Summary
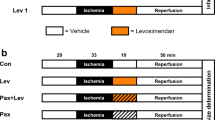
The direct myocardial protective effects of intracoronary infusions of cromakalim and pinacidil were determined in an anesthetized canine model of coronary occlusion and reperfusion. The left circumflex coronary artery was occluded for 90 minutes and reperfused for 5 hours, at which time the infarct size was determined. Cromakalim (0.1 μg/kg/min) or pinacidil (0.09 μg/kg/min) were infused into the left circumflex coronary artery starting 10 minutes preischemia. Cromakalim significantly reduced infarct size as a percent of the left ventricular area at risk (25±5%) compared with vehicle controls (55±7%). Pinacidil did not reduce infarct size at an equimolar dose, but at the higher dose also significantly reduced infarct size. Collateral blood flow was not significantly altered by either drug, though reperfusion flow was significantly higher in cromakalim-treated animals, particularly in the subepicardial region. When the same dose of cromakalim was given starting 2 minutes before the initiation of reperfusion, no significant beneficial effect of cromakalim was observed. In another study, isolated buffer-perfused rat hearts were subjected to 25 minutes of global ischemia and 30 minutes of reperfusion. These hearts were treated with 7 μM cromakalim, either starting 10 minutes before ischemia or only during reperfusion, and its effect on reperfusion function and LDH release were determined. Cromakalim pretreatment (both when given throughout the experiment and when not present in the reperfusion buffer) resulted in significant improvements in the reperfusion function. Reperfusion contracture and LDH were also significantly reduced with this treatment. When given only during reperfusion, cromakalim did not reduce the severity of ischemia when compared with vehicle controls. Thus, both cromakalim and pinacidil reduce ischemic/reperfusion injury, though the timing of treatment may be important.
Similar content being viewed by others
References
Weir SW, Weston AH. The effects of BRL 34915 and nicorandil on electrical and mechanical activity and on86Rb+ efflux in rat blood vessels.Br J Pharmacol 1986;88:121–128.
Bray KM, Newgreen DT, Small RC, et al. Evidence that the mechanism of the inhibitory action of pinacidil in rat and guinea pig smooth muscle differs from that of glyceryl trinitrate.Br J Pharmacol 1987;91:421–429.
Arena JP, Dass RS. Pinacidil, a putative K+-channel agonist, enhances K+-sensitive current in isolated guinea pig ventricular cells.Biophys J 1988;53:461.
Osterreider W. Modification of K+ conductance of heart cell membrane by BRL 34915.Naunyn Schmiedbergs Arch Pharmacol 1988;337:93–97.
Grover GJ, Sleph PG, Stranieri M, et al. Coronary vasodilator and cardiac functional effects of the potassium channel activator cromakalim in ischemic and nonischemic rat and guinea pig hearts.The Physiologist 1988;31:A150.
Flaim SF, Sleph PG, Stranieri M, et al. Effect of pinacidil on coronary flow and function in isolated perfused rat or guinea pig hearts during ischemic or nonischemic conditions.The Physiologist 1988;31:A149.
Grover GJ, McCullough JR, Henry DE, et al. Antiischemic effects of the potassium channel activators pinacidil and cromakalim and the reversal of these effects with the potassium channel blocker glyburide.J Pharmacol Exp Ther 1989;251:98–104.
Dole WP, Jackson DL, Rosenblatt JI, et al. Relative error and variability in blood flow measurements with radio-labelled microspheres.Am J Physiol 1982;243:H371-H378.
Watts JA, Maiorano L. Effects of diltiazem upon globally ischemic rat hearts.Eur J Pharmacol 1987;138:335–342.
Hamm CW, Opie LH. Protective effects of verapamil, nifedipine, and diltiazem in the coronary-ligated, isolated working rat heart.Circ Res 1983;52(Suppl 1):129–138.
van der Laarse A, Vereecke J, Carmeliet E. Ionic currents during hypoxia in voltage-clamped cat ventricular muscle.Circ Res 1980;47:501–508.
Buckingham RE, Clapham JC, Hamilton TC, et al. BRL 34915, a novel antihypertensive agent: Comparision of effects on blood pressure and other hemodynamic parameters with those of nifedipine in animal models.J Cardiovasc Pharmacol 1986;8:798–804.
Cook NS, Quast U, Hof RP, et al. Similarities in the mechanism of action of two new vasodilator drugs: Pinacidil and BRL 34915.J Cardiovasc Pharmacol 1988;11:90–99.
Fosset M, De Weille JR, Green RD, et al. Antidiabetic sulfonylureas control action potential properties in heart cells via high affinity receptors that are linked to ATP-dependent K+ channels.J Biol Chem 1988;263:7933–7936.
