Abstract
Background
Shaping immune responses to prevent tumor-induced tolerance or transplant rejection after solid organ transplantation is a permanently expanding field of research. Immunological tolerance, in this case, is a double-edged sword. Tumors escape immune surveillance by creating an abnormal state of tolerance towards their own antigens, whereas transplantation medicine is challenged to develop new strategies to induce allograft-specific immunological tolerance. One mechanism possibly capable of achieving immunoregulation is based on indoleamine-2,3-dioxygenase (IDO).
Objective
This overview article focuses on IDO-mediated tryptophan catabolism with special regard to its role in cancer and transplantation immunology.
Results
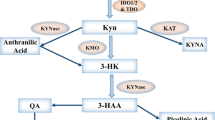
The historical view about IDO as a host’s antimicrobial defence mechanism has been extended by the observation that its expression is essential for successful allogeneic pregnancy. Subsequent studies analysing IDO as an immune-regulatory enzyme describe its implications in cancer immune escape, as chemical abrogation of enzyme activity with 1-methyl-tryptophan (1-MT), results in enhanced antitumor responses in animal models. Therefore, a clinical trial treating cancer patients with 1-MT has been started. IDO also seems to play an essential role in the control of allo- and autoreactive T cell responses. CTLA4-Ig is able to induce IDO expression in dendritic cells (DCs) and consequently renders them tolerogenic, which might provide one explanation for the observed therapeutic effects of abatacept and belatacept.
Conclusion
There is evidence that IDO achieves immune modulation in several animal models. However, in humans, this remains controversially discussed.


Similar content being viewed by others
References
Moffett JR, Namboodiri MA (2003) Tryptophan and the immune response. Immunol Cell Biol 81(4):247–265
Yoshida R, Hayaishi O (1978) Induction of pulmonary indoleamine 2,3-dioxygenase by intraperitoneal injection of bacterial lipopolysaccharide. Proc Natl Acad Sci USA 75(8):3998–4000
Werner ER, Werner-Felmayer G, Fuchs D, Hausen A, Reibnegger G, Wachter H (1989) Parallel induction of tetrahydrobiopterin biosynthesis and indoleamine 2,3-dioxygenase activity in human cells and cell lines by interferon-gamma. Biochem J 262(3):861–866
Werner-Felmayer G, Werner ER, Fuchs D, Hausen A, Reibnegger G, Wachter H (1989) Characteristics of interferon induced tryptophan metabolism in human cells in vitro. Biochim Biophys Acta 1012(2):140–147
Saito K, Markey SP, Heyes MP (1992) Effects of immune activation on quinolinic acid and neuroactive kynurenines in the mouse. Neuroscience 51(1):25–39
Terness P, Chuang JJ, Opelz G (2006) The immunoregulatory role of IDO-producing human dendritic cells revisited. Trends Immunol 27(2):68–73
Munn DH, Sharma MD, Lee JR, Jhaver KG, Johnson TS, Keskin DB, Marshall B, Chandler P, Antonia SJ, Burgess R, Slingluff CL Jr, Mellor AL (2002) Potential regulatory function of human dendritic cells expressing indoleamine 2,3-dioxygenase. Science 297(5588):1867–1870
Hayaishi O (1996) Utilization of superoxide anion by indoleamine oxygenase-catalyzed tryptophan and indoleamine oxidation. Adv Exp Med Biol 398:285–289
Thomas SR, Stocker R (1999) Redox reactions related to indoleamine 2,3-dioxygenase and tryptophan metabolism along the kynurenine pathway. Redox Rep 4(5):199–220
Hirata F, Hayaishi O (1975) Studies on indoleamine 2,3-dioxygenase. I. Superoxide anion as substrate. J Biol Chem 250(15):5960–5966
Pfefferkorn ER (1984) Interferon gamma blocks the growth of Toxoplasma gondii in human fibroblasts by inducing the host cells to degrade tryptophan. Proc Natl Acad Sci USA 81(3):908–912
Munn DH, Zhou M, Attwood JT, Bondarev I, Conway SJ, Marshall B, Brown C, Mellor AL (1998) Prevention of allogeneic fetal rejection by tryptophan catabolism. Science 281(5380):1191–1193
