Abstract
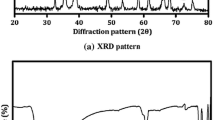
Cu2SnS3 (CTS) nanocrystallites were successfully obtained through a facile solvothermal technique at 140 and 200 °C for 24 h. In this work, the effect of temperature on structural, morphological and optical characteristics was investigated. The X-ray diffraction patterns of the samples proved the polycrystalline nature and the formation of pure cubic Cu2SnS3 structure with (111) preferential orientation. No peak referring to other binary or ternary phases were detected in the patterns. We found that by increasing the temperature, the average crystallite sizes calculated using the Debye–Scherrer formula increased from 10 to 23 nm and the direct optical band gap increased from 1.30 to 1.44 eV. The Magnetic measurements at room temperature reveal paramagnetic behavior. In this study, Cu2SnS3 nanocrystals were prepared through an environmental-friendly facile solvothermal method. It was found that the crystal structure and morphology of the productions were strongly influenced by the reaction conditions including temperature. In our case, the temperature seems to affect the quality of the nanoparticles and may be the cause of the presence of such forms. At increasing more particles on the surface may agglomerate to form bigger grains.









Similar content being viewed by others
References
Z.G. Li, A.L.K. Lui, K.H. Lam, L.F. Xi, Y.M. Lam, Inorg. Chem. 53, 10874–10880 (2014)
J.C. Zhou, L. You, S.W. Li, Y.L. Yang, Mater. Lett. 81, 248–250 (2012)
Z. Seboui, A. Gassoumi, N.K. Turki, Mater. Sci. Semicond. Process. 26, 360–366 (2014)
A. Tombak, Y.S. Ocak, M.F. Genişel, T. Kilicoglu, Mater. Sci. Semicond. Process. 28, 98–102 (2014)
K. Jimbo, R. Kimura, T. Kamimura, S. Yamada, W.S. Maw, H. Araki, K. Oishi, H. Katagiri, Thin Solid Films 515, 5997–5999 (2007)
H. Katagiri, Thin Solid Films 480, 426–432 (2005)
H. Katagiri, K. Saitoh, T. Washio, H. Shinohara, T. Kurumadani, S. Miyajima, Sol. Energy Mater. Sol. Cells 65, 141–148 (2001)
M. Bouaziz, M. Amlouk, S. Belgacem, Thin Solid Films 517, 2527–2530 (2009)
S.W. Shin, J.H. Han, Y.C. Park, G.L. Agawane, C.H. Jeong, J.H. Yun, A.V. Moholkar, J.Y. Lee, J.H. Kim, J. Mater. Chem. 22, 21727–21732 (2012)
R.A. Wibowo, W.S. Kim, E.S. Lee, B. Munir, K.H. Kim, J. Phys. Chem. Solids 68, 1908–1913 (2007)
L.J. Chen, Y.J. Chuang, J. Cryst. Growth 376, 11–16 (2013)
K. Tanaka, Y. Fukui, N. Moritake, H. Uchiki, Sol. Energy Mater. Sol. Cells 95, 838–842 (2011)
N. Kamoun, H. Bouzouita, B. Rezig, Thin Solid Films 515, 5949–5952 (2007)
M. Bouaziz, J. Ouerfelli, M. Amlouk, S. Belgacem, Phys. Status Solidi A 204, 3354–3360 (2007)
M. Adelifard, M.M.B. Mohagheghi, H. Eshghi, Phys. Scr. 85, 035603–035608 (2012)
U. Chalapathi, Y. Jayasree, S. Uthanna, V. Sundara Raja, Phys. Status Solidi A 210, 2384–2390 (2013)
