Summary
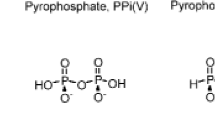
Two types of reactivities of thiophosphates have been demonstrated: one being nucleophilic displacement by the P-S moiety of nucleoside phosphorothioates and the other, phosphorylation via P-S cleavage as the driving force. We have designed a system where both displacement on carbon and P-S cleavage are possible. Adenosine derivatives have been synthesized with 5′-deoxy-5′-chloro and 5′-O-tosyl substitutions as leaving groups utilizing the 3′-O-phosphorothioate as the biphilic center. The main products of cyclization were 5′-O-tosyl and 5′-chloroadenosine 2′:3′-cyclic phosphate. Formation of 3′:5′-S-phosphorothioate was slow even using an excellent leaving group. This is possibly due to hydrogen bonding between the 2′-OH and the neighboring P-O.− KOH hydrolysis of the cyclic phosphorothioate yielded 2′(3′) phosphorothioates in a 1:1 ratio. The 2′ and 3′ isomers were separated and used to study the relative rates of cyclization. The cyclization via P-S cleavage of 2′(3′)-O-phosphorothioates showed that the 2′ isomer was more reactive. This is the first report of superior reactivity of the 3′-OH of a ribonucleoside.
Similar content being viewed by others
References
Bradbury, E., Nagyvary, J.: Nucleic Acids Res.3, 2437 (1976)
Chladek, S., Nagyvary, J. (1972). J. Amer. Chem. Soc.94, 2079
Cook, A.F. (1970). J. Amer. Chem. Soc.92, 190
Dunaway-Mariano, D. (1976). Tetrahedron32, 2991
Eckstein, F. (1970). J. Am. Chem. Soc.92, 4718
Gilman, A.G. (1970). Proc. Nat. Acad. Sci. USA67, 305
Iio, M., Kobayashi, Y., Eto, M., Yano, K., Omura, H., Eto, M. (1975). J. Fac. Agr., Kyushu Univ.19, 73
Kikugawa, K., Ichino, M. (1971). Tetrahedron Letts,87
Ridgway, S.T. (1974). Bull. Am. Astron. Soc.6, 376
Shuman, D.A., Miller, J.P., Scholten, M.B., Simon, L.M., Robins, R.K. (1973). Biochemistry12, 2781
Slabaugh, M.R., Harvey, A.J., Nagyvary, J. (1974). J. Mol. Evol.3, 317
Wieland, Th., Lambert, R. (1956). Chem. Ber.89, 2476
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kapovits, I., Nagyvary, J. Mechanistic possibilities in prebiotic thiophosphate chemistry. J Mol Evol 11, 25–33 (1978). https://doi.org/10.1007/BF01768022
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF01768022