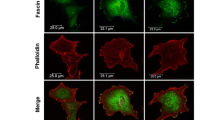
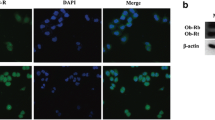
Abstract
Epidemiological and animal studies suggest an association between dietary fatty acids and an increase risk of developing breast cancer. Obesity, which is characterized by hyperlipidemia and an elevation of circulating free fatty acids (FFAs), is also associated with enhanced cancer risk. In breast cancer cells, the FFA oleic acid (OA) induces migration, proliferation, prolong survival, invasion, an increase in cellular Ca2+ concentration, MEK1/2, ERK1/2, FAK and Src activation. However, the role of OA on MMP-9 secretion and invasion has not been studied in detail. We demonstrate here that stimulation of MDA-MB-231 breast cancer cells with 200 μM OA induces an increase on MMP-9 secretion through a PKC, Src, and EGFR-dependent pathway, as revealed by gelatin zymography assays. Furthermore, microtubule network mediates MMP-9 secretion induced by OA. In contrast, OA does not induce an increase on MMP-9 secretion in MCF10A cells, whereas it does not induce MMP-9 secretion in MCF12A mammary non-tumorigenic epithelial cells. In addition, OA induces invasion through an EGFR, Gi/Go proteins, MMPs, PKC and Src-dependent pathway, but it is not able to promote invasion in non-invasive MCF-7 breast cancer cells. In summary, our findings demonstrate that OA promotes an increase on MMP-9 secretion and invasion through a PKC, Src, and EGFR-dependent pathway in breast cancer cells.





Similar content being viewed by others
References
Welsch CW (1992) Relationship between dietary fat and experimental mammary tumorigenesis: a review and critique. Cancer Res 52(7 Suppl):2040s–2048s
Rose DP (1997) Effects of dietary fatty acids on breast and prostate cancers: evidence from in vitro experiments and animal studies. Am J Clin Nutr 66(suppl 6):1513S–1522S
Lee MM, Lin SS (2000) Dietary fat and breast cancer. Annu Rev Nutr 20:221–248
Fay MP, Freedman LS, Clifford CK (1997) Effect of different types and amounts of fat on the development of mammary tumors in rodents: a review. Cancer Res 57:3979–3988
Felber JP, Golay A (2002) Pathways from obesity to diabetes. Int J Obes Relat Metab Disord 26(suppl 2):S39–S45
Calle EE, Kaaks R (2004) Overweight, obesity and cancer: epidemiological evidence and proposed mechanisms. Nat Rev Cancer 4:579–591
McArthur MJ, Atshaves BP, Frolov A (1999) Cellular uptake and intracellular trafficking of long chain fatty acids. J Lipid Res 40:1371–1383
Ferre P (2004) The biology of peroxisome proliferator-activated receptors: relationship with lipid metabolism and insulin sensitivity. Diabetes 53(suppl 1):S43–S50
Louet JF, Chatelain F, Decaux JF (2001) Long-chain fatty acids regulate liver carnitine palmitoyltransferase I gene (L-CPT I) expression through a peroxisome-proliferator-activated receptor alpha (PPARalpha)-independent pathway. Biochem J 354(Pt 1):189–197
Yonezawa T, Katoh K, Obara Y (2004) Existence of GPR40 functioning in a human breast cancer cell line, MCF-7. Biochem Biophys Res Commun 314:805–809
Hirasawa A, Tsumaya K, Awaji T (2005) Free fatty acids regulate gut incretin glucagon-like peptide-1 secretion through GPR120. Nat Med 11:90–94
Itoh Y, Kawamata Y, Harada M (2003) Free fatty acids regulate insulin secretion from pancreatic beta cells through GPR40. Nature 422:173–176
Briscoe CP, Tadayyon M, Andrews JL (2003) The orphan G protein-coupled receptor GPR40 is activated by medium and long chain fatty acids. J Biol Chem 278:11303–11311
Brown AJ, Goldsworthy SM, Barnes AA (2003) The Orphan G protein-coupled receptors GPR41 and GPR43 are activated by propionate and other short chain carboxylic acids. J Biol Chem 278:11312–11319
Xiong Y, Miyamoto N, Shibata K (2004) Short-chain fatty acids stimulate leptin production in adipocytes through the G protein-coupled receptor GPR41. Proc Natl Acad Sci USA 101:1045–1050
