Abstract
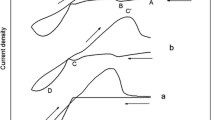
The corrosion behavior of sputter-deposited Al, Al-Cu, and Al-Si alloy thin films in dilute hydrofluoric (HF) acid solution was investigated. These materials maintain a thin aluminum oxide film in dilute HF solutions and, consequently, are susceptible to localized corrosion. Pit densities increase for the alloys with Cu and, to a lesser extent, Si additions. Open circuit potentials (OCP) are more positive for such alloys relative to the OCP of pure Al. Metastable pits in Al-Cu alloys are formed in Cu-depleted zones at grain boundaries which are galvanically coupled to adjacent θ-Al2Cu precipitates. Metastable pits in Al-Si alloys are formed in the Al matrix which is galvanically coupled to adjacent elemental Si nodules. θ-Al2Cu has different electrochemical characteristics than Al, even though both maintain a thin Al oxide in dilute HF solutions. θ-Al2Cu has a more positive OCP than pure Al and facilitates cathodic reactions at enhanced rates relative to pure Al. Hence, its presence raises the potential of the adjacent pure Al grain boundary to potentials which increase the probability of metastable pitting. Evidence is also presented which suggests that metastable pit growth may be cathode limited. A new hypothesis describing one mechanism by which θ-Al2Cu supports cathodic electron transfer re-actions is discussed.
Similar content being viewed by others
References
S. Thomas and H.M. Berg:IEEE Transactions on Components,Hybrides, and Manufacturing Technology (CHMT-10), 1987, pp. 252–57.
S. Mayumi, I. Murozono, H. Nanatsue, and S. Ueda:J. Electrochem. Soc, 1990, vol. 137 (6), pp. 1861–67.
J. Zahavi, M. Rotel, H.C. Huang, and P. Totta:International Corrosion Congress, Toronto, ON, Canada, 1984, pp. 311–16.
J.R. Scully, R.P. Frankenthal, K.J. Hanson, D.J. Siconolfi, and J.D. Sinclair:J. Electrochem., 1990, vol. 137 (5), pp. 1365–73.
J.L. Murray and A.J. McAlister:Bull. Alloy Phase Diagrams, 1984, vol. 5 (1), pp. 74–84.
J. Fleming: Microelectronics Device Development Laboratories, Sandia National Laboratories, July 1990, private communication.
J.O. McCaldin and H. Sankur:Appl. Phys. Lett., 1971, vol. 19 (12), pp. 524–27.
A.P. Bond, G.F. Boiling, H.A. Domian, and H. Biloni:J. Electrochem., 1966, vol. 113, p. 773.
H. Bohni and H.H. Uhlig:J. Electrochem., 1969, vol. 116, p. 906.
M. Pourbaix:Atlas of Electrochemical Equilibria in Aqueous Solutions, NACE, Houston, TX, 1974, pp. 168–76.
J.W. Schultze: inPassivity of Metals, R.P. Frankenthal and J. Kruger, eds., The Electrochemical Soc, Inc., Princeton, NJ, 1978, p. 82.
J.R. Galvele and S.M. de De Micheli:Corros. Sci., 1970, vol. 10, p. 795.
R. Holliger and H. Bohni: inComputer Aided Acquisition and Analysis of Corrosion Data, M.W. Kendig, U. Bertocci, and J.E. Strutt, eds., The Electrochemical Society Proceedings Series, Pennington, NJ, 1985, vol. 85–3, p. 200.
A.K. Covington and J.M. Thain:J. Chem. Ed., 1972, vol. 49 (8), p. 554.
M. Salomen and B.K. Stevenson:J. Chem. Eng. Data, 1974, vol. 19 (1), p. 42.
H.N. Farrer and F.J.C. Rossotti:J. Inorg. Nucl. Chem., 1965, vol. 26, p. 1959.
W.J. Hamer and Y.-C. Wu:J. Research Nat. Bureau Standards-A. Physics and Chemistry, 1970, vol. 74A (6), p. 761.
ASTM Standard G-3, ASTM Annual Book of Standards, ASTM, Philadelphia, PA, 1990, vol. 03.02, p. 60.
H.P. Hack, P.J. Moran, and J.R. Scully:The Measurement and Correction of Electrolyte Resistance in Electrochemical Tests, L.L. Scribner and S.R. Taylor, eds., ASTM STP 1056, ASTM, Philadelphia, PA, 1989, pp. 5–26.
S.J. Ketcham and F.H. Haynie:Corrosion, 1963, vol. 19, pp. 242t-246t.
J.W. Evancho and J.T. Staley:Metall. Trans., 1974, vol. 5, pp. 43–47.
M.J. Carr and A.D. Romig, Jr.:Proc. 44th EMSA Meeting, San Francisco Press, San Francisco, CA, 1988, pp. 916–17.
A.D. Romig, Jr.: Sandia Report SAND82-2938, Sandia National Laboratories, Albuquerque, NM, Mar. 1983.
A.D. Romig, Jr.:Characterization of Interfacial Chemistry by Analytical Electron Microscopy, Defect and Diffusion Forum, Trans Tech Publications, Aedermanns-dorf, Switzerland, 1988, vol. 59, pp. 179–96.
A.D. Romig, Jr., S.J. Plimpton, R.L. Mykelbust, and D.E. Newbury:Microbeam Analysis—1990, San Francisco Press, San Francisco, CA, 1991, pp. 275–80.
