Abstract
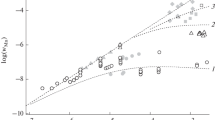
The kinetics of the permanganic oxidation process of glycine, L-alanine and L-leucine in strong acid media were investigated using a spectrophotometric technique. Conclusive evidence has proven that the autocatalytic activity of Mn(II) in these reactions in strong acidic media is analogous to that of weak acid media, but in the former, Mn(II) ions should acquire a critical concentration for them to show autocatalytic characteristics. This critical concentration depends on the nature of the amino acid used. Considering the delayed autocatalytic behavior of Mn(II) ions, we herein present the rate equations and mechanisms satisfying observations for both catalytic and noncatalytic routes. The correspondence of the pseudo-order rate constants of the catalytic and noncatalytic pathways to Eyring law verify both the critical concentration as well as the delayed autocatalytic behavior concepts. In general, the onset of delayed behavior can be attributed to the concentration ratio of Mn(II) to amino acid which can be of a certain value for any particular amino acid.
Similar content being viewed by others
References
R. S. Verma, M. J. Reddy, V. R. Shastry, J. Chem. Soc. Perkin Trans 2 (1976) 469.
A. A. Frost, R.G. Pearson, Kinetic and Mechanisms, Wiley, New York, 1961, p. 152.
C. S. Ameta, P. N. Pande, H. L. Gupta, H. C. Chowhhry, Acta Phys. Chem. 26 (1980) 89.
C. S. Ameta, P. N. Pande, H. L. Gupta, H. C. Chowhhry, Z. Phys. Chem. (Leipzig), 261 (1980) 1222.
C. S. Ameta, P. N. Pande, H. L. Gupta, H. C. Chowhhry, Z. Phys. Chem. (Leipzig), 261 (1980) 802.
C. S. Ameta, P. N. Pande, H. L. Gupta, H. C. Chowhhry, Acta Chim. Acad. Sci. Hung. 110 (1982) 7.
L. M. Bharadwaj, P. C. Nigam, Ind. J. Chem. 8A (1981) 793.
V. S. Rao, B. Sethuram, T.N. Rao, Int. J. Chem. Kinet. 11 (1979) 165.
V. S. Rao, B. Sethuram, T.N. Rao, Oxid. Commun. 9 (1986) 11.
U. D. Mudaliar, V.R. Chourey, R.S. Verma, V.R. Shastry, Ind. J. Chem. Soc. 60 (1983) 561.
H. M. Girgis, R.M. Hassan, A.S. El-Shahawy, Bull. Fac. Sci. Univ. 16 (1987) 41.
R. M. Hassan, M.A. Mousa, M.H. Wahdan, J. Chem. Soc. Dalton. Trans 3 (1988) 605.
H. Iloukani, H. Bahrami, Int. J. Chem. Kinet. 31 (1999) 95.
B. R. Sahu, V.R. Chourey, S. Pandey, L.V. Shastry, V. R. Shastry, Ind. J. Chem. Soc. 76 (2000) 131.
H. Iloukhani, N. Rashidi, M. Moghadasi, Asian J. Chem. 12 (2000) 1209.
H. Iloukhani, S.R. Ekvan, A. A. Rafati, Phys. Chem. Liquids 41 (2003) 25.
M. Moghadasi, N. Rashidi, H. Iloukhani, Phys. Chem. Liquids 2001 (39) 267.
H. Iloukhani, M. Moazenzadeh, Phys. Chem. Liquids 39 (2001) 429.
M. Zahedi, H. Bahrami, Kinet. Catal. 45 (2004) 351.
H. Bahrami, M. Zahedi, Can. J. Chem. 82 (2004) 430.
H. Bahrami, M. Zahedi, Int. J. Chem. Kinet. 38 (2006) 1.
A. I. Vogel, Quimica Analitica Cuantitative, Vol. 1, Kapelusz, Buenos Aires, 1960, p. 382.
F. Felig, Spot Tests in Inorganic Analysis, Elsevier, Amsterdam, 1972, p. 334
A. I. Vogel, Quimica Analitica Cuantitative, Vol. 1, Kapelusz, Buenos Aires, 1953, p. 250.
R. M. Roberts, J. C. Gilbert, L. B. Rodwald, A. S. Wingrove, Modern Experimental Organic Chemistry, 2nd ed., Saunders, Philadelphia, 1985, p. 700.
