Summary
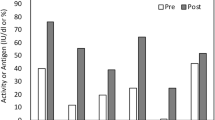
We investigated a new protein-C (PC) concentrate in a child with a type-II homozygous deficiency, concerning tolerance and safety. By means of various functional and antigen assays the in vivo recovery and the half-life were determined. In order to compare the results we reduced the measured values to the average halfrlife of 10.0 ± 0.5 h and to an optimal recovery of 96.6%. Considerable discrepancies observed in the response of functional (clotting) and both the amidolytic and antigen assays are characteristic for type II and anticoagulant treatment. The substituted protein C is activated by the endogenous system. Thus, the efficacy of activation can be determined in deficiency states. The antigen activity of one unit PC concentrate was found to be 120% (or 1.2 U/ml plasma), close to the 100% activity defined for endogenous PC.
Similar content being viewed by others
References
Barrett PN, Eibl J, Wöber G (1989) Virus inactivation of plasma proteins by vapour heating. In: Stoltz JF, Rivat C (eds) Biotechnologie des protéines du plasma. INSERM, Paris, pp 399–404
Bertina RM (1987) An international collaborative study on the performance of protein C antigen assays. Report of the ICTH Subcommittee on Protein C. Thromb Haemost 57: 112–117
Bertina RM, Broekmans AW, Lommenhock-van ES C, van Wijngaaden A (1984) The use of a functional and immunologic assay for plasma protein C in the study of the heterogeneity of congenital protein C deficiency. Thromb Haemost 51: 1–5
Elsinger F, Wöber G, Dorner F, Eibl J, Linnau Y, Philapitsch A, et al. (1986) Steam treatment of freeze-dried plasma O fractions. In: Ciavarella NL, Ruggeri ZM, Zimmerman TSO (eds) Factor Vil/Von Willebrand factor: biological and clinical advances. Wichtig, Milan, pp 297–302
Griffin JH, Bezeand A, Eratt B, Mosher D (1983) Functional and immunologic studies of protein C in thromboembolic disease. Blood 62: 301
Han P, Fung KP, Rahdakrishnan U (1991) Lack of agreement of chromogenic and clotting assays for factor X and protein C in Warfarinised plasma. Thromb Haemost 65: 360–363
Hubbard AR (1988) Standardization of protein C in plasma: establishment of an international standard. Thromb Haemost 59: 464–467
Mannucci PM, Boyer C, Triodi A, Viganò-D'Angelo S, Wolf M, Valsecchi C, D'Angelo A, Meyer D, Larrieu MJ (1987) Multicenter comparison of five functional and two immunological assays for protein C. Thromb Haemost 57: 44–48
Marlar RA, Montgomery RR, Broekmans AW (1989) Diagnosis and treatment of homozygous protein-C deficiency. J Pediatr 114: 528–534
Riess H, Binsack T, Hiller E (1985) Protein C antigen in prothrombin complex concentrates: content, recovery and half-life. Blut 50: 303–306
Schwarz HP, Schramm W, Dreyfus M (1990) Monoclonal antibody purified protein-C concentrate: initial clinical experience. In: Bruley DF, Drohan WN (eds) Protein C and related anticoagulants. Gulf, Houston, pp 83–89
Vinazzer H, Pangraz U (1987) Protein C: comparison of different assays in normal and abnormal plasma samples. Thromb Res 46: 1–8
Vogel G, Machulik M, Lauten G (1987) Thrombophilie infolge eines Protein C-Defektes vom Typ II. Z Ges Inn Med 42: 550–551
Zurborn KH, Broers H, Kirch W, Jäger N, Bruhn HD (1987) Die immunologische und funktioneile Protein C-Bestimmung bei verschiedenen internistischen Erkrankungen. Klin Wochenschr 65: 906–911
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Auberger, K. Evaluation of a new protein-C concentrate and comparison of protein-C assays in a child with congenital protein-C deficiency. Ann Hematol 64, 146–151 (1992). https://doi.org/10.1007/BF01697402
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01697402