Abstract
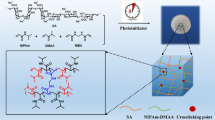
Dual-responsive hydrogel composites, hydrocomposites (HC), composed of magnetite particles and alginate, which undergo drastic changes in volume in response to pH and magnetic fields, were prepared and employed for the removal of methylene blue (MB) from the aqueous environment. The use of magnets allowed for the control of the MB absorption mechanism and the removal of HCs following their application in water, thereby preventing secondary contamination. The HC capacity for water absorption was found to be dependent on the amount of magnetite and could be altered with the application of a magnetic field. In a 25 mg/L MB solution at pH 6.3 and 8, 20 mg of HC removed 97.7% of MB. The presence of 1% magnetite (w/w) is sufficient for composites to be guided by magnetic stimuli, and the resulting material could be reused at least four times through a simple treatment process.
Graphic Abstract





Similar content being viewed by others
Data Availability
All data generated or analyzed during this study are included in this published article and its supplementary information files.
References
Ahmed EM (2015) Hydrogel: preparation, characterization and applications : a review. J Adv Res 6:105–121. https://doi.org/10.1016/j.jare.2013.07.006
Sampatrao V, Vyankatrao A, Jacky R (2016) Citric acid crosslinked cyclodextrin/hydroxypropylmethylcellulose hydrogel films for hydrophobic drug delivery. Int J Biol Macromol 93:75–86. https://doi.org/10.1016/j.ijbiomac.2016.08.072
Peppas NA, Bures P, Leobandung W, Ichikawa H (2000) Hydrogels in pharmaceutical formulations. Eur J Pharm Biopharm 50:24–46
Hoffman AS (2012) Hydrogels for biomedical applications. Adv Drug Deliv Rev 64:18–23. https://doi.org/10.1016/j.addr.2012.09.010
Gupta P, Vermani K, Garg S (2002) Hydrogels : from controlled release to pH-responsive drug delivery. Drug Discov Today 7:569–579
De Oliveira KAL, Sitta DLA, Guilherme MR, Muniz EC, Rubira AF (2017) Design of pH-responsive albumin-alginate hydrogels for drug delivery. J Control Release 259:e5–e195. https://doi.org/10.1016/j.jconrel.2017.03.043
Lima DS, Tenório-neto ET, Lima-tenório MK, Guilherme MR, Scariot DB, Nakamura CV, Muniz EC, Rubira AF (2018) pH-responsive alginate-based hydrogels for protein delivery. J Mol Liq 262:29–36. https://doi.org/10.1016/j.molliq.2018.04.002
Lima-tenório MK, Tenório-neto ET, Guilherme MR, Garcia FP, Nakamura CV, Pineda EAG, Rubira AF (2015) Water transport properties through starch-based hydrogel nanocomposites responding to both pH and a remote magnetic field. Chem Eng J 259:620–629. https://doi.org/10.1016/j.cej.2014.08.045
Okay O (2000) Macroporous copolymer networks. Prog Polym Sci 25:711
Kuang J, Yuk KY, Huh KM (2011) Polysaccharide-based superporous hydrogels with fast swelling and superabsorbent properties. Carbohydr Polym 83:284–290. https://doi.org/10.1016/j.carbpol.2010.07.052
Pourjavadi A, Zeidabadi F, Barzegar S (2010) Alginate-based biodegradable superabsorbents as candidates for diclofenac sodium delivery systems. J Appl Polym Sci 118:2015–2023. https://doi.org/10.1002/app
Chandrawati R (2016) Minireview enzyme-responsive polymer hydrogels for therapeutic delivery. Exp Biol Med. https://doi.org/10.1177/1535370216647186
Gombotz WR, Wee SF (1998) Protein release from alginate matrices. Adv Drug Deliv Rev 31:267–285
