Abstract
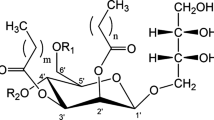
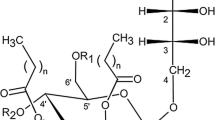
Mannosylerythritol lipids (MELs) are currently one of the most promising biosurfactants because of their multifunctional applications and good biodegradability. Depending on the yeast strain and the feedstock used for the fermentation process, structural variations in the MELs obtained occur. Therefore, MELs produced by Pseudozyma aphidis DSMZ 70725 with a soybean oil feedstock were characterized by chromatography and mass spectrometry (MS). Column chromatography with silica provided fractionation of the different types of MEL. High-performance liquid chromatography combined with MS was employed for the analysis of the MEL fractions and crude mixtures. A characteristic MS pattern for the MELs was obtained and indications of the presence of new MEL homologues, showing the incorporation of longer and more unsaturated fatty acid chains than previously reported, were given. Gas chromatography–MS analysis confirmed the presence of such unsaturated fatty acid chains in the MELs, demonstrating the incorporation of fatty acids with lengths ranging from C8 to C14 and with up to two unsaturations per chain. The incorporation of C16 and C18 fatty acid chains requires further investigation. MS/MS data allowed the unambiguous identification of the fatty acids present in the MELs. The product ion spectra also revealed the presence of a new isomeric class of MELs, bearing an acetyl group on the erythritol moiety.









Similar content being viewed by others
References
Schramm LL, Stasius EN, Gerrard Marangoni D (2003) Ann Rep Prog Chem C 99:3–48
Banat IM, Makkar RS, Cameotra SS (2000) Appl Environ Microbiol 53:495–508
Muthusamy K, Gopalakrishnan S, Ravi TK, Sivachidambaram P (2008) Biosurfactants Curr Sci 94:736–747
Mukherjee S, Das P, Sen R (2006) Trends Biotechnol 24:509–515
Cameotra SS, Makkar RS (2004) Curr Opin Microbiol 7:262–266
Desai JD, Banat IM (1997) Microbiol Mol Biol Rev 61:47–64
Van Bogaert I, Saerens K, De Muynck C, Develter D, Soetaert W (2007) Appl Microbiol Biotechnol 76:23–34
Sullivan ER (1998) Curr Opin Biotechnol 9:263–269
Silva GP, Mack M, Contiero J (2009) Biotechnol Adv 27:30–39
Lang S (2002) Curr Opin Colloid Interface Sci 7:12–20
Kitamoto D, Isoda H, Nakahara T (2002) J Biosci Bioeng 94:187–201
Im JH, Yanagishita H, Ikegami T, Takeyama Y, Idemoto Y, Koura N, Kitamoto D (2003) J Biomed Mater Res 65A:379–385
Kitamoto D, Ghosh SGO, Nakatani Y (2000) Chem Commun 1861–862
Kitamoto D, Yanagishita H, Endo A, Nakaiwa M, Nakane M, Akiya T (2001) Biotechnol Prog 17:362–365
Isoda H, Kitamoto D, Shinmoto H, Matsumura M, Nakahara T (1997) Biosci Biotechnol Biochem 61:609–614
Isoda H, Shinmoto H, Kitamoto D, Matsumura M, Nakahara T (1997) Lipids 32:263–271
Wakamatsu Y, Zhao X, Jin C, Day N, Shibahara M, Nomura N, Nakahara T, Murata T, Yokoyama KK (2001) Eur J Biochem 268:374–383
Zhao X, Wakamatsu Y, Shibahara M, Nomura N, Geltinger C, Nakahara T, Murata T, Yokoyama KK (1999) Cancer Res 59:482–486
Arutchelvi JI, Bhaduri S, Uppara PV, Doble M (2008) J Ind Microbiol Biotechnol 35:1559–1570
Morita T, Konishi M, Fukuoka T, Imura T, Kitamoto D (2008) J Biosci Bioeng 105:493–502
Rau U, Nguyen LA, Roeper H, Koch H, Lang S (2005) Appl Microbiol Biotechnol 68:607–613
Morita T, Masaaki K, Fukuoka T, Imura T, Kitamoto D (2006) Appl Microbiol Biotechnol 73:305–313
Worakitkanchanakul W, Imura T, Fukuoka T, Morita T, Sakai H, Abe M, Rujiravanit R, Chavadej S, Minamikawa H, Kitamoto D (2009) Colloids Surf B Biointerfaces 68:207–212
Fukuoka T, Morita T, Konishi M, Imura T, Kitamoto D (2008) Carbohydr Res 343:555–560
Kitamoto D, Akiba S, Hioki C, Tabuchi T (1990) Agric Biol Chem 54:31–36
Fukuoka T, Kawamura M, Morita T, Imura T, Sakai H, Abe M, Kitamoto D (2008) Carbohydr Res 343:2947–2955
Hewald S, Linne U, Scherer M, Marahiel MA, Kamper J, Bolker M (2006) Appl Environ Microbiol 72:5469–5477
Konishi M, Morita T, Fukuoka T, Imura T, Kakugawa K, Kitamoto D (2007) Appl Microbiol Biotechnol 75:521–531
Morita T, Konishi M, Fukuoka T, Imura T, Kitamoto D (2007) J Biosci Bioeng 104:78–81
Rau U, Nguyen LA, Roeper H, Koch H, Lang S (2005) Eur J Lipid Sci Technol 107:373–380
Banoub JH, Newton RP, Esmans E, Ewing DF, Mackenzie G (2005) Chem Rev 105:1869–1915
Peter-Katalinic J (2005) Methods Enzymol 405:139–171
Wiesner J, Premsler T, Sickmann A (2008) Proteomics 8:4466–4483
Cuyckens F, Claeys M (2004) J Mass Spectrom 39:1–15
Fukuoka T, Morita T, Konishi M, Imura T, Kitamoto D (2007) Biotechnol Lett 29:1111–1118
Rau U, Nguyen LA, Schulz S, Wray V, Nimtz M, Roeper H (2005) Appl Microbiol Biotechnol 66:551–559
Kitamoto D, Yanagishita H, Haraya K, Kitamoto HK (1998) Biotechnol Lett 20:813–818
Kitamoto D, Isoda H, Nakahara T (2002) J Biosci Bioeng 94:187–201
Acknowledgements
M.O. is grateful to the team at the University of Valencia for allowing him to spend some months in their laboratory. T.G. and A.C. acknowledge Research Foundation Flanders (FWO) for financial support. The authors would like to thank Dionex for providing the Acclaim surfactant column and Waters for the provision of the hydrophilic interaction liquid chromatography column. This study was financed by the University of Antwerp (KARAMEL UA-BOF project). Filip Lemière thanks the Hercules foundation for financial support in purchasing the MS instrumentation.
Author information
Authors and Affiliations
Corresponding author
Additional information
Footnote: Published in the special issue Advances in Analytical Separations with Guest Editors Yolanda Pico and Joan O. Grimalt.
Rights and permissions
About this article
Cite this article
Onghena, M., Geens, T., Goossens, E. et al. Analytical characterization of mannosylerythritol lipid biosurfactants produced by biosynthesis based on feedstock sources from the agrofood industry. Anal Bioanal Chem 400, 1263–1275 (2011). https://doi.org/10.1007/s00216-011-4741-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00216-011-4741-9