Abstract
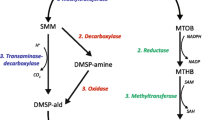
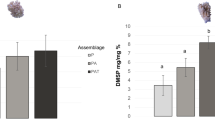
Many groups of tropical cnidarians including scleractinian corals, octocorals, corallimorphs, and anemones contain the tertiary sulfonium compound dimethylsulfoniopropionate (DMSP). It is not known if the compound is synthesized by the animals, their microalgal symbionts, or derived through their diet. We determined the source of the DMSP in several species of tropical and temperate anemones using three approaches: (1) conducting comparative measurements of DMSP in aposymbiotic and zooxanthellate anemones of three species that harbor zooxanthellae, and similar measurements in one species that can harbor both zooxanthellae and zoochlorellae, (2) manipulating the presence or absence of zooxanthellae by inoculating juvenile aposymbiotic anemones (Aiptasia pallida) with their symbiont, Symbiodinium bermudense, and (3) manipulating the numbers of S. bermudense by growing aposymbiotic and zooxanthellate A. pallida in the light and the dark. DMSP was present in zooxanthellate anemones in concentrations of 3.4–15 μmol g−1 fresh mass (FM). In aposymbiotic Aiptasia spp. and Anthopleura elegantissima that lacked large numbers of zooxanthellae, concentrations ranged from being undetectable to 0.43 μmol g−1 FM. When aposymbiotic A. pallida were inoculated with zooxanthellae, concentrations of DMSP were an average of 4.24 μmol g−1 FM after 5 weeks; DMSP was undetectable in uninoculated control animals. Aposymbiotic anemones maintained in the light or the dark for 6 weeks contained no DMSP or zooxanthellae. Zooxanthellate anemones in the light contained five times as many zooxanthellae and approximately 7.5 times as much DMSP as zooxanthellate anemones maintained in the dark. Taken together, these data show that the zooxanthellae are the sole source of DMSP in A. pallida. The trends in DMSP concentrations in other species of zooxanthellate anemones suggest that this phenomenon is not limited to A. pallida but may be more generally true for other anemones or even other cnidarians hosting species of Symbiodinium.

Similar content being viewed by others
References
Augustine L, Muller-Parker G (1998) Selective predation by the mosshead sculpin Clinocottus globiceps on the sea anemone Anthopleura elegantissima and its two algal symbionts. Limnol Oceanogr 43:711–715
Banaszak A, Iglesias-Prieto R, Trench R (1993) Scrippsiella velellae sp. nov. (Peridiniales) and Gloeodinium viscum sp. nov. (Phytodiniales), dinoflagellate symbionts of two hydrozoans (Cnidaria). J Phycol 29:517–528
Bates TS, Charlson RJ, Gammon RH (1987) Evidence for climatic role of marine biogenic sulphur. Nature 329:319–321
Brasseur G, Orlando J, Tyndall G (1999) Atmospheric chemistry and global change. Oxford, New York
Broadbent A, Jones G (2004) DMS and DMSP in mucus ropes, coral mucus, surface films and sediment pore waters from coral reefs in the Great Barrier Reef. Mar Freshwater Res 55:849–855
Broadbent A, Jones G, Jones R (2002) DMSP in corals and benthic algae from the Great Barrier Reef. Estuar Coast Shelf Sci 55:547–555
Buchanan BB, Gruissem W, Jones RL (2000) Biochemistry and molecular biology of plants. Wiley, Somerset
Cantoni G, Anderson D (1956) Enzymatic cleavage of dimethylpropiothetin by Polysiphonia lanosa. J Biol Chem 222:171–177
Challenger F, Simpson M (1948) Studies on biological methylation. Part 12. A precursor of the dimethyl sulphide evolved by Polysiphonia fastigiata. Dimethyl-2-carboxyethylsulphonium hydroxide and its salts. J Chem Soc 3:1591–1597