Le Poul E, Loison C, Struyf S (2003) Functional characterization of human receptors for short chain fatty acids and their role in polymorphonuclear cell activation. J Biol Chem 278:25481–25489
Navarro-Tito N, Robledo T, Salazar EP (2008) Arachidonic acid promotes FAK activation and migration in MDA-MB-231 breast cancer cells. Exp Cell Res 314:3340–3355
Soto-Guzman A, Robledo T, Lopez-Perez M, Salazar EP (2008) Oleic acid induces ERK1/2 activation and AP-1 DNA binding activity through a mechanism involving Src kinase and EGFR transactivation in breast cancer cells. Mol Cell Endocrinol 294:81–91
Navarro-Tito N, Soto-Guzman A, Castro-Sanchez L, Martinez-Orozco R, Salazar EP (2010) Oleic acid promotes migration on MDA-MB-231 breast cancer cells through an arachidonic acid-dependent pathway. Int J Biochem Cell Biol 42:306–317
Hardy S, St-Onge GG, Joly E, Langelier Y, Prentki M (2005) Oleate promotes the proliferation of breast cancer cells via the G protein-coupled receptor GPR40. J Biol Chem 280:13285–13291
Germain E, Chajes V, Cognault S, Lhuillery C, Bougnoux P (1998) Enhancement of doxorubicin cytotoxicity by polyunsaturated fatty acids in the human breast tumor cell line MDA-MB-231: relationship to lipid peroxidation. Int J Cancer 75:578–583
Przybytkowski E, Joly E, Nolan CJ, Hardy S, Francoeur AM, Langelier Y, Prentki M (2007) Upregulation of cellular triacylglycerol—free fatty acid cycling by oleate is associated with long-term serum-free survival of human breast cancer cells. Biochem Cell Biol 85:301–310
Menendez JA, Mehmi I, Atlas E, Colomer R, Lupu R (2004) Novel signaling molecules implicated in tumor-associated fatty acid synthase-dependent breast cancer cell proliferation and survival: role of exogenous dietary fatty acids, p53–p21WAF1/CIP1, ERK1/2 MAPK, p27KIP1, BRCA1, and NF-kappaB. Int J Oncol 24:591–608
Martinez-Orozco R, Navarro-Tito N, Soto-Guzman A, Castro-Sanchez L, Salazar EP (2010) Arachidonic acid promotes epithelial-to-mesenchymal-like transition inmammary epithelial cells MCF10A. Eur J Cell Biol 89:476–488
Coussens LM, Fingleton B, Matrisian LM (2002) Matrix metalloproteinase inhibitors and cancer: trials and tribulations. Science 295:2387–2392
Curran S, Murray GI (1999) Matrix metalloproteinases in tumour invasion and metastasis. J Pathol 189:300–308
Folgueras AR, Pendas AM, Sanchez LM, Lopez-Otin C (2004) Matrix metalloproteinases in cancer: from new functions to improved inhibition strategies. Int J Dev Biol 48:411–424
Egeblad M, Werb Z (2002) New functions for the matrix metalloproteinases in cancer progression. Nat Rev Cancer 2:161–174
Sternlicht MD, Werb Z (2001) How matrix metalloproteinases regulate cell behavior. Annu Rev Cell Dev Biol 17:463–516
Mendes O, Kim HT, Stoica G (2005) Expression of MMP2, MMP9 and MMP3 in breast cancer brain metastasis in a rat model. Clin Exp Metastasis 22:237–246
Pellikainen JM, Ropponen KM, Kataja VV, Kellokoski JK, Eskelinen MJ, Kosma VM (2004) Expression of matrix metalloproteinase (MMP)-2 and MMP-9 in breast cancer with a special reference to activator protein-2, HER2, and prognosis. Clin Cancer Res 10:7621–7628
Duffy MJ, Maguire TM, Hill A, McDermott E, O’Higgins N (2000) Metalloproteinases: role in breast carcinogenesis, invasion and metastasis. Breast Cancer Res 2:252–257
Jones JL, Glynn P, Walker RA (1999) Expression of MMP-2 and MMP-9, their inhibitors, and the activator MT1-MMP in primary breast carcinomas. J Pathol 189:161–168
Cortes-Reynosa P, Robledo T, Macias-Silva M, Wu SV, Salazar EP (2008) Src kinase regulates metalloproteinase-9 secretion induced by type IV collagen in MCF-7 human breast cancer cells. Matrix Biol 27:220–231
Juarez J, Clayman G, Nakajima M, Tanabe KK, Saya H, Nicolson GL, Boyd D (1993) Role and regulation of expression of 92-kDa type-IV collagenase (MMP-9) in 2 invasive squamous-cell-carcinoma cell lines of the oral cavity. Int J Cancer 55:10–18
