Summary
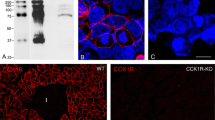
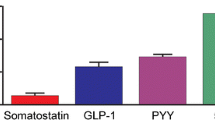
By employing polyclonal antibodies for retinol-binding protein (RBP), its distribution in the human pancreas and digestive tract mucosa was compared with those of transthyretin (TTR) and various peptide hormones. The materials used included surgically removed pancreas, esophagus, stomach, small and large intestines. Paraffin sections were stained by the indirect immunoenzyme method. The results indicate that RBP-containing cells are found in the pancreas and the gastro-intestinal mucosa, but most frequently in the gastric antrum and duodenum. In the pancreas, RBP-containing cells are found in the islets and among acinar and ductal epithelial cells, and consistently stain for chromogranin A. RBP-containing cells in the gastrointestinal mucosa showed typical features of endocrine cells and also stained for chromogranin A. The distribution of TTR in these tissue sites resembled that of RBP, but the immunoreactive intensities of both peptides altered independently. Comparison of the distribution of RBP, TTR, and various gastrointestinal peptide hormones revealed that the distribution of RBP coincided with none of the other peptides, although some of the RBP-containing cells stained for most of the peptides examined and vice versa. These results suggest that RBP may be a consistent component of gastrointestinal endocrine cells.
Similar content being viewed by others
References
Goodman DS (1984) Plasma retinol-binding protein. In: Sporn MB, Roberts AB, Goodman DS (eds) The retinoids, vol 2. Academic Press, New York, pp 41–88
Jacobsson B, Carlström A, Collins VP, Grimelius L (1989a) Trans-thyretin in endocrine pancreatic tumors. Am J Pathol 134: 465–471
Jacobsson B, Collins VP, Grimelius L, Pettersson T, Sandstedt B, Carlström A (1989b) Transthyretin immunoreactivity in human and porcine liver, choroid plexus, and pancreatic islets. J Histochem Cytochem 37: 31–37
Kameko M, Ichikawa M, Katsuyama T, Kanai M, Kato M, Akamatsu T (1986) Immunohistochemical localization of plasma retinol-binding protein and prealbumin in human pancreatic islets. Histochem J 18: 164–168
Kanai M, Raz A, Goodman DS (1968) Retinol-binding protein: the transport protein for vitamin A in human plasma. J Clin Invest 47: 2025–2044
Katoh M, Kanai M, Kameko M, Ohno S, Fujii Y, Nagata T (1982) Localization of retinol-binding protein and prealbumin in the human kidney with an unlabeled enzyme immunohistochemical method. Acta Histochem Cytochem 15: 68–75
Kato M, Kato K, Blaner WS, Chertow BS, Goodman DS (1985) Plasma and cellular retinoid-binding proteins and transthyretin (prealbumin) are all localized in the islets of Langerhans in the rat. Proc Natl Acad Sci USA 82: 2488–2492
Katsuyama T, Ono K, Nakayama J, Kanai M (1985) Recent advances in mucosubstance histochemistry. In: Kawai K (ed) Gastric mucus and mucous secreting cells. Excerpta Medica, Amsterdam, pp 3–18
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 277: 680–685
Makover A, Soprano DR, Wyatt ML, Goodman DS (1989a) Localization of retinol-binding protein messenger RNA in the rat kidney and in perinephric fat tissue. J Lipid Res 30: 171–180
Makover A, Soprano DR, Wyatt ML, Goodman DS (1989b) An in situ-hybridization study of the localization of retinol-binding protein and transthyretin messenger RNAs during fetal development in the rat. Differentiation 40: 17–25
Nanba K, Aoki J, Sasaki N (1987) A new enzyme immunohistochemical technique using alkaline phosphatase-labeled avidin and new fuchsin. Pathology and Clinical Medicine 5: 333–339 (in Japanese)
Ota H, Katsuyama T, Ishii K, Nakayama J, Shiozawa T, Tsukahara Y (1991) A dual staining method for identifying mucins of different gastric epithelial mucous cells. Histochem J 23: 22–28
Polak JM (1989) Endocrine cells of the gut. In: Schults SG, Makhlouf GM, Rauner BB (eds) Handbook of physiology, vol 2. Oxford University Press, New York, pp 79–96
Rindi G, Buffa R, Sessa F, Tortora O, Solcia E (1986) Chromogranin A, B and C immunoreactivities of mammalian endocrine cells. Distribution, distinction from costored hormones/prohormones and relationship with the argyrophil component of secretory granules. Histochemistry 85: 19–28
Smith JE, Muto Y, Goodman DS (1975) Tissue distribution and subcellular localization of retinol-binding protein in normal and vitamin A-deficient rats. J Lipid Res 16: 318–323
Soprano DR, Herbert J, Soprano KJ, Schon EA, Goodman DS (1985) Demonstration of transthyretin mRNA in the brain and other extrahepatic tissues in the rat. J Biol Chem 260: 11793–11798
Soprano DR, Soprano KJ, Goodman DS (1986) Retinol-binding protein messenger RNA levels in the liver and in extrahepatic tissues of the rat. J Lipid Res 27: 166–171
Suhara A, Kato M, Kanai M (1990) Ultrastructural localization of plasma retinol-binding protein in rat liver. J Lipid Res 31: 1669–1681
Towbin H, Staehelin T, Gordon J (1979) Electrophoretic transfer of proteins from polyacrylamide gels to nitrocellulose sheets: procedure and some applications. Proc Natl Acad Sci USA 76: 4350–4354
Tsutsumi Y, Osamura RY, Nagura H, Watanabe K, Yanaihara N (1983) Immunohistochemical studies on gastrointestinal hormones in the intestinal metaplasia of the stomach. In: Miyoshi A (ed) Gut peptides and ulcer, Biomedical Research Foundation, Tokyo, pp 171–179
Usuda N, Kameko M, Kanai M, Nagata T (1983) Immunocytochemical demonstration of retinol-binding protein in the lysosomes of the proximal tubules of the human kidney. Histochemistry 78: 487–490
Yanaihara C (1989) Sequences of natural gut peptides, related peptides, and their precursors. In: Schults SG, Makhlouf GM, Rauner BB (eds) Handbook of physiology, vol 2. Oxford University Press, New York, pp 45–62
Wilson BS, Lloyd RV (1984) Detection of chromogranin in neuroendocrine cells with a monoclonal antibody. Am J Pathol 115: 458–468
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kameko, M., Ota, H., Ishii, K. et al. Distribution of retinol-binding protein in the human digestive tract. Virchows Archiv B Cell Pathol 61, 315–322 (1992). https://doi.org/10.1007/BF02890433
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02890433