Charlson RJ, Lovelock JE, Andreae MO, Warren SG (1987) Oceanic phytoplankton, atmospheric sulphur, cloud albedo and climate. Nature 326:655–661
Clayton WJ, Lasker H (1985) Individual and population growth in the asexually reproducing anemone Aiptasia pallida Verrill. J Exp Mar Biol Ecol 90:249–258
Cook C, D’Elia C, Muller-Parker G (1988) Host feeding and nutrient sufficiency for zooxanthellae in the sea anemone Aiptasia pallida. Mar Biol 98:253–262
DeBose J, Lema S, Nevitt G (2008) Dimethylsulfoniopropionate as a foraging cue for reef fishes. Science 319:1356
Fitzgerald L, Szmant A (1997) Biosynthesis of “essential” amino acids by scleractinian corals. Biochem J 322:213–221
Gröne T, Kirst GO (1992) The effects of nutrient deficiency, methionine, and inhibitors of methionine metabolism on the DMSP contents of Tetraselmis subcordiformis (Stein). Mar Biol 112:497–503
Hill RW, Dacey JWH, Krupp DA (1995) Dimethylsulfoniopropionate in reef corals. Bull Mar Sci 57:489–494
Hill RW, Dacey JWH, Edward A (2000) Dimethylsulfoniopropionate in giant clams (Tridacnidae). Biol Bull 199:108–115
Johnson C, Field C (1993) Using fixed-effects model multivariate analysis of variance in marine biology and ecology. Oceanogr Mar Biol Ann Rev 31:177–221
Jones G, Trevena A (2005) The influence of coral reefs on atmospheric dimethylsulphide over the Great Barrier Reef, Coral Sea, Gulf of Papua and Solomon and Bismark Seas. Mar Freshwater Res 56:85–93
Jones G, Curran M, Broadbent A (1994) Dimethylsulphide in the South Pacific. 6th Pacific Congr Mar Sci Technol:183–190
Kettle A, Andreae MO (2000) Flux of dimethylsulfide from the oceans: a comparison of updated data sets and flux models. J Geophys Res 26:26793–26808
Kiene RP, Visscher P, Keller M, Kirst GO (1996) Biological and environmental chemistry of DMSP and related sulfonium compounds. Plenum Press, New York
LaJeunesse T, Trench R (2000) The biogeography of two species of Symbiodinium (Freudenthal) inhabiting the intertidal anemone, Anthopleura elegantissima (Brandt). Biol Bull 199:126–134
Lewis L, Muller-Parker G (2004) Phylogenetic placement of “zoochlorellae” (Chlorophyta), algal symbiont of the temperate sea anemone Anthopleura elegantissima. Biol Bull 207:87–92
Liss PS, Malin G, Turner SM, Holligan PM (1994) Dimethyl sulphide and Phaeocystis: a review. J Mar Syst 5:41–53
Lovelock JE, Maggs C, Rasmussen R (1972) Atmospheric dimethyl sulfide and the natural sulfur cycle. Nature 237:452–453
Malin G (1996) The role of DMSP and DMS in the global sulfur cycle and climate regulation. In: Kiene RP, Visscher P, Keller M, Kirst GO (eds) Biological and environmental chemistry of DMSP and related sulfonium compounds. Plenum Press, New York, pp 177–189
Malin G (1997) Sulphur, climate and the microbial maze. Nature 387:857–859
Malin G, Kirst GO (1997) Algal production of dimethyl sulfide and its atmospheric role. J Phycol 33:889–896
Malin G, Turner SM, Liss PS (1992) Sulfur: the plankton/climate connection. J Phycol 28:590–597
McNally K, Govind N, Thomé P, Trench R (1994) Small-subunit ribosomal DNA sequence analyses and a reconstruction of the inferred phylogeny among symbiotic dinoflagellates (Pyrrophyta). J Phycol 30:316–329
Muller-Parker G (1987) Seasonal variation in light-shade adaptation of natural populations of the symbiotic sea anemone Aiptasia pulchella (Carlgren, 1943) in Hawaii. J Exp Mar Biol Ecol 112:165–183
