Abstract
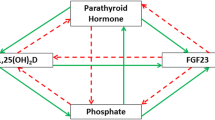
The heat-stable protein kinase inhibitor (PKI) protein is a specific and potent competitive inhibitor of the catalytic subunit of cAMP-dependent protein kinase (PKA). Previously, it has been shown that vitamin D status affects chick kidney PKI activity: a 5- to 10-fold increase in PKI activity was observed in kidneys of chronically vitamin D-deficient chicks and treatment with 1,25-dihydroxyvitamin D3 (1,25[OH]2D3) in cultured kidney cells resulted in a 95% decrease in PKI activity. The authors have recently cloned the cDNA for chick kidney PKI and have used the coding sequence to study the regulation of PKI mRNA. Northern analysis showed the expression of two PKI messages, which are 2.7 and 3.3 kb in size. These mRNAs are expressed in brain, muscle, testis, and kidney, but not in pancreas, liver, or intestine. PKI mRNA steady-state levels are downregulated by 47% in kidneys from vitamin D-replete chicks as compared to vitamin D-deficient chicks. PKI mRNA levels in brain, muscle, and testis are not affected by vitamin D status. Treatment of primary chick kidney cultures treated with 10−7 M, 1,25(OH)2D3 for 24 h resulted in a 20–30% decrease in PKI mRNA. 1,25(OH)2D3 treatment does not affect the stability of PKI mRNA as determined by treatment of cell cultures with actinomycin D. This study shows that 1,25(OH)2D3 directly and tissue-specifically downregulates PKI mRNA in the chick kidney.
Similar content being viewed by others
References
Posner, J. B., Stern, R., and Krebs, E. G. (1965).J. Biol. Chem. 240, 982–985.
Ashby, C. D. and Walsh, D. A. (1972).J. Biol. Chem. 247, 6637–6642.
Wen, W., Harootunian, A. T., Adams, S. R., Feramisco, J., Tsien, R. Y., Meinkoth, J. L., and Taylor, S. S. (1994).J. Biol. Chem. 269, 32,214–32,220.
Wen, W., Meinkoth, J. L., Tsien, R. Y., and Taylor, S. S. (1995).Cell 82, 463–473.
Szmigielski, A., Guidotti, A., and Costa, E. (1977).J. Biol. Chem. 252, 3848–3853.
Walsh, D. A., Ashby, C. D., Gonzalez, C., Calkins, D., Fischer, E. H., and Krebs, E. G. (1971).J. Biol. Chem. 246, 1977–1985.
Skala, J. P., Drummond, G. I., and Hahn, P. (1974).Biochem. J. 138, 195–199.
Henry, H. L., Al-Abdaly, F. A., and Noland, T. A. (1983).Comp. Biochem. Physiol. 74B, 715–718.
Ashby, C. D. and Walsh, D. A. (1973).J. Biol. Chem. 248, 1255–1261.
Walsh, D. A. and Ashby, C. D. (1973).Recent. Prod. Hormone Res. 29, 329–359.
Kuo, J. F. (1975).Biochem. Biophys. Res. Commun. 65, 1214–1220.
Costa, M. (1977).Biochem. Biophys. Res. Commun. 78, 1311–1218
Tash, J. S., Welsh, M. J., and Means, A. R. (1981).Endocrinology 108, 427–434.
Rudack-Garcia, D. and Henry, H. L. (1981).J. Biol. Chem. 256, 10,781–10,785.
Al-Abdaly, F. A. and Henry, H. L. (1989).Endocrinology 124, 2901–2906.
Henry, H. L., Cunningham, N. C., and Noland, T. A. (1983).Endocrinology 113, 1942–1949.
Harrison, H. C., Harrison, H. E., and Park E. A. (1958).Am. J. Physiol. 192, 432–436.
Marchetto, G. S. and Henry, H. L. (1995).Gene 158, 303,304.
Van Patten, S. M., Howard, P., Walsh, D. A., and Maurer, R. A. (1992).Mol. Endo. 6, 2114–2122.
Walters, M. R. (1992).Endocr. Rev. 13, 719–764.
Okazaki, T., Igarashi, T., and Kronenberg, H. M. (1988).J. Biol. Chem. 263 2203–2208.
Naveh-Many, T. and Silver, J. (1988).J. Clin. Invest. 81, 270–273.
Lichtler, A., Stover, M. L., Angilly, J., Kream, B., and Rowe, D. W. (1989).J. Biol. Chem. 264, 3072–3077.
Simpson, R. U., Hsu, T., Begley, D. A., Mitchell, B. S., and Alizadeh, B. N. (1987).J. Biol. Chem. 262, 4104–4108.
Norman, A. W., Midgett, R. J., Myrtle, J. F., and Nowicki, H. G. (1971).Biochem. Biophys. Res. Commun. 42, 1082–1087.
Sanger, F., Nicklen, S., and Coulson, A. R. (1977).Proc. Natl. Acad. Sci. USA 74, 5463–5467.
Chomczynski, P. and Sacchi, N. (1987).Anal. Biochem. 162, 156–159.
Puissant, C. and Houdebine, L. M. (1990).Biotechniques 8, 148,149.
Lehrach, H., Diamond, D., Wozney, J. M., and Boedtker, H. (1977).Biochemistry 16, 4743–4751.
Henry, H. L. (1979).J. Biol. Chem. 254, 2722–2729.
Chou, S.-Y., Hannah, S., Lowe, K. E., Norman, A. W., and Henry, H. L. (1995).Endocrinology 136, 5520–5526.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Marchetto, G.S., Henry, H.L. Tissue-specific expression and regulation by 1,25(OH)2D3 of chick protein kinase inhibitor (PKI) mRNA. Endocr 6, 5–10 (1997). https://doi.org/10.1007/BF02738795
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02738795