C.P. Chan, H. Lam, C. Surya, Sol. Energy Mater. Sol. Cells 94, 207–211 (2010)
Q.M. Chen, X.M. Dou, Z.Q. Li, S.Y. Cheng, S.L. Zhuang, Adv. Mater. Res. 335, 1406–1411 (2011)
Q. Guo, H.W. Hillhouse, R.J. Agrawal, Am. Chem. Soc. 131, 11672–11673 (2009)
Y. Zhou, W. Zhou, Y. Du, M. Li, S. Wu, Mater. Lett. 65, 1535–1537 (2011)
Y. Wang, H.J. Gong, Alloys Compd. 509, 9627–9630 (2011)
K. Woo, Y. Kim, J. Moon, Energy Environ. Sci. 5, 5340–5345 (2012)
S.A. Vanalakar, G.L. Agawane, S.W. Shin, H.S. Yang, P.S. Patil, J.Y. Kim, J.H. Kim, Acta Mater. 85, 314–321 (2015)
S.A. Vanalakar, S.J. Yeo, P.S. Patil, J.Y. Kim, J.H. Kim, Z. Phys. Chem. 228, 917–926 (2014)
S.A. Vanalakar, R.C. Pawar, M.P. Suryawanshi, S.S. Mali, D.S. Dalavi, A.V. Moholkar, Mater. Lett. 65, 548–551 (2011)
S. Rabaoui, H. Dahman, N. Ben Mansour, L. El Mir, J. Mater. Sci.: Mater. Electron. 26, 1119–1124 (2015)
X. Liang, Q. Cai, W. Xiang, Z. Chen, J. Zhong, Y. Wang, M. Shao, Z. Li, J. Mater. Sci. Technol. 29, 231–236 (2013)
H. Saeki, H. Tabata, T. Kawai, Solid State Commun. 120, 439–443 (2001)
G.K. Williamson, W.H. Hall, Acta Metall. 1, 22–31 (1953)
P.A. Fernandes, P.M.P. Salome, A.F. da Cunha, J. Phys. D Appl. Phys. 43, 215403–215414 (2010)
B. Li, Y. Xie, J.X. Huang, Y.T. Qian, J. Solid State Chem. 153, 170–173 (2000)
B. Rezig, H. Dahman, M. Kenzari, Renew. Energy 2, 125–128 (1992)
H. Dahman, M. Khalifa, B. Rezig, M. Brunel, Thin Solid Films 280, 56–60 (1996)
G. Moh, Chemie der Erde 34, 1–61 (1975)
P.A. Fernandes, P.M.P. Salome, A.F. da Cunha, Phys. Status Solidi 7, 901–904 (2010)
H. Hahn, H. Schulze, Naturwiss 52, 426 (1965)
N. Wang, N. Jahrb, Mineral 241, 424–431 (1974)
M. Onoda, X.A. Chen, A. Sato, H. Wada, Mater. Res. Bull. 35, 1563–1570 (2000)
X. Chen, X. Wang, C. An, J. Liu, Y. Qian, J. Cryst. Growth 256, 368–376 (2003)
D. Avallaneda, M.T.S. Nair, P.K. Nair, J. Electrochem. Soc. 187, 346–352 (2010)
S. Kahraman, S. Çetinkaya, S. Yaşar, İ. Bilican, Philos. Mag. 94, 3149–3161 (2014)
S. Yaşar, S. Kahraman, S. Çetinkaya, I. Bilican, J. Alloys Compd. 618, 217–221 (2015)
C.H.L. Weijtens, P.A.C. Vanloon, Thin Solid Films 196, 1–10 (1991)
D. Tiwari, T.K. Chaudhuri, T. Shripathi, U. Deshpande, R. Rawat, Sol. Energy Mater. Sol. Cells 113, 165–170 (2013)
B.H. Liu, J. Ding, Appl. Phys. Lett. 88, 042506–042508 (2006)
B.H. Liu, J. Ding, Z.L. Dong, C.B. Boothroyd, J.H. Yin, J.B. Yi, Phys. Rev. B 74, 184427–184436 (2006)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Rabaoui, S., Dahman, H., Omri, K. et al. Controlled solvothermal synthesis and properties of Cu2SnS3 nanoparticles. J Mater Sci: Mater Electron 28, 3090–3097 (2017). https://doi.org/10.1007/s10854-016-5897-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10854-016-5897-z