Abstract
Purpose
To investigate the effect of titanium dioxide (TiO2) nanoparticles on the inhibition of the in vitro cellular activity of human Tenon’s fibroblasts (HTFs) under UVA exposure.
Methods
The effects of TiO2 nanoparticles on human Tenon’s fibroblasts were evaluated after 1, 4, 6, and 24 h of exposure to UVA at levels of 2.5, 5.0, and 10 J/cm2. The methyl thiazolyl tetrazolium (MTT) assay was performed to measure the suppression of cellular metabolic activity. The lactate dehydrogenase (LDH) assay was performed to determine the extent of cell membrane damage. Flow cytometric analysis and inverted phase-contrast and electron microscopy were performed. The scratch wound assay was performed to visualize suppression of cellular migration.
Results
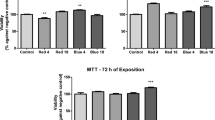
MTT assay values were similar between the UVA-exposed groups and the control group without UVA exposure. However, the combined exposure of TiO2 nanoparticles and UVA exposure induced significant dose-dependent inhibition of cellular viability and damage to HTFs, especially at concentrations of TiO2 equal to or greater than 100 μg/mL and 2.5 J/cm2 of UVA irradiation. Changes in cellular morphology increased in a dose-dependent pattern with a TiO2 concentration greater than 100 μg/mL under UVA exposure. At a TiO2 concentration of 150 μg/mL, damage to the cellular morphology of the HTFs was significantly increased, and nanoparticles were seen inside of the cytoplasm in the affected HTFs exposed to UVA. There was a significant reduction of cellular migration at TiO2 concentrations higher than 150 μg/mL.
Conclusion
TiO2 nanoparticles inhibited the cellular activity of HTFs under UVA irradiation and showed potential for use to prevent the wound scarring of Tenon’s fibroblasts. Further studies will be necessary to determine the optimal concentration of TiO2 nanoparticles and UVA exposure dose for clinical applications.








Similar content being viewed by others
References
Lockwood A, Brocchini S, Khaw PT (2013) New developments in the pharmacological modulation of wound healing after glaucoma filtration surgery. Curr Opin Pharmacol 13(1):65–71. https://doi.org/10.1016/j.coph.2012.10.008
Jampel HD, Pasquale LR, Dibernardo C (1992) Hypotony maculopathy following trabeculectomy with mitomycin C. Arch Ophthalmol 110(8):1049–1050
Chawla A, Mercieca K, Fenerty C, Jones NP (2013) Outcomes and complications of trabeculectomy enhanced with 5-fluorouracil in adults with glaucoma secondary to uveitis. J Glaucoma 22(8):663–666. https://doi.org/10.1097/IJG.0b013e318255dc07
Higginbotham EJ, Stevens RK, Musch DC, Karp KO, Lichter PR, Bergstrom TJ, Skuta GL (1996) Bleb-related endophthalmitis after trabeculectomy with mitomycin C. Ophthalmology 103(4):650–656
Akova YA, Bulut S, Dabil H, Duman S (1999) Late bleb-related endophthalmitis after trabeculectomy with mitomycin C. Ophthalmic Surg Lasers 30(2):146–151
Stamper RL, McMenemy MG, Lieberman MF (1992) Hypotonous maculopathy after trabeculectomy with subconjunctival 5-fluorouracil. Am J Ophthalmol 114(5):544–553
Darian-Smith E, Toh T (2014) Corneal decompensation following bleb needling with 0.01% mitomycin C: a case study. Clin Exp Ophthalmol 42(9):901–902. https://doi.org/10.1111/ceo.12345
Chen X, Selloni A (2014) Introduction: titanium dioxide (TiO2) nanomaterials. Chem Rev 114(19):9281–9282. https://doi.org/10.1021/cr500422r
Chen X, Mao SS (2007) Titanium dioxide nanomaterials: synthesis, properties, modifications, and applications. Chem Rev 107(7):2891–2959. https://doi.org/10.1021/cr0500535