Draget KI (2016) Alginates: fundamental properties and food applications. Ref Modul Food Sci. https://doi.org/10.1016/B978-0-08-100596-5.03182-6
Byrom D (1991) Biomaterials: novel materials from biological sources. Palgrave Macmillan, New York
De Lima HHC, Kupfer VL, Moisés MP, Guilherme MR (2018) Bionanocomposites based on mesoporous silica and alginate for enhanced drug delivery. Carbohydr Polym 196:126–134. https://doi.org/10.1016/j.carbpol.2018.04.107
Panão CO, Campos ELS, Lima HHC, Rinaldi AW, Lima-tenório MK, Tenório-neto ET, Guilherme MR, Asefa T, Rubira AF (2019) Ultra-absorbent hybrid hydrogel based on alginate and SiO 2 microspheres: a high-water-content system for removal of methylene blue. J Mol Liq 276:204–213. https://doi.org/10.1016/j.molliq.2018.11.157
Ito A, Shinkai M, Honda H, Kobayashi T (2005) Medical application of functionalized magnetic nanoparticles. J Biosci Bioeng 100:1–11. https://doi.org/10.1263/jbb.100.1
Gao Y, Liu Y, Xu C (2014) Magnetic Nanoparticles for Biomedical Applications : From Diagnosis to Treatment to Regeneration. London. https://doi.org/10.1007/978-1-4471-4372-7
Lu A, Salabas EL, Schüth F (2007) Magnetic nanoparticles : synthesis, protection, functionalization, and application. Angew Chem Int Ed 46:1222–1244. https://doi.org/10.1002/anie.200602866
Hu X, Nian G, Liang X, Wu L, Yin T, Lu H, Qu S, Yang W (2019) Adhesive tough magnetic hydrogels with high Fe3O4 content. Appl Mater Interfaces 11:10292–10300. https://doi.org/10.1021/acsami.8b20937
Li Y, Huang G, Zhang X, Li B, Chen Y, Lu T (2012) Magnetic hydrogels and their potential biomedical applications. Adv Funct Mater 23:660–672. https://doi.org/10.1002/adfm.201201708
Wang ZJ, Zhu CN, Hong W, Wu ZL, Zheng Q (2017) Cooperative deformations of periodically patterned hydrogels. Sci Adv 3:1–8
Wu ZL, Moshe M, Greener J, Therien-aubin H, Nie Z, Sharon E, Kumacheva E (2013) Three-dimensional shape transformations of hydrogel sheets induced by small-scale modulation of internal stresses. Nat Commun 4:1–7. https://doi.org/10.1038/ncomms2549
Mu K, Mohr H (1998) Hepatitis C and human immunodeficiency virus RNA degradation by methylene blue/light treatment of human plasma. J Med Virol 56:239–245
Tretter L, Koml T (2017) Neuropharmacology Methylene blue stimulates substrate-level phosphorylation catalysed by succinyl-CoA ligase in the citric acid cycle. Neuropharmacology 2:1–12. https://doi.org/10.1016/j.neuropharm.2017.05.009
Ferreira-strixino J (2017) Evaluation of methylene blue as photosensitizer in promastigotes of Leishmania major and Leishmania braziliensis. Photodiagn Photodyn Ther 18:325–330. https://doi.org/10.1016/j.pdpdt.2017.04.009
Aspiroz C, Sevil M, Toyas C, Gilaberte Y (2017) Photodynamic therapy with methylene blue for skin ulcers infected with pseudomonas aeruginosa and Fusarium spp. Acta Dermosifiliogr 108:e45–e48. https://doi.org/10.1016/j.adengl.2017.05.010
Ozal E, Kuralay E, Yildirim V, Kilic S, Bolcal C, Kucukarslan N, Gunay C, Demirkilic U, Tatar H (2005) Preoperative methylene blue administration in patients at high risk for vasoplegic syndrome. Ann Thorac Surg 79:1615–1619. https://doi.org/10.1016/j.athoracsur.2004.10.038
Leyh RG, Kofidis T, Struber M, Fischer S, Knobloch K, Hagl C, Simon AR, Haverich A (2003) Methylene blue: the drug of choice for catecholamine- refractory vasoplegia after cardiopulmonary bypass? J Thorac Cardiovasc Surg 125:1426–1431. https://doi.org/10.1016/S0022-5223(02)73284-4
