Abstract
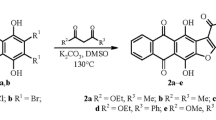
I-Acetylenyl-9,10-anthraquinones react with an excess of NH2NH2 at 80–115°C to give a mixture of substituted 7H-dibenzo[de,h]quinolin-7-ones and anthra[9,1-cd]-1,2-diazepin-8-ones. The latter compounds undergo reductive contraction of the sevenmembered ring to give the corresponding 7H-dibenzo[de,h]quinolin-7-ones. Bulky substituents in position 2 of the initial acetylenylanthraquinones prevent the formation of the sevenmembered heterocycle. A scheme of the cyclocondensation was proposed.
Similar content being viewed by others
References
I. D. Ivanchikova, R. N. Myasnikova, and M. S. Shvartsberg,Izv. Akad. Nauk, Ser. Khim., 1998, 2031 [Russ. Chem. Bull., 1998,47, 1975 (Engl. Transl.)].
M. S. Shvartsberg, I. D. Ivanchikova, and S. F. Vasilevsky,Tetrahedron Lett., 1994,35, 2077.
Weigand-Hilgetag,Praktische organische Chemie, [Practical Organic Chemistry], Springer, Leipzig 1965.
A. V. Piskunov, A. A. Moroz, and M. S. Shvartsberg,Izv. Akad. Nauk SSSR, Ser. Khim., 1987, 828 [Bull. Acad. Sci. USSR, Div. Chem. Sci., 1987,36, 755 (Engl. Transl.)].
A. A. Moroz, A. V. Piskunov, and M. S. Shvartsberg,Izv. Akad. Nauk SSSR, Ser. Khim., 1981, 386 [Bull. Acad. Sci. USSR, Div. Chem. Sci., 1981,30 (Engl. Transl.)].
I. D. Ivanchikova, A. A. Moroz, and M. S. Shvartsberg,Izv. Akad. Nauk SSSR, Ser. Khim, 1991, 1447 [Bull. Acad. Sci. USSR, Div. Chem. Sci., 1991,40, 1286 (Engl. Transl.)].
A. A. Moroz, D. Sc. (Chem.) Thesis, Novosibirsk, 1988, 332 pp.
Author information
Authors and Affiliations
Additional information
Translated fromIzvestiya Akademii Nauk. Seriya Khimicheskaya, No. 10, pp. 2027–2030, October, 1998.
Rights and permissions
About this article
Cite this article
Shvartsberg, M.S., Ivanchikova, I.D. & Vasilevsky, S.F. A novel heterocyclization of 1-acetylenyl-9,10-anthraquinones. Russ Chem Bull 47, 1971–1974 (1998). https://doi.org/10.1007/BF02494508
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF02494508