Summary
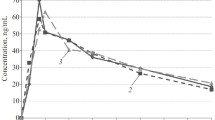
The interaction of diclofenac and ketoprofen, both applied intraperitoneally in a dose of 8 mg/kg for twenty-eight days, was assessed with cardioactive drugs in rats. Interaction was assessed on the basis of ECG records after the infusion of adrenaline, verapamil or lidocaine to the rats treated with diclofenac or ketoprofen vs control. The infusion time was measured in seconds to the moment of the appearance of the first heart reaction to the infusion of the cardioactive drug, then to the appearance of more frequent changes in the ECG record, and finally, to the occurrence of the toxic effect. It was also measured the plasma concentrations of sodium and potassium ions. As well as diclofenac and ketoprofen concentration, 2 hours after single and 28th dose. ECG patterns revealed no occurrence of cardiotoxic action of diclofenac and ketoprofen. The treatment with diclofenac caused significantly lower sodium plasma concentrations whereas the concentration of potassium was increased. Diclofenac concentrations were the same after a single and multiple doses, whereas concentrations of ketoprofen were significantly higher after a single dose than after its multiple applications.
Similar content being viewed by others
References
McGettigan P., Henry D. (2006): Cardiovascular risik and inhibition of cyclooxygenase: a systematic review of the observational studies of selective and nonselective inhibitors of cyclooxigenase 2. JAMA, 296, 1633–1634.
Ray W.A., Stein C.M., Hall K. (2002): Daugherty J.R., Griffin M.R. Non steroidal anti-inflammatory drugs and risk of serious coronary heart disease; an observational cohort. Lancet, 359, 118–123.
Andersohn F. (2006): Use of first- and second-generation cyclooxygenase-2-selective nonsteroidal anti-inflammatory drugs and risk of acute myocardial infarction. Circulation, 113, 1950–1957.
Motola D., Vaccheri A., Silvani M.C., Poluzzi E., Bottoni A., De Ponti F., Montanaro N. (2004): Pattern of NSAID use in the Italian general population: a questionnaire-based survey. Eur. J. Clin. Pharmacol., 60, 731–738.
Jick H., Kaye J.A., Russmann S., Jick S.S (2006): Nonsteroidal antiimflammatory drugs and acute myocardial infarction in patients with no major risk factors. Pharmacotherapy, 26, 1379–1387.
EMEA press release on non-selective NSAIDS. August 2nd, 2005,http://www.emea.eu.int/pdfs/humanlpress/pr/24732305en. pdf Accessed 23 August 2006.
Arun P.R., Azeez P.A. (2004): Vulture population decline, diclofenac and avian gout. Cun. Sci., 87, 565–568.
Singh G., Wu 0., Ranghorne P., Madhok R. (2006): Risk of acute myocardial infarction with nonselective non-steroidal anti-inflammatory drugs: a meta-analysis. Arthritis Res. Ther., 8, 153–158.
Jakovljevic V., Stanulovic M., Sabo A., Popovic M.(1989): Effect of chronic consumption of caffeine on the autonomous system of isolated rat heart. Yugoslav. Physiol. Pharmacol. Acta, 25,51–53.
Jakovljevic V., Sabo A., Popovic M., Stanulovic M. (1990): Interaction between methaixantine and cardioactive drugs. (Abstract). Eur. J. Pharmacol., 183.
Vasovic V., Vukmirovic S., Poša M., Mikov M., Raškovic A., Jakovljevic V. (2006): Effect of rat pretreatment with aqueous solution of stevioside and bile acids on the action of certain cardioactive drugs. Eur. J. Drug Metabol. Pharmacokinet., 31, 311–314.
Turner R.A.: Screening Methods in Pharmacology, Academic Press, New York and London, 1965.
Owen S.G., Michael S.R., William T.F. (1987): Rapid High-performance liquid chromatographic assay for the simultaneus analysis of non-steroidal antiinflamatory drugs in plasma. J. Chromatogr., 416, 293–302.
