ghoraeian P. and Torabinejad S. assisted on western blot analysis.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Disclosure
This study partially supported by University of Zabol (grant code: UOZ-GR-9517-13).
Rights and permissions
About this article
Cite this article
Askari, H., Abazari, M.F., Ghoraeian, P. et al. Ameliorative effects of hydrogen sulfide (NaHS) on chronic kidney disease-induced brain dysfunction in rats: implication on role of nitric oxide (NO) signaling. Metab Brain Dis 33, 1945–1954 (2018). https://doi.org/10.1007/s11011-018-0301-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11011-018-0301-8