Park MJ, Park IC, Hur JH, Rhee CH, Choe TB, Yi DH, Hong SI, Lee SH (2000) Protein kinase C activation by phorbol ester increases in vitro invasion through regulation of matrix metalloproteinases/tissue inhibitors of metalloproteinases system in D54 human glioblastoma cells. Neurosci Lett 290:201–204
Simon C, Goepfert H, Boyd D (1998) Inhibition of the p38 mitogen-activated protein kinase by SB 203580 blocks PMA-induced Mr 92, 000 type IV collagenase secretion and in vitro invasion. Cancer Res 58:1135–1139
Vacaresse N, Lajoie-Mazenc I, Auge N, Suc I, Frisach MF, Salvayre R, Negre-Salvayre A (1999) Activation of epithelial growth factor receptor pathway by unsaturated fatty acids. Circ Res 85:892–899
Kim S, Choi JH, Lim HI, Lee SK, Kim WW, Cho S, Kim JS, Kim JH, Choe JH, Nam SJ, Lee JE, Yang JH (2009) EGF-induced MMP-9 expression is mediated by the JAK3/ERK pathway, but not by the JAK3/STAT-3 pathway in a SKBR3 breast cancer cell line. Cell Signal 21:892–898
Liu JF, Crepin M, Liu JM, Barritault D, Ledoux D (2002) FGF-2 and TPA induce matrix metalloproteinase-9 secretion in MCF-7 cells through PKC activation of the Ras/ERK pathway. Biochem Biophys Res Commun 293:1174–1182
Yao J, Xiong S, Klos K, Nguyen N, Grijalva R, Li P, Yu D (2001) Multiple signaling pathways involved in activation of matrix metalloproteinase-9 (MMP-9) by heregulin-beta1 in human breast cancer cells. Oncogene 20:8066–8074
Sliva D, English D, Lyons D, Lloyd FP Jr (2002) Protein kinase C induces motility of breast cancers by upregulating secretion of urokinase-type plasminogen activator through activation of AP-1 and NF-kappaB. Biochem Biophys Res Commun 290:552–557
Toullec D, Pianetti P, Coste H, Bellevergue P, Grand-Perret T, Ajakane M, Baudet V, Boissin P, Boursier E, Loriolle F et al (1991) The bisindolylmaleimide GF 109203X is a potent and selective inhibitor of protein kinase C. J Biol Chem 266:15771–15781
Blake RA, Broome MA, Liu X, Wu J, Gishizky M, Sun L, Courtneidge SA (2000) SU6656, a selective src family kinase inhibitor, used to probe growth factor signaling. Mol Cell Biol 20:9018–9027
Sun R, Gao P, Chen L, Ma D, Wang J, Oppenheim JJ, Zhang N (2005) Protein kinase C zeta is required for epidermal growth factor-induced chemotaxis of human breast cancer cells. Cancer Res 65:1433–1441
Pettersson F, Couture MC, Hanna N, Miller WH (2004) Enhanced retinoid-induced apoptosis of MDA-MB-231 breast cancer cells by PKC inhibitors involves activation of ERK. Oncogene 23:7053–7066
Moss J, Vaughan M (1988) ADP-ribosylation of guanyl nucleotide-binding regulatory proteins by bacterial toxins. Adv Enzymol Relat Areas Mol Biol 61:303–379
Imada K, Ito A, Sato T, Namiki M, Nagase H, Mori Y (1997) Hormonal regulation of matrix metalloproteinase 9/gelatinase B gene expression in rabbit uterine cervical fibroblasts. Biol Reprod 56:575–580
Kim HS, Luo L, Pflugfelder SC, Li DQ (2005) Doxycycline inhibits TGF-beta1-induced MMP-9 via Smad and MAPK pathways in human corneal epithelial cells. Invest Ophthalmol Vis Sci 46:840–848
Deery WJ, Weisenberg RC (1981) Kinetic and steady-state analysis of microtubules in the presence of colchicine. Biochemistry 20:2316–2324
Wodnicka M, Pierzchalska M, Bereiter-Hahn J, Kajstura J (1992) Comparative study on effects of cytochalasins B and D on F-actin content in different cell lines and different culture conditions. Folia Histochem Cytobiol 30:107–111
Cortesio CL, Chan KT, Perrin BJ, Burton NO, Zhang S, Zhang ZY, Huttenlocher A (2008) Calpain 2 and PTP1B function in a novel pathway with Src to regulate invadopodia dynamics and breast cancer cell invasion. J Cell Biol 180:957–971
