Abstract
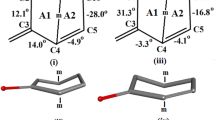
The structure of the title compound C26H38N2OH has been determined by direct methods from diffractometer data and refined by full-matrix least-squares. Crystals are monoclinicP21,a=6.237(1),b=10.295(2),c=18.485(2) Å, β=98.76(1)o,Z=2,D c=1.12 g cm−3,R=0.055 for 1059 observed reflections. The structure has two N-containing functional groups, one each in the pyrrolidine ring and carbonitrile groups, with interatomic separation of 11.527 Å. The 16-carbonitrile group is α-oriented. All steroid rings are trans-connected, A and C being in chair conformations and B and D half-charis. N(23) in the carbonitrile side-chair forms a weak tail-tail hydrogen-bond network by linking with the neighboring OH(20) hydroxy group at 1−x, 1/2+y, 1−z.
Similar content being viewed by others
References
Gandiha, A., Marshall, I. G., Paul, D., Roger, I. W., Scott, W., and Singh, H. (1975)Clin. Exp. Pharmacol. 2, 159–170.
Karaulov, S. (1973) SNOOPI Molecular Plotting Program: (Univ. of Wales, Cardiff).
Khoung Huu-Lainé, F., and Pinto-Scognamiglio, W. (1964)Arch. Int. Pharmacodyn. Ther. 147, 209–213.
Palmer, R. A., Kalam, M. A., Singh, H., and Paul, D. (1980)J. Cryst. Mol. Struct. 10, 31–53.
Quevauviller, A., and Lainé, F. (1960)Ann. Pharm. Fr. 18, 678–680.
Sheldrick, G. M. (1976) SHELX-76. Program for Crystal Structure Determination (Univ. of Cambridge, England).
Sheldrick, G. M. (1987) SHELX-S Program for Crystal Structure Solution (Univ. of Gottingen).
Singh, H., Chaudhary, A. K., Bhardwaj, T. R., and Paul, D. (1984)J. Sci. Ind. Res. 43, 306–315.
Singh, H. (1989) Personal Communication.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Chattopadhyay, T.K., Palmer, R.A. & Lisgarten, J.N. Crystal structure of 20ζ-hydroxy-3β-pyrrolidino-5-pregnene-16α-carbonitrile (HS1175): A novel design for neuromuscular blocking drugs. J Chem Crystallogr 24, 231–234 (1994). https://doi.org/10.1007/BF01672415
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF01672415