USP. The United States Pharmacopeia. The national formulary. USP 26th revision (January 1, 2003). NF 21st ed (Januar 1, 2003). Rockville, MD: The United States Pharmacopoeia Convention, Inc., 2002.
Clasco R.K., Ed.: USP DI Drug information for the Health Care P professional. Thomson MICROMEDEX, Inc 2003 P. IV/3 17
Johnston S.A., Fox S.M. (1997): Mechanism of action of anti-inflammatory medications used for the treatment of osteoarthritis. J. Am. Veterin. Med. Assoc., 210, 1486–1492.
Oaks J.L., Gilbert M., Virani M.Z., Watson R.T., Meteyer C.U., Rideout B.A., Shivaprasad H.L., Ahmed S., Chaudhry M.J., Arshad M., Mahmood S., Au A., Khan A.A (2004): Diclofenac residues as the cause of vulture population decline in Pakistan. Nature, 427, 630–633.
Noory T., Jarrar B.M. (2006): Histochemical Alterations in the Spleen of Rabbits Induced by Diclofenac Sodium (Voltaren). Science, 19, 21–29.
Masubuchi Y., Nakayama S., Hone T. (2002): Role of mitochondrial permeability transition in diclofenac-induced hepatocyte injury in rats. Hepatology, 35, 544–551.
Hunter R.P., Isaza R., Koch D.E. (2003): Oral bioavailability and pharmacokinetic characteristics of ketoprofen enantiomers after oral and intravenous administration in Asian elephants (Elephas maximus). Am. J. Veterin. Res., 64, 109–114.
Ishizaki T., Sasaki T., Suganuma T., Horai Y., Chiba K., Watanabe M., Asuke W., Hoshi H. (1980): Pharmacokinetics of ketoprofen following single oral, intramuscular and rectal doses and after repeated oral. Eur. J. Clin. Pharmacol., 18, 407–414.
Aberg G., Ciofalo V.B., Pendleton R.G., et al. (1995): Inversion of (R)- to (S)-ketoprofen in eight animal species. Chirality, 7, 383–387.
Lees P., Taylor P.M., Landoni F.M., Arifah A.K., Waters C. (2003): Ketoprofen in the Cat. Pharmacodyn. Chiral Pharmacokinet. Veterin. J. 165, 21–35.
McGettigan P., Henry D. (2006): Cardiovascular risik and inhibition of cyclooxygenase: a systematic review of the observational studies of selective and nonselective inhibitors of cyclooxigenase 2. JAMA, 296, 1633–1634.
Ray W.A., Stein C.M., Hall K. (2002): Daugherty J.R., Griffin M.R. Non steroidal anti-inflammatory drugs and risk of serious coronary heart disease; an observational cohort. Lancet, 359, 118–123.
Andersohn F. (2006): Use of first- and second-generation cyclooxygenase-2-selective nonsteroidal anti-inflammatory drugs and risk of acute myocardial infarction. Circulation, 113, 1950–1957.
Motola D., Vaccheri A., Silvani M.C., Poluzzi E., Bottoni A., De Ponti F., Montanaro N. (2004): Pattern of NSAID use in the Italian general population: a questionnaire-based survey. Eur. J. Clin. Pharmacol., 60, 731–738.
Jick H., Kaye J.A., Russmann S., Jick S.S (2006): Nonsteroidal antiimflammatory drugs and acute myocardial infarction in patients with no major risk factors. Pharmacotherapy, 26, 1379–1387.
EMEA press release on non-selective NSAIDS. August 2nd,2005) http://www.emea.eu.intlpdfs/humanlpress/pr/24732305en.pd f Accessed 23 August 2006.
Arun P.R., Azeez P.A. (2004): Vulture population decline, diclofenac and avian gout. Curr. Sci., 87, 565–568.