Kratos XSAM 800 commercial software package, Kratos Limited Analytical Instruments, Manchester, United Kingdom.
D.R. Frear, J.E. Sanchez, A.D. Romig, Jr., and J.W. Morris, Jr.:Metall. Trans. A, 1990, vol. 21A, pp. 2449–58.
H.B. Aaron and H.I. Aaronson:Acta Metall., 1968, vol. 16, p. 789.
J. Kruger and K. Rhyne:Nucl. Chem. Waste Management, 1982, vol. 3, p. 205.
Y. Murty, T. Kattamis, and O. Devereux:Corrosion, June 1975, vol. 31 (6),
B. Mazurkiewicz and A. Piotrowski:Corros. Sci., 1983, vol. 23 (7), pp. 697–707.
R.H. Brown, W.L. Fink, and M.S. Hunter:Trans. AIME, 1941, vol. 143, p. 115.
ASM Metals Handbook on Corrosion, ASM, Metals Park, OH, 1987, vol. 13, p. 584.
J.R. Scully: inCritical Factors in Localized Corrosion, G.S. Frankel and R.C. Newman, eds., The Electrochemical Society, Pennington, NJ, 1992, vol. 92–9, pp. 144–56.
K.J. Vetter and H.H. Streblow:Localized Corrosion, NACE-3,, R.W. Staehle, B.F. Brown, J. Kruger, and A. Agrawal, eds., NACE, Houston, TX, 1974, pp. 240–60.
G.S. Frankel:Corros. Sci., 1990, vol. 30 (12), pp. 1203–18.
G.S. Frankel, L. Stockert, F. Hunkeler, and H. Bohni:Corros. J., 1987, vol. 43 (7), pp. 429–36.
D.E. Peebles and J.R. Scully: Sandia National Laboratories, Albuquerque, NM, unpublished research, 1990.
G.E. McGuire, G.K. Schweitzer, and T.A. Carlson:Inorg. Chem., 1973, vol. 12, p. 2450.
P.E. Larson:J. Electron Spectroscopy Rel. Phenom., 1974, vol. 4, p. 213.
N.S. McIntyre and M.G. Cook:Anal. Chem., 1975, vol. 47, p. 2208.
L.C. Feldman and J.W. Mayer:Fundamentals of Surface and Thin Film Analysis, North-Holland, NY, 1986, p. 125.
J.A. Richardson and G.C. Wood:J. Electrochem. Soc, 1973, vol. 120, p. 193.
F. Mansfeld and H. Shih:J. Electrochem. Soc, 1988, vol. 135, p. 1171.
J.R. Scully: “Characterization of the Corrosion of Al Thin Films Using Electrochemical Impedance Methods,” ASTM STP1188 on EIS-Analysis and Interpretation, J.R. Scully, D. Silverman, and M. Kendig, eds., ASTM, Philadelphia, PA, in press.
Introduction to Ceramics: W.D. Kingery, H.K. Bowen, and D.R. Uhlmann, eds., John Wiley and Son, New York, NY, 1976, p. 903.
G.D. Davis, W.C. Moshier, J.S. Ahearn, H.F. Hough, and G.O. Gote:J. Vac. Sci. Technol. A, 1987, vol. 5 (4), p. 1152.
H. Hladky and J.L. Dawson:Corros. Sci., 1981, vol. 21 (4), pp. 317–22.
H. Hladky and J.L. Dawson:Corros. Sci., 1982, vol. 22 (3), pp. 231–37.
J. Painot and J. Augustynski:Electrochim. Acta, 1975, vol. 20, p. 747.
F. Hunkeler and H. Bohni:Corrosion, 1981, vol. 37, p. 645.
J.W. Schultz and L. Elfenthal:J. Electroanal. Chem., 1986, vol. 204, pp. 153–71.
K. Shimamura, K. Miura, A. Kawashima, K. Asami, and K. Hashimoto:Proc. Symp. on Corrosion and Electrochemical Catalysis on Metallic Glasses, Proc. Electrochem. Soc, The Electrochemical Society, Pennington, NJ, 1988, vol. 88–1, p. 232.
T.N. Taylor and J.A. Martin:J. Vac. Sci. Tech., 1991, vol. A9, pp. 1840–46.
H.H. Strehblow and C.J. Doherty:J. Electrochem. Soc, 1978, vol. 125 (1), pp. 30–33.
W.D. Mackintosh, F. Brown, and H.H. Plattner:J. Electrochem. Soc, 1974, vol. 121 (10), pp. 1281–86.
H.H. Streblow, C.M. Melliar-Smith, and W.M. Augustyniak:J. Electrochem. Soc, 1978, vol. 125 (6), pp. 915–19.
G.C. Farlow, P.S. Sklad, C.W. White, and C.J. McHargue:J. Mater. Res., 1990, vol. 5 (7), p. 1502.
B. Abies, P. Sheng, M.D. Courts, and Y. Arie:Adv. Phys., 1975, vol. 24 (3), p. 407.
J.W. Schultze and U. Stimming:Z. Phys. Chem., 1985, vol. 98, p. 285.
J.W. Schultze and M.M. Haga:Z. Phys. Chem., 1977, vol. 104, p. 73.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Scully, J.R., Peebles, D.E., Romig, A.D. et al. Metallurgical factors influencing the corrosion of aluminum, Al-Cu, and Al-Si alloy thin films in dilute hydrofluoric solution. Metall Trans A 23, 2641–2655 (1992). https://doi.org/10.1007/BF02658068
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF02658068