Summary
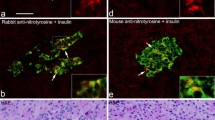
Immunoreactivity for islet amyloid polypeptide (IAPP) in the islets of Langerhans of non-insulin-dependent diabetic patients and non-diabetic patients of a non-selected post-mortem series was studied with a new polyclonal IAPP antibody. Out of 133 patients examined, 124 exhibited immunoreactivity for IAPP. Immunoreactivity was localized intra- and extracellularly and was limited to the islets of Langerhans. No extracellular immunoreactivity was observed in amyloid-negative cases. Co-localization of insulin and IAPP in the same islet-cells was verified by double staining with monoclonal insulin and polyclonal IAPP antibodies. Of 100 patients with non-insulin-dependent diabetes mellitus (NIDDM) and islet amyloid, 98 exhibited IAPP-positive deposits and 71 exhibited intracellular immunoreactivity. Evaluation of intracellular immunoreactivity and degree of islet amyloid deposition in cases of overt NIDDM revealed an inverse relationship, in that intracellular IAPP immunoreactivity were reduced in patients with developing islet amyloid deposition. Our data are consistent with the hypothesis of primaryβ-cell dysfunction leading to amyloid formation, with subsequent disturbance ofβ-cell homeostasis.
Similar content being viewed by others
References
Bell ET (1952) Hyalinization of the islets of Langerhans in diabetes mellitus. Diabetes 1:341–344
Berends MJH, Nieuwenhuis MG, Lips CJM, Blok APR, Ooms ECM, Veldhuizen RWA (1990) A comparative study between human B-cell insulin and islet amyloid polypeptide (IAPP) storage and amyloid deposition in type 2 diabetes mellitus and in non-diabetics. In: Natvig JB, Forre O, Husby G, Husebekk A, Skogen B, Sletten K, Westermark P (eds) Amyloid and amyloidosis 1990. Kluwer, Dordrecht, pp 457–461
Clark A, Wells CA, Buley ID, Cruickshank JK, Vanhegan RI, Matthews DR, Cooper GJS, Holman RR, Turner RC (1988) Islet amyloid, increased A-cells, reduced B-cells and exocrine fibrosis: quantitative changes in the pancreas in type 2 diabetes. Diabetes Res 9:151–159
Clark A, Saad MF, Nezzer T, Uren C, Knowler WC, Bennett PH, Turner RC (1990) Islet amyloid polypeptide in diabetic and non-diabetic Pima Indiens. Diabetologia 33:285–289
Cooper GJS, Willis AC, Clark A, Turner RC, Sim RB, Reid KBM (1987) Purification and characterization of a peptide from amyloid-rich pancreases of type 2 diabetic patients. Proc Natl Acad Sci USA 84:8628–8632
Cooper GJS, Day AJ, Willis AC, Roberts AN, Reid KBM, Leighton B (1989) Amylin and the amylin gene: structure, function and relationship to islet amyloid and to diabetes mellitus. Biochim Biophys Acta 1014:247–258
Ehrlich JC, Ratner IM (1961) Amyloidosis of the islets of Langerhans. Am J Pathol 38:49–59
Glenner GG, Eanes ED, Wiley CA (1988) Amyloid fibrils formed from a segment of the pancreatic islet amyloid protein. Biochem Biophys Res Commun 155:608–614
Johnson KH, O'Brien TD, Jordan K, Westermark P (1989a) Impaired glucose tolerance is associated with increased islet amyloid polypeptide (IAPP) immunoreactivity in pancreatic beta cells. Am J Pathol 135:245–250
Johnson KH, O'Brien TD, Betsholtz C, Westermark P (1989b) Islet amyloid, islet-amyloid polypeptide, and diabetes mellitus. N Engl J Med 321:513–518
Johnson KH, O'Brien TD, Westermark P (1991) Newly identified pancreatic protein islet amyloid polypeptide. Diabetes 40:310–314
Narita R, Toshimori H, Nakazato M, Kuribayashi T, Toshimori T, Kawabata K, Takahashi K, Masukura S (1992) Islet amyloid polypeptide (IAPP) and pancreatic islet amyloid deposition in diabetic and non-diabetic patients. Diabetes Res Clin Pract 15:3–14
Porte D, Kahn SE (1989) Hyperproinsulinemia and amyloid in NIDDM. Clues to etiology of isletβ-cell dysfunction? Diabetes 38:1333–1336
Puchtler H, Sweat F, Levine M (1962) On the binding of congo red by amyloid. J Histochem Cytochem 10:355–364
Schneider HM, Störkel S, Will W (1980) Das Amyloid der Langer-hansschen Inseln und seine Beziehung zum Diabetes mellitus. Dtsch Med Wochenschr 105:1143–1147
Scifert G (1959) Die pathologische Morphologie der Langerhansschen Inseln besonders bei Diabetes mellitus des Menschen. Verb Dtsch Ges Pathol 42:50–84
Steiner DF, Ohagi S, Nagamatsu S, Bell GI, Nishi M (1991) Isislet amyloid polypeptide a significant factor in pathogenesis or pathophysiology of diabetes? Diabetes 40:305–309
Toshimori H, Narita R, Nakazato M, Asai J, Mitsukawa T, Kangawa K, Matsuo H, Takahashi K, Matsukura S (1991) Islet amyloid polypeptide in insulinoma and in the islets of the pancreas of non-diabetic and diabetic subjects. Virchows Arch [A] 418:411–417
Westermark P (1973) Fine structure of islets of Langerhans in insular amyloidosis. Virchows Arch [A] 359:1–18
Westermark P, Wilander E (1978) The influence of amyloid deposits on the islet volume in maturity onset diabetes mellitus. Diabetologia 15:417–421
Westermark P, Wernstedt C, Wilander E, Hayden DW, O'Brien TD, Johnson KH (1987a) Amyloid fibrils in human insulinoma and islets of Langerhans of the diabetic cat are derived from a neuropeptide-like protein also present in normal islet cells. Proc Natl Acad Sci USA 84:3881–3885
Westermark P, Wilander E, Westermark GT, Johnson KH (1987b) Islet amyloid polypetide-like immunoreactivity in the islet B cells of type 2 (non-insulin-dependent) diabetic and non-diabetic individuals. Diabetologia 30:887–892
Wullbrandt A, Saeger W, Missmahl HP, Voigt C, Linke RP (1990) Amyloid in intervertebral discs of surgery and autopsy material. A new class of amyloid? Virchows Arch [A] 416:335–341
Author information
Authors and Affiliations
Additional information
Supported by the Johanna and Fritz Buch-Gedächtnisstiftung (Hamburg, FRG)
Rights and permissions
About this article
Cite this article
Röcken, C., Linke, R.P. & Saeger, W. Immunohistology of islet amyloid polypeptide in diabetes mellitus: Semi-qantitative studies in a post-mortem series. Vichows Archiv A Pathol Anat 421, 339–344 (1992). https://doi.org/10.1007/BF01660981
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01660981