Abstract
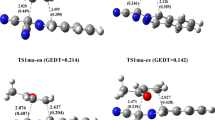
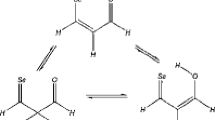
The semi-empirical PM3 SCF-MO method is used to investigate the Wagner-Meerwein migration of various groups during the pinacol-pinacolone rearrangement of some acyclic systems. Pinacol first protonates and dehydrates to form a carbocation that undergoes a 1,2-migration to form a protonated ketone, which then deprotonates to yield the pinacolone product. We study the Wagner-Meerwein migration of hydride, methyl, ethyl, isopropyl,t- butyl, phenyl and heterocylic 2-, 3-and 4-pyridyl groups in various acyclic 1,2-diol (pinacol) systems as they rearrange to pinacolones. This 1,2-migration involves a three-centred moiety in the cationic transition state. The migratory aptitude predicted here follows the order: hydride >t-butyl > isopropyl > ethyl > methyl > phenyl, which accords well with available experimental data and/or chemical intuition, reflecting also on the ability of the group involved to carry positive charge in the transition state. The structure of the migrating group (whether aliphatic or aromatic) within the transition state also supports the stabilising role of delocalisation of positive charge for reaction feasibility. Geometrical and thermodynamic considerations coincide in assigning the following order to relative “earliness” of the transition state along the reaction pathway:t-butyl > isopropyl > phenyl > methyl > 2-pyridyl > 4-pyridyl
Similar content being viewed by others
References
Olah G A and Schleyer P v R 1969Carbonium ions (New York, London, Sydney, Toronto: Wiley-Interscience) vol. 2; Olah G A and Schleyer P v R 1971Carbonium ions (New York, London, Sydney, Toronto: Wiley-Interscience) vol. 3; Creary X 1991Chem. Rev. 91 1625
Vogel P 1985Organic chemistry: Studies in organic chemistry 21 (New York: Elsevier Science) ch. 10; Saunders M and Jiminez-Vazquez H A 1991Chem. Rev. 91 375
Pine S H 1987Organic chemistry 5th edn (Singapore: McGraw-Hill) p. 973
Sykes P 1986A guide book to mechanism in organic chemistry 6th edn (New Delhi: Orient Longman) p. 1ll
Fittig W 1859Annalen 110 23; Fittig W 1860Annalen 114 54
Collins CJ 1960Q. Rev. 375
Cecchi P, Cipollini R, Pizzabiocca A, Renzi G and Speranza M 1988Tetrahedron 44 4847
Suzuki K, Ohkuma T, Miyazawa M and Tsichihashi G 1986Tetrahedron Lett. 27 373
Suzuki K, Katayama E and Tsuchihashi G 1984Tetrahedron Lett. 25 1817
Suzuki K, Katayamma E and Tsuchihashi G 1983Tetrahedron Lett. 24 4997
Stiles M and Meyer R P 1959J. Am. Chem. Soc. 81 1499
Shono T, Fujita K, Kumai S, Watanabe T and Nishiguchi I 1972Tetrahedron Lett. 31 3249
Pilkington J W and Waring A J 1973Tetrahedron Lett. 44 4345
Tsuchihashi G, Tomooka K and Suzuki K 1984Tetrahedron Lett. 25 4253
Shine H J 1967Aromatic rearrangements (Amsterdam, London, New York: Elsevier) ch. 1
Collins C J 1955J. Am. Chem. Soc. 77 5517
Carlin R B and Shivaramakrishnan K P 1970J. Org. Chem. 35 2368
Mayo P D 1980Rearrangement in ground and excited states (New York: Academic Press) p. 35
Hammond G S 1955J. Am. Chem. Soc. 77 334
Stewart J J P 1989J. Comput. Chem. 10 209
Stewart J JP 1983QCPEBull. 3 43
Shano D F J 1985Opt. Theory Appl. 46 87; Stewart J J P 1990 Frank J Seiler Research Laboratory, United States Air Force Academy, CO 80840
Maquestiau A, Flammang R, Flammang-Barbieux M and Mispreuve H 1980Tetrahedron 36 1994
Young-Sook P H 1988Bull. Korean Chem. Soc. 10 151
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Pachuau, Z., Lyngdoh, R.H.D. Molecular orbital studies on the Wagner-Meerwein migration in some acyclic pinacol—pinacolone rearrangements. J Chem Sci 116, 83–91 (2004). https://doi.org/10.1007/BF02708200
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF02708200