Lin KL, Su JC, Chien CM, Tseng CH, Chen YL, Chang LS, Lin SR (2010) Naphtho[1, 2-b]furan-4, 5-dione disrupts Janus kinase-2 and induces apoptosis in breast cancer MDA-MB-231 cells. Toxicol In Vitro 24:1158–1167
Hanke JH, Gardner JP, Dow RL, Changelian PS, Brissette WH, Weringer EJ, Pollok BA, Connelly PA (1996) Discovery of a novel, potent, and Src family-selective tyrosine kinase inhibitor. Study of Lck- and FynT-dependent T cell activation. J Biol Chem 271:695–701
Das R, Mahabeleshwar GH, Kundu GC (2004) Osteopontin induces AP-1-mediated secretion of urokinase-type plasminogen activator through c-Src-dependent epidermal growth factor receptor transactivation in breast cancer cells. J Biol Chem 279:11051–11064
Ricono JM, Huang M, Barnes LA, Lau SK, Weis SM, Schlaepfer DD, Hanks SK, Cheresh DA (2009) Specific cross-talk between epidermal growth factor receptor and integrin alphavbeta5 promotes carcinoma cell invasion and metastasis. Cancer Res 69:1383–1391
Curran S, Murray GI (2000) Matrix metalloproteinases: molecular aspects of their roles in tumour invasion and metastasis. Eur J Cancer 36:1621–1630
Peyri N, Berard M, Fauvel-Lafeve F, Trochon V, Arbeille B, Lu H, Legrand C, Creoin M (2009) Breast tumor cells transendothelial migration induces endothelial cell anoikis through extracellular matrix degradation. Anticancer Res 29:2347–2355
Augustin S, Berard M, Kellaf S, Peyri N, Fauvel-Lafeve F, Legrand C, He L, Crepin M (2009) Matrix metalloproteinases are involved in both type I (apoptosis) and type II (autophagy) cell death induced by sodium phenylacetate in MDA-MB-231 breast tumour cells. Anticancer Res 29:1335–1343
Boyd NF, Stone J, Vogt KN, Connelly BS, Martin LJ, Minkin S (2003) Dietary fat and breast cancer risk revisited: a meta-analysis of the published literature. Br J Cancer 89:1672–1685
Lahmann PH, Hoffmann K, Allen N, van Gils CH, Khaw KT, Tehard B, Berrino F, Tjonneland A, Bigaard J, Olsen A, Overvad K, Clavel-Chapelon F, Nagel G, Boeing H, Trichopoulos D, Economou G, Bellos G, Palli D, Tumino R, Panico S, Sacerdote C, Krogh V, Peeters PH, Bueno-de-Mesquita HB, Lund E, Ardanaz E, Amiano P, Pera G, Quiros JR, Martinez C, Tormo MJ, Wirfalt E, Berglund G, Hallmans G, Key TJ, Reeves G, Bingham S, Norat T, Biessy C, Kaaks R, Riboli E (2004) Body size and breast cancer risk: findings from the European prospective investigation into cancer and nutrition (EPIC). Int J Cancer 111:762–771
Byon CH, Hardy RW, Ren C, Ponnazhagan S, Welch DR, McDonald JM, Chen Y (2009) Free fatty acids enhance breast cancer cell migration through plasminogen activator inhibitor-1 and SMAD4. Lab Invest 89:1221–1228
Kim MS, Lee EJ, Kim HR, Moon A (2003) p38 kinase is a key signaling molecule for H-Ras-induced cell motility and invasive phenotype in human breast epithelial cells. Cancer Res 63:5454–5461
Kim JH, Lee KW, Lee MW, Lee HJ, Kim SH, Surh YJ (2006) Hirsutenone inhibits phorbol ester-induced upregulation of COX-2 and MMP-9 in cultured human mammary epithelial cells: NF-kappaB as a potential molecular target. FEBS Lett 580:385–392
Suzuki I, Iigo M, Ishikawa C, Kuhara T, Asamoto M, Kunimoto T, Moore MA, Yazawa K, Araki E, Tsuda H (1997) Inhibitory effects of oleic and docosahexaenoic acids on lung metastasis by colon-carcinoma-26 cells are associated with reduced matrix metalloproteinase-2 and -9 activities. Int J Cancer 73:607–612
Polette M, Huet E, Birembaut P, Maquart FX, Hornebeck W, Emonard H (1999) Influence of oleic acid on the expression, activation and activity of gelatinase A produced by oncogene-transformed human bronchial epithelial cells. Int J Cancer 80:751–755
Hoshino D, Tomari T, Nagano M, Koshikawa N, Seiki M (2009) A novel protein associated with membrane-type 1 matrix metalloproteinase binds p27(kip1) and regulates RhoA activation, actin remodeling, and matrigel invasion. J Biol Chem 284:27315–27326