K. K. Banerji, P. Nath, Bull. Chem. Soc. Jap. 42 (1969) 2038.
F. J. Andrés Ordax, A. Arrizabalaga, J.I. Martinez de Ilarduya, An. Quim. 80 (1984) 531.
F. J. Andrés Ordax, A. Arrizabalaga, R. Martinez Perez de mendiola, Studia. Chemica. 11 (1986) 303.
F. J. Andrés Ordax, A. Arrizabalaga, K. Ortega, An. Quim. 85 (1989) 218.
J. F. Perez Benito, F. Mata Perez, E. Brillas, Can. J. Chem. 65 (1987) 2329.
E. Brillas, J. A. Garrido, J. F. Perez Benito, Collect. Czech. Chem. Comun. 53 (1988) 479.
J. A. Garrido, J. F. Perez Benito, R. M. Rodrigouez, J. De Andrés, E. Brillas, J. Chem. Res. 11 (1987) 380.
J. De Andrés, E. Brillas, J. A. Garrido, J. F. Perez Benito, J. Chem. Soc. Perkin Trans 2 (1988) 107.
R. M. Rodrigouez, J. De Andrés, E. Brillas, J. A. Garrido, J. F. Perez Benito, New J. Chem. 2 (1988) 143.
J. De Andrés, E. Brillas, J. A. Garrido, J. F. Perez Benito, Gazz. Chim. Ital. 118 (1988) 203.
K. A. Kovacs, P. Grof, L. Burai, M. Riedel, J. Phys. Chem. A 108 (2004) 11026.
W. A. Waters, Q. Rev. Chem. Soc. 12 (1958) 277.
N. Ganapathisubramanian, J. Phys. Chem. 92 (1988) 414.
R. T. Powell, T. Oskin, N. Ganapathisubramanian, J. Phys. Chem. 93 (1989) 2718.
A. Arrizabalaga, F. J. Andrés Ordax, M. Y. Fernández Aránguiz, R. Peche, Int. J. Chem. Kinet. 29 (1997) 181.
F. J. Andrés Ordax, A. Arrizabalaga, J. Casado, R. Peche, React. Kinet. Catal. Lett. 44 (1991) 293.
F. J. Andrés Ordax, A. Arrizabalaga, R. Peche, M.A. Quintana, An. Quim. 87 (1992) 828.
F. J. Andrés Ordax, A. Arrizabalaga, R. Peche, M.A. Quintana, An. Quim. 88 (1992) 440.
M. J. Insausti, F. Mata-Pèrez, M.P. Alvarez-Macho, Int. J. Chem. Kinet. 27 (1995) 507.
A. Arrizabalaga, F. J. Andrés Ordax, M.Y. Fernández Aránguiz, R. Peche. Int. J. Chem. Kinet. 28 (1996) 799.
W. A. Waters, Q. Rev. Chem. Soc. 12 (1958) 277.
P. S. Radhakrishnanurti, M. D. Rao, Indian J. Chem. Soc. A 15 (1977) 524.
Y. Takezaki, C. Takeuchi, J. Chem. Phys. 22 (1954) 1527.
J.W. Moore, R. G. Pearson, Kinetic and Mechanisms, Wiley, New York, 1981, p. 42.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bahrami, H., Zahedia, M. Conclusive evidence for delayed autocatalytic behavior of Mn(II) ions at a critical concentration. JICS 5, 535–545 (2008). https://doi.org/10.1007/BF03246131
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF03246131