Abstract
Rationale
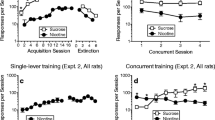
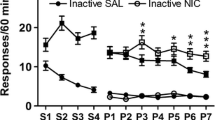
Nicotine self-administration in rats is modest when response-contingent nicotine infusions are delivered alone (primary reinforcement) but robust when nicotine infusions are combined with a mildly reinforcing non-pharmacological stimulus. Furthermore, response-independent (non-contingent) nicotine administration also elevates responding for that same non-pharmacological stimulus, suggesting that in addition to primary reinforcement, nicotine can enhance the incentive value of other reinforcers.
Objectives
In this study, we tested the hypothesis that the reinforcement-enhancing effects of non-contingent nicotine are more dependent on the reinforcing strength of the non-pharmacological stimulus than are the effects of contingent nicotine.
Materials and methods
A weakly reinforcing light-tone stimulus was established as a conditioned reinforcer by repeated pairings with sucrose for some rats, or by delivery in an explicitly unpaired design with sucrose to other rats. Subsequently, both groups lever pressed for the stimulus with contingent nicotine, non-contingent nicotine (0.06 mg kg−1 per infusion, freebase), or non-contingent saline, according to fixed ratio and progressive ratio reinforcement schedules.
Results
Compared to sucrose-unpaired training, repeated association with sucrose established the light-tone stimulus as a robust conditioned reinforcer. Contingent and non-contingent nicotine equally elevated responding for this conditioned stimulus. Conversely, for the less reinforcing (sucrose-unpaired) stimulus contingent nicotine more effectively elevated behavior compared to non-contingent nicotine.
Conclusions
The reinforcement-enhancing effect of nicotine increases behavior controlled by both conditioned and unconditioned reinforcers; however, for less salient stimuli associative processes derived from the primary reinforcing effects of contingent nicotine may also be important. These data suggest that nicotine present in tobacco may differentially modulate stimulus-driven behavior in smokers.





Similar content being viewed by others
References
Balfour DJ (2004) The neurobiology of tobacco dependence: a preclinical perspective on the role of the dopamine projections to the nucleus. Nicotine Tob Res 6:899–912
Balfour DJ, Wright AE, Benwell ME, Birrell CE (2000) The putative role of extra-synaptic mesolimbic dopamine in the neurobiology of nicotine dependence. Behav Brain Res 113:73–83
Brody AL, Mandelkern MA, London ED, Childress AR, Lee GS, Bota RG, Ho ML, Saxena S, Baxter LR Jr, Madsen D, Jarvik ME (2002) Brain metabolic changes during cigarette craving. Arch Gen Psychiatry 59:1162–1172
Caggiula AR, Donny EC, White AR, Chaudhri N, Booth S, Gharib MA, Hoffman A, Perkins KA, Sved AF (2001) Cue dependency of nicotine self-administration and smoking. Pharmacol Biochem Behav 70:515–530
Caggiula AR, Donny EC, White AR, Chaudhri N, Booth S, Gharib MA, Hoffman A, Perkins KA, Sved AF (2002a) Environmental stimuli promote the acquisition of nicotine self-administration in rats. Psychopharmacology (Berl) 163:230–237
Caggiula AR, Donny EC, Chaudhri N, Perkins KA, Evans-Martin FF, Sved AF (2002b) Importance of nonpharmacological factors in nicotine self-administration. Physiol Behav 77:683–687
Chaudhri N, Caggiula AR, Donny EC, Booth S, Gharib MA, Craven LA, Allen SS, Sved AF, Perkins KA (2005) Sex differences in the contribution of nicotine and nonpharmacological stimuli to nicotine self-administration in rats. Psychopharmacology (Berl) 180:258–266
Chaudhri N, Caggiula AR, Donny EC, Palmatier MI, Liu X, Sved AF (2006) Complex interactions between nicotine and nonpharmacological stimuli reveal multiple roles for nicotine in reinforcement. Psychopharmacology (Berl) 184:353–366
Childress AR, Mozley PD, McElgin W, Fitzgerald J, Reivich M, O’Brien CP (1999) Limbic activation during cue-induced cocaine craving. Am J Psychiatry 156(1):11–18
Cohen C, Perrault G, Griebel G, Soubrie P (2005) Nicotine-associated cues maintain nicotine-seeking behavior in rats several weeks after nicotine withdrawal: reversal by the cannabinoid (CB1) receptor antagonist, rimonabant (SR141716). Neuropsychopharmacology 30:145–155