Singh G., Wu O., Ranghorne P., Madhok R. (2006) Risk of acute myocardial infarction with nonselective non-steroidal anti-inflammatory drugs: a meta-analysis. Arthritis Res. Ther., 8, 153–158.
Jakovljevic V., Stanulovic M., Sabo A., Popovic M.(l989): Effect of chronic consumption of caffeine on the autonomous system of isolated rat heart. Yugoslav. Physiol. Pharmacol. Acta, 25, 51–53. 33. Jakovljevic V., Sabo A., Popovic M., Stanulovic M. (1990): Interaction between methalxantine and cardioactive drugs. (Abstract). Eur. J. Pharmacol., 183.
Vasovic V., Vukmirovic S., Poša M., Mikov M., Raškovic A., Jakovljevic V. (2006): Effect of rat pretreatment with aqueous solution of stevioside and bile acids on the action of certain cardioactive drugs. Eur. J. Drug Metabol. Pharmacokinet., 31,311–314.
Turner R.A.: Screening Methods in Pharmacology, Academic Press, New York and London, 1965.
Owen S.G., Michael S.R., William T.F. (1987): Rapid High-performance liquid chromatographic assay for the simultaneus analysis of non-steroidal antiinflamatory drugs in plasma. J. Chromatogr., 416, 293–302.
USP. The United States Pharmacopeia. The national formulary. USP 26th revision (January 1, 2003). NF 2lst ed (January 1, 2003). Rockville, MD: The United States Pharmacopoeia Convention, Inc., 2002.
Clasco R.K., Ed.: USP DI Drug information for the Health Care P professional. Thomson MICROMEDEX, Inc 2003 P. IV/317
Johnston S.A., Fox S.M. (1997): Mechanism of action of anti-inflammatory medications used for the treatment of osteoarthritis. J. Am. Veterin. Med. Assoc., 210, 1486–1492.
Oaks J.L., Gilbert M., Virani M.Z., Watson R.T., Meteyer C.U., Rideout B.A., Shivaprasad H.L., Ahmed S., Chaudhry M.J., Arshad M., Mahmood S., Ali A., Khan A.A (2004): Diclofenac residues as the cause of vulture population decline in Pakistan. Nature, 427, 630–633.
Noory T., Jarrar B.M.(2006): Histochemical Alterations in the Spleen of Rabbits Induced by Diclofenac Sodium (Voltaren). Science, 19, 21–29.
Masubuchi Y., Nakayama S., Hone T. (2002): Role of mitochondrial permeability transition in diclofenac-induced hepatocyte injury in rats. Hepatology, 35, 544–551.
Hunter R.P., Isaza, R., Koch D.E. (2003): Oral bioavailability and pharmacokinetic characteristics of ketoprofen enantiomers after oral and intravenous administration in Asian elephants (Elephas maximus). Am. J. Veterin. Res., 64, 109–114.
Ishizaki T., Sasaki T., Suganuma T., Horai Y., Chiba K., Watanabe M., Asuke W., Hoshi H. (1980): Pharmacokinetics of ketoprofen following single oral, intramuscular and rectal doses and after repeated oral. Eur. J. Clin. Pharmacol., 18, 407–414.
Aberg G., Ciofalo V.B, Pendleton R.G., et al. (1995): Inversion of (R)- to (S)-ketoprofen in eight animal species. Chirality, 7, 383–387.
Lees P., Taylor P.M., Landoni F.M., Arifah A.K., Waters C. (2003): Ketoprofen in the Cat: Pharmacodyn. Chiral Pharmacokinet. Veterin. J. 165, 21–35.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Jakovljevic, V., Sabo, A., Tomić, Z. et al. Interaction of diclofenac and ketoprofen with cardioactive drugs in rats. Eur. J. Drug Metabol. Pharmacokinet. 34, 11–17 (2009). https://doi.org/10.1007/BF03191378
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF03191378