Mellor AL, Munn DH (1999) Tryptophan catabolism and T-cell tolerance: immunosuppression by starvation? Immunol Today 20(10):469–473
Moffett JR, Espey MG, Saito K, Namboodiri MA (1994) Quinolinic acid immunoreactive cells in the choroid plexus, leptomeninges and brain vasculature of the immune-stimulated gerbil. J Neuroimmunol 54(1–2):69–73
Terness P, Bauer TM, Rose L, Dufter C, Watzlik A, Simon H, Opelz G (2002) Inhibition of allogeneic T cell proliferation by indoleamine 2,3-dioxygenase-expressing dendritic cells: mediation of suppression by tryptophan metabolites. J Exp Med 196(4):447–457
Bauer TM, Jiga LP, Chuang JJ, Randazzo M, Opelz G, Terness P (2005) Studying the immunosuppressive role of indoleamine 2,3-dioxygenase: tryptophan metabolites suppress rat allogeneic T-cell responses in vitro and in vivo. Transpl Int 18(1):95–100
Ehrlich P (1909) Über den jetzigen Stand der Karzinomforschung. Ned Tijdschr Geneeskd 5:273–290
Burnet FM (1970) The concept of immunological surveillance. Prog Exp Tumor Res 13:1–27
Thomas L (1959) Discussion in cellular and humoral aspects of the hypersensitive state. In: Lawrence HS (ed) Hoeber, New York, p 529. ff
Khong HT, Restifo NP (2002) Natural selection of tumor variants in the generation of “tumor escape” phenotypes. Nat Immunol 3(11):999–1005
Melief CJ, Toes RE, Medema JP, van der Burg SH, Ossendorp F, Offringa R (2000) Strategies for immunotherapy of cancer. Adv Immunol 75:235–282
Pardoll D (2003) Does the immune system see tumors as foreign or self? Annu Rev Immunol 21:807–839
Fuchs EJ, Matzinger P (1996) Is cancer dangerous to the immune system? Semin Immunol 8(5):271–280
Dong H, Strome SE, Salomao DR, Tamura H, Hirano F, Flies DB, Roche PC, Lu J, Zhu G, Tamada K, Lennon VA, Celis E, Chen L (2002) Tumor-associated B7-H1 promotes T-cell apoptosis: a potential mechanism of immune evasion. Nat Med 8(8):793–800
O’Connell J, Bennett MW, O’Sullivan GC, Collins JK, Shanahan F (1999) The Fas counterattack: cancer as a site of immune privilege. Immunol Today 20(1):46–52
Lopez CB, Rao TD, Feiner H, Shapiro R, Marks JR, Frey AB (1998) Repression of interleukin-2 mRNA translation in primary human breast carcinoma tumor-infiltrating lymphocytes. Cell Immunol 190(2):141–155
Sheu BC, Lin RH, Ho HN, Huang SC (1997) Down-regulation of CD25 expression on the surface of activated tumor-infiltrating lymphocytes in human cervical carcinoma. Hum Immunol 56(1–2):39–48
Radoja S, Saio M, Schaer D, Koneru M, Vukmanovic S, Frey AB (2001) CD8(+) tumor-infiltrating T cells are deficient in perforin-mediated cytolytic activity due to defective microtubule-organizing center mobilization and lytic granule exocytosis. J Immunol 167(9):5042–5051
Sutmuller RP, van Duivenvoorde LM, van Elsas A, Schumacher TN, Wildenberg ME, Allison JP, Toes RE, Offringa R, Melief CJ (2001) Synergism of cytotoxic T lymphocyte-associated antigen 4 blockade and depletion of CD25(+) regulatory T cells in antitumor therapy reveals alternative pathways for suppression of autoreactive cytotoxic T lymphocyte responses. J Exp Med 194(6):823–832
Yu P, Lee Y, Liu W, Krausz T, Chong A, Schreiber H, Fu YX (2005) Intratumor depletion of CD4+ cells unmasks tumor immunogenicity leading to the rejection of late-stage tumors. J Exp Med 201(5):779–791
Bubenik J (2004) MHC class I down-regulation: tumour escape from immune surveillance? (review). Int J Oncol 25(2):487–491
Zou W (2005) Immunosuppressive networks in the tumour environment and their therapeutic relevance. Nat Rev Cancer 5(4):263–274
Uyttenhove C, Pilotte L, Theate I, Stroobant V, Colau D, Parmentier N, Boon T, Van den Eynde BJ (2003) Evidence for a tumoral immune resistance mechanism based on tryptophan degradation by indoleamine 2,3-dioxygenase. Nat Med 9(10):1269–1274