Corrigall WA, Coen KM (1989) Nicotine maintains robust self-administration in rats on a limited-access schedule. Psychopharmacology (Berl) 99:473–478
Corrigall WA, Coen KM (1991) Selective dopamine antagonists reduce nicotine self-administration. Psychopharmacology (Berl) 104:171–176
Corrigall WA, Franklin KB, Coen KM, Clarke PB (1992) The mesolimbic dopaminergic system is implicated in the reinforcing effects of nicotine. Psychopharmacology (Berl) 107:285–289
Corrigall WA, Coen KM, Adamson KL (1994) Self-administered nicotine activates the mesolimbic dopamine system through the ventral tegmental area. Brain Res 653:278–284
Corrigall WA, Coen KM, Adamson KL, Chow BL, Zhang J (2000) Response of nicotine self-administration in the rat to manipulations of mu-opioid and gamma-aminobutyric acid receptors in the ventral tegmental area. Psychopharmacology (Berl) 149:107–114
Dani JA, De Biasi M (2001) Cellular mechanisms of nicotine addiction. Pharmacol Biochem Behav 70:439–446
Depoortere RY, Li DH, Lane JD, Emmett-Oglesby MW (1993) Parameters of self-administration of cocaine in rats under a progressive-ratio schedule. Pharmacol Biochem Behav 45:539–548
Di Ciano P, Everitt BJ (2004) Conditioned reinforcing properties of stimuli paired with self-administered cocaine, heroin or sucrose: implications for the persistence of addictive behaviour. Neuropharmacology 47(Suppl 1):202–213
Donny EC, Chaudhri N, Caggiula AR, Evans-Martin FF, Booth S, Gharib MA, Clements LA, Sved AF (2003) Operant responding for a visual reinforcer in rats is enhanced by non-contingent nicotine: implications for nicotine self-administration and reinforcement. Psychopharmacology (Berl) 169:68–76
Donny EC, Caggiula AR, Mielke MM, Booth S, Gharib MA, Hoffman A, Maldovan V, Shupenko C, McCallum SE (1999) Nicotine self-administration in rats on a progressive ratio schedule of reinforcement. Psychopharmacology (Berl) 147:135–142
Donny EC, Caggiula AR, Mielke MM, Jacobs KS, Rose C, Sved AF (1998) Acquisition of nicotine self-administration in rats: the effects of dose, feeding schedule, and drug contingency. Psychopharmacology (Berl) 136:83–90
Donny EC, Caggiula AR, Knopf S, Brown C (1995) Nicotine self-administration in rats. Psychopharmacology (Berl) 122:390–394
Due DL, Huettel SA, Hall WG, Rubin DC (2002) Activation in mesolimbic and visuospatial neural circuits elicited by smoking cues: evidence from functional magnetic resonance imaging. Am J Psychiatry 159:954–960
Field M, Duka T (2002) Cues paired with a low dose of alcohol acquire conditioned incentive properties in social drinkers. Psychopharmacology (Berl) 159(3):325–334
Fletcher PJ, Korth KM, Chambers JW (1999) Selective destruction of brain serotonin neurons by 5,7-dihydroxytryptamine increases responding for a conditioned reward. Psychopharmacology (Berl) 147:291–299
Goldberg SR, Spealman RD, Goldberg DM (1981) Persistent behavior at high rates maintained by intravenous self-administration of nicotine. Science 214:573–575
Jose Lanca A, Sanelli TR, Corrigall WA (2000) Nicotine-induced fos expression in the pedunculopontine mesencephalic tegmentum in the rat. Neuropharmacology 39:2808–2817
Lanca AJ, Adamson KL, Coen KM, Chow BL, Corrigall WA (2000) The pedunculopontine tegmental nucleus and the role of cholinergic neurons in nicotine self-administration in the rat: a correlative neuroanatomical and behavioral study. Neuroscience 96:735–742
Lehman RS (1995) Statistics in the behavioral sciences. A conceptual approach. Brooks/Cole Publishing Company, Brooks/Cole Publishing Company
Lesage MG, Burroughs D, Dufek M, Keyler DE, Pentel PR (2004) Reinstatement of nicotine self-administration in rats by presentation of nicotine-paired stimuli, but not nicotine priming. Pharmacol Biochem Behav 79:507–513
Liu X, Caggiula AR, Palmatier MI, Donny EC, Chaudhri N, Sved AF (2005) Reinforcement-enhancing effect of nicotine depends on the reinforcement valence of nondrug stimulus. Society for Neuroscience Abstract 1027.14
Mansvelder HD, McGehee DS (2000) Long-term potentiation of excitatory inputs to brain reward areas by nicotine. Neuron 27:349–357
