Abstract
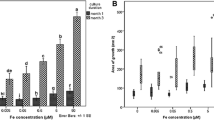
Transmembrane ferricyanide reduction in whole cells of normal and of transformed tobacco (Nicotiana tabacum) callus tissue was compared. It was found that low concentrations of indoleacetic acid (IAA, 0.1 μM), gibberellic acid (GA, 0.3 μM), and benzyl adenine (BA, 0.03 μM) stimulate external ferricyanide reduction in normal tobacco callus cells, but inhibit this reaction up to 67% in transformed cells when hormones are applied to cells 10 min prior to assay. Higher concentrations of these growth regulators (1 μM or greater) inhibit transmembrane ferricyanide reduction in both types of cells, with the exception of IAA, giving an initial stimulation of the rate (12%), followed by 24% inhibition after 2 min. The observed external ferricyanide reduction by whole tobacco callus cells may be explained on the basis of a transplasmalemma redox system, which may be associated with the iron metabolism of these cells.
Similar content being viewed by others
References
Bienfait HF, Duivenvoorden J. Verkerke W (1982) Ferric reduction by roots of chlorotic bean plants: Indications for an enzymatic process. J Plant Nutr 5:451–456
Chaney RL, Brown JC, Tiffin JC (1972) Obligatory reduction of ferric chelates in iron uptake by soybeans. Plant Physiol 50:208–213
Clark MG, Partick EJ, Crane FL (1982) Effects of catecholamines on the transplasma membrane redox system in rat liver. Biochem Internat 5:711–717
Clark MG, Partick EJ, Patten GS, Crane FL, Löw H, Grebing C (1981) Evidence for the extra-cellular reduction of ferricyanide by rat liver. Biochem J 200:565–572
Craig TA, Crane FL (1981) Evidence for a transplasma membrane electron transport system in plant cells. Proc Ind Acad Sci 90:150–155
Craig TA, Crane FL (1982) Hormonal control of a transplasma membrane electron transport system in plant cells. Proc Ind Acad Sci 91:150–154
Crane FL, Roberts H, Linnane AW, Löw H (1982) Transmembrane ferricyanide reduction by cells of the yeastSaccharomyces cerevisiae. J Bioenerget Biomembr 14:191–205
Löw H, Crane FL, Grebing C, Hall K, Tally M (1980) Metabolic milieu and insulin action. In: Waldhäusl WK (ed) Diabetes 1979. Excerpta Medica, Amsterdam, pp 209–213
MacKellar WC, Crane FL, Morré DJ, Ramasarma T, Löw H, Grebing C (1979) Orientation of NADH dehydrogenase activity in plasma membranes. J Cell Biol 83:286a
Mishra RK, Passow H (1969) Induction of intracellular ATP synthesis by extracellular ferricyanide in human red blood cells. J Membr Biol 1:214–224
Murashige T, Skoog F (1962) A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiol Plant 15:473–497
Orringer EP, Roer MES (1979) An ascorbate-mediated transmembrane-reducing system of the human erythrocyte. J Clin Invest 63:53–58
Polevoy VV, Salamatova TS (1977) Auxin, proton pump and cell trophies. In: Marré E, Ciferri O (eds) Regulation of cell membrane activities in plants. Elsevier/North-Holland Biomedical Press, Amsterdam, pp 209–215
Römheld V, Marschner H (1983) Mechanism of iron uptake by peanut plants. I. FeIII reduction, chelate splitting, and release of phenolics. Plant Physiol 71:949–954
Sijmons PC, Bienfait HF (1983) Source of electrons for extracellular Fe (III) reduction in iron-deficient bean roots. Physiol Plant (in press)
Thomashow MF, Nutter R, Montoya AL, Gordon MP, Nester EW (1980) Integration and organization of Ti plasmid sequences in crown gall tumors. Cell 19:729–739
Wang C-S, Alaupovic P (1978) Isolation and partial characterization of human erythrocyte membrane NADH:(acceptor) oxidoreductase. J Supramol Struct 9:1–14
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Barr, R., Crane, F.L. & Craig, T.A. Transmembrane ferricyanide reduction in tobacco callus cells. J Plant Growth Regul 2, 243–249 (1983). https://doi.org/10.1007/BF02042253
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF02042253