Curti A, Pandolfi S, Valzasina B, Aluigi M, Isidori A, Ferri E, Salvestrini V, Bonanno G, Rutella S, Durelli I, Horenstein AL, Fiore F, Massaia M, Colombo MP, Baccarani M, Lemoli RM (2006) Modulation of tryptophan catabolism by human leukemic cells results in the conversion of CD25− into CD25+ T regulatory cells. Blood 109:2871–2877
Brandacher G, Perathoner A, Ladurner R, Schneeberger S, Obrist P, Winkler C, Werner ER, Werner-Felmayer G, Weiss HG, Gobel G, Margreiter R, Konigsrainer A, Fuchs D, Amberger A (2006) Prognostic value of indoleamine 2,3-dioxygenase expression in colorectal cancer: effect on tumor-infiltrating T cells. Clin Cancer Res 12(4):1144–1151
Ino K, Yoshida N, Kajiyama H, Shibata K, Yamamoto E, Kidokoro K, Takahashi N, Terauchi M, Nawa A, Nomura S, Nagasaka T, Takikawa O, Kikkawa F (2006) Indoleamine 2,3-dioxygenase is a novel prognostic indicator for endometrial cancer. Br J Cancer 95(11):1555–1561
Galon J, Costes A, Sanchez-Cabo F, Kirilovsky A, Mlecnik B, Lagorce-Pages C, Tosolini M, Camus M, Berger A, Wind P, Zinzindohoue F, Bruneval P, Cugnenc PH, Trajanoski Z, Fridman WH, Pages F (2006) Type, density, and location of immune cells within human colorectal tumors predict clinical outcome. Science 313(5795):1960–1964
Spiotto MT, Yu P, Rowley DA, Nishimura MI, Meredith SC, Gajewski TF, Fu YX, Schreiber H (2002) Increasing tumor antigen expression overcomes “ignorance” to solid tumors via crosspresentation by bone marrow-derived stromal cells. Immunity 17(6):737–747
Banchereau J, Steinman RM (1998) Dendritic cells and the control of immunity. Nature 392(6673):245–252
Munn DH, Sharma MD, Hou D, Baban B, Lee JR, Antonia SJ, Messina JL, Chandler P, Koni PA, Mellor AL (2004) Expression of indoleamine 2,3-dioxygenase by plasmacytoid dendritic cells in tumor-draining lymph nodes. J Clin Invest 114(2):280–290
Fallarino F, Grohmann U, Hwang KW, Orabona C, Vacca C, Bianchi R, Belladonna ML, Fioretti MC, Alegre ML, Puccetti P (2003) Modulation of tryptophan catabolism by regulatory T cells. Nat Immunol 4(12):1206–1212
Mellor AL, Chandler P, Baban B, Hansen AM, Marshall B, Pihkala J, Waldmann H, Cobbold S, Adams E, Munn DH (2004) Specific subsets of murine dendritic cells acquire potent T cell regulatory functions following CTLA4-mediated induction of indoleamine 2,3 dioxygenase. Int Immunol 16(10):1391–1401
Lee JH, Torisu-Itakara H, Cochran AJ, Kadison A, Huynh Y, Morton DL, Essner R (2005) Quantitative analysis of melanoma-induced cytokine-mediated immunosuppression in melanoma sentinel nodes. Clin Cancer Res 11(1):107–112
Lee JR, Dalton RR, Messina JL, Sharma MD, Smith DM, Burgess RE, Mazzella F, Antonia SJ, Mellor AL, Munn DH (2003) Pattern of recruitment of immunoregulatory antigen-presenting cells in malignant melanoma. Lab Invest 83(10):1457–1466
Lechler RI, Garden OA, Turka LA (2003) The complementary roles of deletion and regulation in transplantation tolerance. Nat Rev Immunol 3(2):147–158
Mellor AL, Munn DH (2001) Tryptophan catabolism prevents maternal T cells from activating lethal anti-fetal immune responses. J Reprod Immunol 52(1–2):5–13
Schrocksnadel K, Widner B, Bergant A, Neurauter G, Schrocksnadel H, Fuchs D (2003) Tryptophan degradation during and after gestation. Adv Exp Med Biol 527:77–83
Mellor AL, Baban B, Chandler P, Marshall B, Jhaver K, Hansen A, Koni PA, Iwashima M, Munn DH (2003) Cutting edge: induced indoleamine 2,3 dioxygenase expression in dendritic cell subsets suppresses T cell clonal expansion. J Immunol 171(4):1652–1655
Collins AV, Brodie DW, Gilbert RJ, Iaboni A, Manso-Sancho R, Walse B, Stuart DI, van der Merwe PA, Davis SJ (2002) The interaction properties of costimulatory molecules revisited. Immunity 17(2):201–210
Moreland L, Bate G, Kirkpatrick P (2006) Abatacept. Nat Rev Drug Discov 5(3):185–186
