Abstract
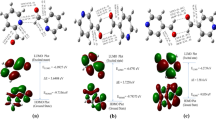
The absorption bands in the UV spectra of isomeric nitropyrazoles were assigned by the calculations in the semiempirical AMI (CI) approximation. The long-wave absorption of nitropyrazoles is caused by π→π* and η0→π* transitions. The charge-transfer band is the most intense. The π→π* transitions undergo a considerable bathochromic shift in the deprotonation. The first ionization potential (PI) of the 4-nitropyrazole anion was estimated from the empirical dependence of the energy of the excited π-state on PI of alkyl-substituted 4-nitropyrazoles. The PI of the 4-nitropyrazole anion is 3 eV lower than that of a neutral molecule. This is evidence for a substantial destabilization of the boundary β-orbital in the heterolytic cleavage of the N−H bond. The analysis of the UV and NMR spectra of 3(5)-nitropyrazole confirms the viewpoint that the 3-nitro tautomer predominates in solution.
Similar content being viewed by others
References
C. L. Habraken, P. C. M. Van Woerom, H. W. De Wind, and C. G. M. Kallenberg,Rec. Trav. Chim., 1966,85, 1191.
C. L. Habraken, C. I. M. Beenakker, and J. Brussee,J. Heterocycl. Chem., 1972,9, 939.
J. W. A. Janssen, C. G. Kruse, H. J. Koeners, and C. L. Habraken,J. Heterocycl. Chem., 1973,10, 1055.
D. Dumanevic, J. Ciric, A. Muk, and V. Nicolic,Talanta, 1975,22, 819.
M. A. Andreeva, M. I. Bolotov, Sh. G. Isaev, R. Ya. Mushii, V. P. Perevalov, V. I. Seraya, and B. I. Stepanov,Zh. Obshch. Khim., 1980,50, 2116 [J. Gen. Chem. USSR, 1980,50 (Engl. Transl.)].
V. P. Perevalov, L. I. Baryshnenkova, G. P. Sennikov, I. B. Savina, Sh. G. Isaev, M. A. Andreeva, and B. I. Stepanov,Khim. Geterotsikl. Soedin., 1986, 1629 [Chem. Heterocycl. Compd., 1985 (Engl. Transl.)].
V. P. Perevalov, V. V. Redchenko, G. V. Shelban, A. B. Kostitsin, L. I. Baryshnenkova, and B. I. Stepanov,Khim. Geterotsikl. Soedin., 1986, 1629 [Chem. Heterocycl. Compd., 1986 (Engl. Transl.)].
V. K. Turchaninov, D. K. Danovich, A. F. Ermikov, V. G. Zakzhevskii, and L. A. Es'kova,Izv. Akad. Nauk SSSR, Ser. Khim., 1991, 2503 [Bull. Acad. Sci USSR, Div. Chem. Sci., 1991,40, 2177 (Engl. Transl.)].
J. Del Bene and H. H. Jaffe,J. Chem. Phys., 1986,48 4050.
J. L. Finar,J. Chem. Soc. (B), 1968, 725.
J. W. A. M. Janssen, H. J. Koeners, C. G. Kruse, and C. L. Habraken,J. Org. Chem., 1973,38, 1777.
R. Huttel and F. Buchele,Chem. Ber., 1995,88, 1586.
L. Knorr,Lieb. Ann. Chem., 1984,279, 234.
O. V. Sverdlova,Elektronnye spektry v organicheskoi khimii [Electronic States in Organic Chemistry], Khimiya, Leningrad, 1985, 84 (in Russian).
M. R. Grimmett, K. H. R. Lim, and R. T. Weavers,Aust. J. Chem., 1979,32, 2203.
A. Fruchier, V. Pellergrin, R. M. Claramunt, and J. Elguero,Org. Magn. Reson., 1984,22, 473.
P. Cabildo, R. M. Claramunt, and J. Elguero,Org. Magn. Reson., 1984,22, 603.
R. M. Claramunt, J. Elguero, C. Marzin, and J. Seita,An. Quim., 1979,75, 701.
M. Begtrup, G. Boyer, P. Cabildo, C. Cativiela, R. M. Claramunt, J. Elguero, J I. Garcia, C. Toiron, and P. Vedso,Magn. Reson. Chem., 1993,31, 107.
C. Lopez, R. M. Claramunt, S. Trofimenko, and J. Elguero,Can. J. Chem., 1993,71, 678.
M. T. Chenon, C. Coupry, D. M. Grant, and R. J. Pugmire,J. Org. Chem., 1977,42, 659.
H. Dorn,J. Prakt. Chem., 1973,315, 382.
Author information
Authors and Affiliations
Additional information
Translated fromIzvestiya Akademii Nauk. Seriya Khimicheskaya. No. 2, pp. 310–314, February, 1997.
Rights and permissions
About this article
Cite this article
Vokin, A.I., Komarova, T.N., Larina, L.I. et al. Investigation of UV spectra of isomeric nitropyrazoles by the semiempirical AM1 (CI) method. Russ Chem Bull 46, 297–301 (1997). https://doi.org/10.1007/BF02494366
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF02494366