Kang SJ, Lee YJ, Kim BM, Choi YJ, Chung HW (2011) Cytotoxicity and genotoxicity of titanium dioxide nanoparticles in UVA-irradiated normal peripheral blood lymphocytes. Drug Chem Toxicol 34(3):277–284. https://doi.org/10.3109/01480545.2010.546800
Tu M, Huang Y, Li HL, Gao ZH (2012) The stress caused by nitrite with titanium dioxide nanoparticles under UVA irradiation in human keratinocyte cell. Toxicology 299(1):60–68. https://doi.org/10.1016/j.tox.2012.05.011
Sayes CM, Wahi R, Kurian PA, Liu Y, West JL, Ausman KD, Warheit DB, Colvin VL (2006) Correlating nanoscale titania structure with toxicity: a cytotoxicity and inflammatory response study with human dermal fibroblasts and human lung epithelial cells. Toxicol Sci 92(1):174–185. https://doi.org/10.1093/toxsci/kfj197
Yin JJ, Liu J, Ehrenshaft M, Roberts JE, Fu PP, Mason RP, Zhao B (2012) Phototoxicity of nano titanium dioxides in HaCaT keratinocytes--generation of reactive oxygen species and cell damage. Toxicol Appl Pharmacol 263(1):81–88. https://doi.org/10.1016/j.taap.2012.06.001
Sanders K, Degn LL, Mundy WR, Zucker RM, Dreher K, Zhao B, Roberts JE, Boyes WK (2012) In vitro phototoxicity and hazard identification of nano-scale titanium dioxide. Toxicol Appl Pharmacol 258(2):226–236. https://doi.org/10.1016/j.taap.2011.10.023
Seo JW, Chung H, Kim MY, Lee J, Choi IH, Cheon J (2007) Development of water-soluble single-crystalline TiO2 nanoparticles for photocatalytic cancer-cell treatment. Small 3(5):850–853. https://doi.org/10.1002/smll.200600488
Cai R, Kubota Y, Shuin T, Sakai H, Hashimoto K, Fujishima A (1992) Induction of cytotoxicity by photoexcited TiO2 particles. Cancer Res 52(8):2346–2348
Adams LK, Lyon DY, McIntosh A, Alvarez PJ (2006) Comparative toxicity of nano-scale TiO2, SiO2 and ZnO water suspensions. Water Sci Technol 54(11–12):327–334
Hirakawa K, Mori M, Yoshida M, Oikawa S, Kawanishi S (2004) Photo-irradiated titanium dioxide catalyzes site specific DNA damage via generation of hydrogen peroxide. Free Radic Res 38(5):439–447
Desmouliere A, Redard M, Darby I, Gabbiani G (1995) Apoptosis mediates the decrease in cellularity during the transition between granulation tissue and scar. Am J Pathol 146(1):56–66
Liao CM, Chiang YH, Chio CP (2009) Assessing the airborne titanium dioxide nanoparticle-related exposure hazard at workplace. J Hazard Mater 162(1):57–65. https://doi.org/10.1016/j.jhazmat.2008.05.020
Rehn B, Seiler F, Rehn S, Bruch J, Maier M (2003) Investigations on the inflammatory and genotoxic lung effects of two types of titanium dioxide: untreated and surface treated. Toxicol Appl Pharmacol 189(2):84–95
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical standard
This article does not contain any studies with human participants or animals performed by any of the authors.
Ethical approval
All procedures performed in studies were in accordance with the ethical standards of the Medical Research Institution of Pusan National University Hospital and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Rights and permissions
About this article
Cite this article
Lee, SU., Lee, JE., Kim, SJ. et al. Effects of titanium dioxide nanoparticles on the inhibition of cellular activity in human Tenon’s fibroblasts under UVA exposure. Graefes Arch Clin Exp Ophthalmol 256, 1895–1903 (2018). https://doi.org/10.1007/s00417-018-4091-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00417-018-4091-9