Vincenti F, Larsen C, Durrbach A, Wekerle T, Nashan B, Blancho G, Lang P, Grinyo J, Halloran PF, Solez K, Hagerty D, Levy E, Zhou W, Natarajan K, Charpentier B (2005) Costimulation blockade with belatacept in renal transplantation. N Engl J Med 353(8):770–781
Hainz U, Obexer P, Winkler C, Sedlmayr P, Takikawa O, Greinix H, Lawitschka A, Potschger U, Fuchs D, Ladisch S, Heitger A (2005) Monocyte-mediated T-cell suppression and augmented monocyte tryptophan catabolism after human hematopoietic stem-cell transplantation. Blood 105(10):4127–4134
Steckel NK, Kuhn U, Beelen DW, Elmaagacli AH (2003) Indoleamine 2,3-dioxygenase expression in patients with acute graft-versus-host disease after allogeneic stem cell transplantation and in pregnant women: association with the induction of allogeneic immune tolerance? Scand J Immunol 57(2):185–191
Brandacher G, Cakar F, Winkler C, Schneeberger S, Obrist P, Bosmuller C, Werner-Felmayer G, Werner ER, Bonatti H, Margreiter R, Fuchs D (2007) Non-invasive monitoring of kidney allograft rejection through IDO metabolism evaluation. Kidney Int 71(1):60–67
Brown RR, Ozaki Y, Datta SP, Borden EC, Sondel PM, Malone DG (1991) Implications of interferon-induced tryptophan catabolism in cancer, auto-immune diseases and AIDS. Adv Exp Med Biol 294:425–435
Friberg M, Jennings R, Alsarraj M, Dessureault S, Cantor A, Extermann M, Mellor AL, Munn DH, Antonia SJ (2002) Indoleamine 2,3-dioxygenase contributes to tumor cell evasion of T cell-mediated rejection. Int J Cancer 101(2):151–155
Muller AJ, DuHadaway JB, Donover PS, Sutanto-Ward E, Prendergast GC (2005) Inhibition of indoleamine 2,3-dioxygenase, an immunoregulatory target of the cancer suppression gene Bin1, potentiates cancer chemotherapy. Nat Med 11(3):312–319
Hou DY, Muller AJ, Sharma MD, DuHadaway J, Banerjee T, Johnson M, Mellor AL, Prendergast GC, Munn DH (2007) Inhibition of indoleamine 2,3-dioxygenase in dendritic cells by stereoisomers of 1-methyl-tryptophan correlates with antitumor responses. Cancer Res 67(2):792–801
Metz R, Duhadaway JB, Kamasani U, Laury-Kleintop L, Muller AJ, Prendergast GC (2007) Novel tryptophan catabolic enzyme IDO2 is the preferred biochemical target of the antitumor indoleamine 2,3-dioxygenase inhibitory compound d-1-methyl-tryptophan. Cancer Res 67(15):7082–7087
Lob S, Ebner S, Wagner S, Weinreich J, Schafer R, Konigsrainer A (2007) Are indoleamine-2,3-dioxygenase producing human dendritic cells a tool for suppression of allogeneic T-cell responses? Transplantation 83(4):468–473
Terness P, Chuang JJ, Bauer T, Jiga L, Opelz G (2005) Regulation of human auto- and alloreactive T cells by indoleamine 2,3-dioxygenase (IDO)-producing dendritic cells: too much ado about IDO? Blood 105(6):2480–2486
Kudo Y, Boyd CA (2001) Characterisation of l-tryptophan transporters in human placenta: a comparison of brush border and basal membrane vesicles. J Physiol 531(Pt 2):405–416
Agaugue S, Perrin-Cocon L, Coutant F, Andre P, Lotteau V (2006) 1-Methyl-tryptophan can interfere with TLR signaling in dendritic cells independently of IDO activity. J Immunol 177(4):2061–2071
Vottero E, Balgi A, Woods K, Tugendreich S, Melese T, Andersen RJ, Mauk AG, Roberge M (2006) Inhibitors of human indoleamine 2,3-dioxygenase identified with a target-based screen in yeast. Biotechnol J 1(3):282–288
Grohmann U, Fallarino F, Puccetti P (2003) Tolerance, DCs and tryptophan: much ado about IDO. Trends Immunol 24(5):242–248
Acknowledgments
The authors thank Lynne Yakes and Hans-Georg Rammensee for assistance in preparing the manuscript.
This work was supported by a Fortüne grant of the University of Tübingen (1636-0-0 to S.L.).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Löb, S., Königsrainer, A. Is IDO a key enzyme bridging the gap between tumor escape and tolerance induction?. Langenbecks Arch Surg 393, 995–1003 (2008). https://doi.org/10.1007/s00423-007-0245-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00423-007-0245-7