Schmid-Antomarchi H, De Weille J, Fosset M, et al. The receptor for antidiabetic sulfonylureas controls the activity of the ATP-modulated K+ channel in insulin-secreting cells.J Biol Chem 1987;262:15840–15844.
Imai N, Liang C, Stone CK, et al. Comparative effects of nitroprusside and pinacidil on myocardial blood flow and infarct size in awake dogs with acute myocardial infarction.Circulation 1988;7:705–711.
Reimer KA, Jennings RB, Cobb FR, et al. Animal models for protective ischemic myocardium: Results of the NHLBI cooperative study. Comparison of unconscious and conscious dog models.Circ Res 1985;56:651–665.
Gotanda K, Satoh K, Taira N. Is the cardiovascular profile of BRL 34915 characteristic of potassium channel activators?J Cardiovasc Pharmacol 1988;12:239–246.
Longman SD, Clapham JC, Wilson C, et al. Cromakalim, a potassium channel activator: A comparison of its cardiovascular haemodynamic profile and tissue specificity with those of pinacidil and nicorandil.J Cardiovasc Pharmacol 1988;12:535–542.
Steinberg MI, Ertel P, Smallwood J, et al. The relationship between vascular relaxant and cardiac electrophysiological effects of pinacidil.J Cardiovasc Pharmacol 1988;12(Suppl 2):S30-S40.
Angersbach D, Nicholson CD. Enhancement of muscle blood cell flux andpO2 by cromakalim (BRL 34915) and other compounds enhancing membrane K+ conductance, but not by Ca2+ antagonists or hydralazine, in an animal model of occlusive arterial disease.Naunyn Schmiedebergs Arch Pharmacol 1988;337:341–346.
Sanguinetti MC, Scott AL, Zingaro GJ, et al. BRL 34915 (cromakalim) activates ATP-sensitive K+ current in cardiac muscle.Proc Natl Acad Sci USA 1988;85:8360–8364.
Pieper GM, Gross GJ. Protective effect of nicorandil on postischemic function and tissue adenine nucleotides following a brief period of low-flow global ischemia in the isolated perfused rat heart.Pharmacology 1989;38:205–213.
Endo T, Nejima J, Kiuchi K, et al. Reduction of size of myocardial infarction with nicorandil, a new antianginal drug, after coronary artery occlusion in dogs.J Cardiovasc Pharmacol 1988;12:587–592.
Hiroaka M, Fan Z. Activation of ATP-sensitive outward K+ current by nicorandil (2-nicotinamidoethyl nitrate) in isolated ventricular myocytes.J Pharmacol Exp Ther 1988;250:278–285.
Grover GJ, Sleph PG, Parham CS. Effect of diltiazem on infarct size, reperfusion flow, and flow reserve: The effect of timing of treatment.J Pharmacol Exp Ther 1988;246:263–269.
Lo H, Kloner RA, Braunwald E. Effect of intracoronary verapamil on infarct size in the ischemic reperfused canine heart: Critical importance of the timing of treatment.Am J Cardiol 1985;56:672–677.
Grover GJ, Parham CS. Effect of intracoronary diltiazem on ST-segment elevation and myocardial blood flow during pacing-induced ischemia.J Cardiovasc Pharmacol 1987;10:548–554.
Cheung JH, Leaf A, Bonventre JV. Mechanism of protection by verapamil and nifedipine from anoxic injury in isolated cardiac myocytes.Am J Physiol 1984;246(Cell Physiol 15):C323–329.
Watts JA, Whipple JP, Hatley AA. A low concentration of nisoldipine reduces ischemic heart injury: Enhanced reflow and recovery of contractile function without energy preservation during ischemia.J Mol Cell Cardiol 1987;19:809–817.
Kleber AG, Janse MJ, van Capelle FJL, et al. Mechanism and time course of S-T and T-Q segment changes during acute regional myocardial ischemia in the pig heart determined by extracellular and intracellular recordings.Circ Res 1978;42:603–613.
Hill JL, Gettes LS. Effect of acute coronary artery occlusion on local extracellular K+ activity in swine.Circulation 1980;61:768–778.
Noma A, Shibasaki T. Intracellular ATP and cardiac membrane currents. In Narahashi T, ed.Ion channels, Vol 1. 1988;183–212.
Crompton M, Ellinger H, Costi A. Inhibition by cyclosporin A of a Ca2+ dependent probe in heart mitochondria activated by inorganic phosphate and oxidative stress.Biochem J 1988;255:357–360.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Grover, G.J., Dzwonczyk, S., Parham, C.S. et al. The protective effects of cromakalim and pinacidil on reperfusion function and infarct size in isolated perfused rat hearts and anesthetized dogs. Cardiovasc Drug Ther 4, 465–474 (1990). https://doi.org/10.1007/BF01857755
Issue Date:
DOI: https://doi.org/10.1007/BF01857755