Markou A, Weiss f, Gold LH, Caine SB, Schulteis G, Koob GF (1993) Animal models of drug craving. Psychopharmacology (Berl) 112(2–3):163–182
Matteson GL, Palmatier MI, Liu X, Chaudhri N, Caggiula AR, Donny EC, Sved AF (2006) The reinforcement enhancing effect of nicotine depends on the incentive value of a nonpharmacological reinforcer. The 12th annual meeting of the Society for Research on Nicotine and Tobacco, Orlando, FL, February 15–18, 2006
Olausson P, Jentsch JD, Taylor JR (2004a) Nicotine enhances responding with conditioned reinforcement. Psychopharmacology (Berl) 171:173–178
Olausson P, Jentsch JD, Taylor JR (2004b) Repeated nicotine exposure enhances responding with conditioned reinforcement. Psychopharmacology (Berl) 173:98–104
Palmatier MI, Evans-Martin FF, Hoffman A, Caggiula AR, Chaudhri N, Donny E, Liu X, Booth S, Gharib MA, Craven LA, Sved AF (2006) Dissociating the primary reinforcing and reinforcement-enhancing effects of nicotine using a rat self-administration paradigm with concurrently available drug and environmental reinforcers. Psychopharmacology (Berl) 184:391–400
Paterson NE, Froestl W, Markou A (2005) Repeated administration of the GABAB receptor agonist CGP44532 decreased nicotine self-administration, and acute administration decreased cue-induced reinstatement of nicotine-seeking in rats. Neuropsychopharmacology 30:119–128
Perkins KA, Gerlach D, Vender J, Grobe J, Meeker J, Hutchison S (2001) Sex differences in the subjective and reinforcing effects of visual and olfactory cigarette smoke stimuli. Nicotine Tob Res 3:141–150
Pidoplichko VI, Noguchi J, Areola OO, Liang Y, Peterson J, Zhang T, Dani JA (2004) Nicotinic cholinergic synaptic mechanisms in the ventral tegmental area contribute to nicotine addiction. Learn Mem 11:60–69
Reid MS, Mickalian JD, Delucchi KL, Berger SP (1999) A nicotine antagonist, mecamylamine, reduces cue-induced cocaine craving in cocaine-dependent subjects. Neuropsychopharmacology 20:297–307
Rice ME, Cragg SJ (2004) Nicotine amplifies reward-related dopamine signals in striatum. Nat Neurosci 7:583–584
Robbins TW (1978) The acquisition of responding with conditioned reinforcement: effects of pipradrol, methylphenidate, d-amphetamine, and nomifensine. Psychopharmacology (Berl) 58:79–87
Robbins TW, Koob GF (1978) Pipradrol enhances reinforcing properties of stimuli paired with brain stimulation. Pharmacol Biochem Behav 8:219–222
Rose JE (2005) Nicotine and nonnicotine factors in cigarette addiction. Psychopharmacology (Berl):1–12
Rose JE, Levin ED (1991) Inter-relationships between conditioned and primary reinforcement in the maintenance of cigarette smoking. Br J Addict 86:605–609
Stafford D, LeSage MG, Glowa JR (1998) Progressive-ratio schedules of drug delivery in the analysis of drug self-administration: a review. Psychopharmacology (Berl) 139(3):169–184
Stewart J (1960) Reinforcing effects of light as a function of intensity and reinforcement schedule. J Comp Physiol Psychol 53:187–193
Taylor JR, Robbins TW (1984) Enhanced behavioural control by conditioned reinforcers following microinjections of d-amphetamine into the nucleus accumbens. Psychopharmacology (Berl) 84:405–412
Acknowledgements
The authors would like to thank Dr. Peter Olausson for his thoughtful advice about the conditioning procedures used in this experiment. “Principles of laboratory animal care” (NIH No. 85-23, revised 1985) were followed throughout all experiments. The University of Pittsburgh Institutional Animal Care and Use Committee, Assurance Number A3187-01 approved this research. Preparation of this review was supported by National Institute on Drug Abuse research grants DA-10464 and DA-12655 and by a Howard Hughes Predoctoral Research Fellowship awarded to N. Chaudhri.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chaudhri, N., Caggiula, A.R., Donny, E.C. et al. Operant responding for conditioned and unconditioned reinforcers in rats is differentially enhanced by the primary reinforcing and reinforcement-enhancing effects of nicotine. Psychopharmacology 189, 27–36 (2006). https://doi.org/10.1007/s00213-006-0522-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00213-006-0522-0