Hu XT, Zhang FB, Fan YC, Shu XS, Wong AH, Zhou W, Shi QL, Tang HM, Fu L, Guan XY, Rha SY, Tao Q, He C (2009) Phospholipase C delta 1 is a novel 3p22.3 tumor suppressor involved in cytoskeleton organization, with its epigenetic silencing correlated with high-stage gastric cancer. Oncogene 28:2466–2475
Yilmaz M, Christofori G (2009) EMT, the cytoskeleton, and cancer cell invasion. Cancer Metastasis Rev 28:15–33
Hiscox S, Morgan L, Green T, Nicholson RI (2006) Src as a therapeutic target in anti-hormone/anti-growth factor-resistant breast cancer. Endocr Relat Cancer 13(suppl 1):S53–S59
Zou D, Yoon HS, Anjomshoaa A, Perez D, Fukuzawa R, Guilford P, Humar B (2009) Increased levels of active c-Src distinguish invasive from in situ lobular lesions. Breast Cancer Res 11:R45
Chakrabarti S, Zee JM, Patel KD (2006) Regulation of matrix metalloproteinase-9 (MMP-9) in TNF-stimulated neutrophils: novel pathways for tertiary granule release. J Leukoc Biol 79:214–222
O’Brian C, Vogel VG, Singletary SE, Ward NE (1989) Elevated protein kinase C expression in human breast tumor biopsies relative to normal breast tissue. Cancer Res 49:3215–3217
Blobe GC, Obeid LM, Hannun YA (1994) Regulation of protein kinase C and role in cancer biology. Cancer Metastasis Rev 13:411–431
Iakoubov R, Izzo A, Yeung A, Whiteside CI, Brubaker PL (2007) Protein kinase Czeta is required for oleic acid-induced secretion of glucagon-like peptide-1 by intestinal endocrine L cells. Endocrinology 148:1089–1098
Johnson MD, Torri JA, Lippman ME, Dickson RB (1999) Regulation of motility and protease expression in PKC-mediated induction of MCF-7 breast cancer cell invasiveness. Exp Cell Res 247:105–113
Aziz MH, Hafeez BB, Sand JM, Pierce DB, Aziz SW, Dreckschmidt NE, Verma Ak (2010) Protein kinase Cvarepsilon mediates Stat3Ser727 phosphorylation, Stat3-regulated gene expression, and cell invasion in various human cancer cell lines through integration with MAPK cascade (RAF-1, MEK1/2, and ERK1/2). Oncogene 29:3100–3109
Ellerbroek SM, Hudson LG, Stack MS (1998) Proteinase requirements of epidermal growth factor-induced ovarian cancer cell invasion. Int J Cancer 78:331–337
Cox G, Jones JL, O’Byrne KJ (2000) Matrix metalloproteinase 9 and the epidermal growth factor signal pathway in operable non-small cell lung cancer. Clin Cancer Res 6:2349–2355
Schwartzberg PL (1998) The many faces of Src: multiple functions of a prototypical tyrosine kinase. Oncogene 17:1463–1468
Liu J, Yue P, Artym VV, Mueller SC, Guo W (2009) The Role of the exocyst in matrix metalloproteinase secretion and actin dynamics during tumor cell invadopodia formation. Mol Biol Cell 20:3763–3771
Acknowledgments
We are grateful to Nora Ruiz for her technical assistance. This work was supported by grants from CONACYT (43370 and 83802). A. S-G is a recipient of a postdoctoral fellowship from ICyTDF. L. C-S, R. M-O and N. N-T were supported by a CONACYT predoctoral training grant.
Author information
Authors and Affiliations
Corresponding author
Additional information
Adriana Soto-Guzman and Napoleon Navarro-Tito contributed equally to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
10585_2010_9340_MOESM1_ESM.tif
Oleic acid induces invasiveness in MDA-MB-231 breast cancer cells MDA-MB-231 cells held in suspension were treated for 2 h in the absence (-) or presence (+) of 12 μM mitomicyn C (Mit.C), as indicated and then cells were plated on the top of matrigel and treated with 200 μM oleic acid for various times, as indicated. Cell invasion was evaluated after incubation. (TIFF 1733 kb)
Rights and permissions
About this article
Cite this article
Soto-Guzman, A., Navarro-Tito, N., Castro-Sanchez, L. et al. Oleic acid promotes MMP-9 secretion and invasion in breast cancer cells. Clin Exp Metastasis 27, 505–515 (2010). https://doi.org/10.1007/s10585-010-9340-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10585-010-9340-1