Muller-Parker G, Lee K, Cook C (1996) Changes in the ultrastructure of symbiotic zooxanthellae (Symbiodinium sp., Dinophyceae) in fed and starved sea anemones maintained under high and low light. J Phycol 32:987–994
Nevitt G (1999) Foraging by seabirds on an olfactory landscape. Amer Scient 87:46–53
Nevitt G, Bonadonna F (2005) Sensitivity to dimethyl sulphide suggests a mechanism for olfactory navigation by seabirds. Biol Lett 1:303–305
Nevitt GA, Veit RR, Kareiva P (1995) Dimethyl sulphide as a foraging cue for Antarctic procellariiform seabirds. Nature 376:680–682
Otte ML, Wilson G, Morris J, Moran B (2004) Dimethylsulphoniopropionate (DMSP) and related compounds in higher plants. J Exp Bot 55:1919–1925
Simó R (2001) Production of atmospheric sulfur by oceanic plankton: biochemical, ecological, and evolutionary links. Trends Ecol Evol 16:287–294
Stefels J (2000) Physiological aspects of the production and conversion of DMSP in marine algae and higher plants. J Sea Res 43:183–197
Todd J, Rogers R, Li Y, Wexler M, Bond P, Sun L, Curson A, Malin G, Steinke M, Johnson A (2007) Structural and regulatory genes required to make the gas dimethyl sulfide in bacteria. Science 315:666–669
Van Alstyne KL (2008) Ecological and physiological roles of dimethylsulfoniopropionate (DMSP) and its products in marine macroalgae. In: Amsler CD (ed) Algal chemical ecology. Springer, Heidelberg, pp 173–194
Van Alstyne KL, Puglisi MP (2007) DMSP in marine macroalgae and macroinvertebrates: distribution, function, and ecological impacts. Aquat Sci 69:394–402
Van Alstyne KL, Wolfe GV, Freidenburg TL, Neill A, Hicken C (2001) Activated defense systems in marine macroalgae: evidence for an ecological role for DMSP cleavage. Mar Ecol Prog Ser 213:53–65
Van Alstyne KL, Schupp P, Slattery M (2006) The distribution of dimethylsulfoniopropionate (DMSP) in tropical Pacific coral reef invertebrates. Coral Reefs 25:321–327
Van Alstyne KL, Koellermeier L, Nelson T (2007) Spatial variation in dimethylsulfoniopropionate (DMSP) production in Ulva lactuca (Chlorophyta) from the Northeast Pacific. Mar Biol 150:1127–1135
Van Bergeijk SA, Stal LJ (2001) Dimethylsulfoniopropionate and dimethylsulfide in the marine flatworm Convoluta roscoffensis and its algal symbiont. Mar Biol 138:209–216
Wirtz M, Droux M (2005) Synthesis of the sulfur amino acids: cysteine and methionine. Photosynth Res 86:345–362
Wolfe GV, Steinke M, Kirst GO (1997) Grazing-activated chemical defence in a unicellular marine alga. Nature 387:894–897
Yoch DC (2002) Dimethylsulfoniopropionate: its sources, role in the marine food web, and biological degradation to dimethylsulfide. Appl Environ Microbiol 68:5804–5815
Acknowledgments
We thank A. Gehman for assistance with laboratory analyses and two anonymous reviewers for their comments, which improved this manuscript. This work was funded by National Science Foundation grants to K. Van Alstyne (OCE 0526644) and the Shannon Point Marine Center (OCE 0228618).
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Biology Editor Dr. Michael Lesser
Rights and permissions
About this article
Cite this article
Van Alstyne, K.L., Dominique, V.J. & Muller-Parker, G. Is dimethylsulfoniopropionate (DMSP) produced by the symbionts or the host in an anemone–zooxanthella symbiosis?. Coral Reefs 28, 167–176 (2009). https://doi.org/10.1007/s00338-008-0443-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00338-008-0443-y