Cowett RM, Hakanson DO, Ocon RW, Oh W (1976) Untoward neonatal effect of intraamniotic administration of methylene blue. Obstet Gynecol 48:74S-75S
Crooks J (1982) Haemolytic jaundice in methylene blue after intra-amniotic injection of methylene blue. Arch Dis Child 57:872–873. https://doi.org/10.1136/adc.57.11.872
Fish WH, Chazen EM (1992) Toxic effects of methylene blue on the fetus. Arch Dis Child 142:1412–1413
Plunkett GD (1973) Neonatal complications. Obstet Gynecol Int J 41:476–477
Porat R, Gilbert S, Magilner D (1996) Methylene blue-induced phototoxicity: an unrecognized complication. Pediatrics 97:717–721
Serota FT, Bernbaum JC, Schwartz E (1979) The methylene-blue baby. Lancet 2:1142–1143
Bi T (1989) The methylene blue baby. N Engl J Med 320:1756–1757
Vincer MJ, Allen AC, Evans JR, Nwaesei C, Stinson DA (1987) Methylene-blue-induced hemolytic anemia in a neonate. Can Med Assoc J 136:503–504
Gillaman PK (2006) Methylene blue implicated in potentially fatal serotonin toxicity. Anaesthesia 61:1007–1019
Dewachter P, Mouton-Faivre C, Tréchot P, Lleu J-C, Mertes PM (2005) Severe anaphylactic shock with methylene blue instillation. Anesth Analg. https://doi.org/10.1213/01.ANE.0000153497.60047.80
Van Diepen S, Sobey A, Lewanczuk R, Mullen JC (2013) A case of acute respiratory distress syndrome responsive to methylene blue during a carcinoid crisis. Can J Anesth 60:1085–1088. https://doi.org/10.1007/s12630-013-0026-4
Chatterjee S, Chatterjee S, Chatterjee BP, Guha AK (2007) Adsorptive removal of congo red, a carcinogenic textile dye by chitosan hydrobeads: binding mechanism, equilibrium and kinetics. Colloids Surf A Biointerfaces 299:146–152. https://doi.org/10.1016/j.colsurfa.2006.11.036
Mittal A, Mittal J, Malviya A, Kaur D, Gupta VK (2010) Adsorption of hazardous dye crystal violet from wastewater by waste materials. J Colloid Interface Sci 343:463–473. https://doi.org/10.1016/j.jcis.2009.11.060
Korsmeyer RW, Lustig SR, Nikolaos A (1986) Solute and penetrant diffusion in swellable polymers. I. Mathematical modeling. J Polym Sci Polym Phys Ed 24:395–408
Lobo MS, Costa P (2001) Modeling and comparison of dissolution profiles. Eur J Pharm Sci 13:123–133
Tang J, Tong Z, Xia Y, Liu M, Lv Z, Gao Y, Lu T, Xie S, Pei Y, Fangb D, Wang TJ (2018) Despite, Super tough magnetic hydrogels for remotely triggered shape morphing. J Mater Chem B 6:2713–2722. https://doi.org/10.1039/C8TB00568K
Copello GJ, Mebert AM, Raineri M, Pesenti MP, Diaz LE (2011) Removal of dyes from water using chitosan hydrogel/SiO2 and chitin hydrogel/SiO2 hybrid materials obtained by the sol – gel method. J Hazard Mater 186:932–939. https://doi.org/10.1016/j.jhazmat.2010.11.097
Acknowledgements
The authors acknowledge the funding through Coordenação de Aperfeiçoamento Pessoal de Nível Superior (CAPES)—Brazil and Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) – Brazil for the fellowships. A.F.R and M.R.G acknowledge the financial supports given by CNPq and ICETI, respectively.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Costa, P.G., Lima-Tenório, M.K., Tenório-Neto, E.T. et al. Synthesis of Dual-Responsive Alginate-Inspired Hydrocomposites for the Absorption of Blue Methylene. J Polym Environ 29, 1643–1650 (2021). https://doi.org/10.1007/s10924-020-01988-1
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10924-020-01988-1