Abstract
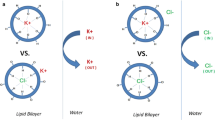
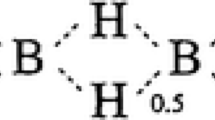
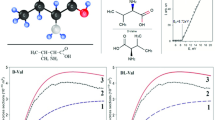
The electrostatic potential of valinomycin in various conformations as obtained by the crystal structures (uncomplexed, complexed) and theoretical considerations have been evaluated and compared. The potential energy profiles along the æ axis of the bracelet-like structures show a systematic variation from the uncomplexed to the complexed structure. This type of conformational change and the potential variation are probably associated with different states of ion transport, like the capture and release of ions by the ionophore. Also, the asymmetry of the molecule due to D-HyIV on one side and L-Lac on the other side is reflected in the potential values along the Z-axis, the magnitude of which, is considerable in the uncomplexed structure. The evaluation of the potential at the ab-initio level on smaller fragments indicate that the order of liganding capacity of oxygen is amide > ether > ester. Also, the inductive effects due to alkyl substitution is negligible as evidenced by the potential studies on the substituted amides and esters.
Similar content being viewed by others
References
Blow, D. M. (1960)Acta. Crystalogr.,13, 168.
Catalan, J. and Yanez, M. (1980)Tetrahedron,36, 665.
Devarajan, S., Nair, C. M. K., Easwaran, K. R. K. and Vijayan, M. (1980)Nature (London),286, 640.
Devarajan, S. and Easwaran, K. R. K. and Vijayan, M. (1984)Int. J. Peptide Protein Res.,23, 324.
Etchebest, C., Lavery, R. and Pullman, A. (1982)Stud. Biophys.,90, 7.
Hamilton, J. A., Sabesan, M. N. and Steinrauf, L. K. (1981)J. Am. Chem. Soc.,103, 5880.
Hayes, D. M. and Kollman, P. A. (1976)J. Am. Chem. Soc.,98, 3335.
Hehre, W. J., Stewart, R. F. and Pople, J. A. (1969)J. Chem. Phys.,51, 2657.
Karle, I. L. (1975)J. Am. Chem. Soc.,97, 4379.
Mayers, D. F. and Urry, D. W. (1972)J. Am. Chem. Soc.,94, 77.
Neupert-Laves, K. and Dobbler, M. (1975)Helv. Chim. Acta.,58, 432.
Ovchinnikov, Yu. A. and Ivanov, V. T. (1975)Tetrahedron,31, 2177.
Ovichinnikov, Yu. A., Ivanov, V. T., and Shkrob, A. M. (1974)Membrane active complexones, (Amsterdam: Elsevier).
Perricaudet, M. and Pullman, A. (1973)FEBS Lett.,34, 222.
Scrocco, E. and Tomasi, J. (1973)Top. Curr. Chem.,42, 95.
Smith, G. D., Dnax, W. L., Langs, D. A., De Titta, G. T., Edmonds, J. W., Rohrer, D. C. and Weeks, C. M. (1975)J. Am. Chem. Soc.,97, 7242.
Srebrenik, S., Weinstein, H. and Pauncz, (1973)Chem. Phys. Lett.,20, 419.
Steinrauf, L. K., Hamilton, J. A. and Sabesan, M. N. (1982)J. Am. Chem. Soc.,104, 4085.
Sundaram, K. and Tyagi, K. S. (1978)Int. J. Quantum Chem.,13, 17.
Szabo, G., Eisenman, G., Laprade, R., Ciani, S. M. and Krasne, S. (1973) inMembranes (ed. George Eisenman) (New York: Marcel Dekker Inc) p. 291.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Sreerama, N., Vishveshwara, S. Studies involving the electrostatic potential of valinomycin. J. Biosci. 8, 315–327 (1985). https://doi.org/10.1007/BF02703985
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF02703985