Summary
The relationship between the phosphate potential (I) and the amount of phosphate (Q), added to the soil has been examined by equilibrating soil samples with 0.001M or 0.01M CaCl2 solutions containing various amounts of phosphate.
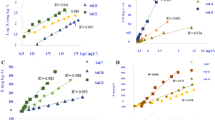
For one neutral and two alkaline soils the Q/I relationship depends on the CaCl2 concentration and the pH in such a way that the apparent values of I decrease when the CaCl2 concentration increases from 0.001 M to 0.01M. The difference between the two values increases when the pH increases. When correction is made for the formation of the soluble calcium phosphate complex, CaHPO4, the Q/I relationship becomes independent of the CaCl2 concentration. The initial phosphate potential (I0) determined by interpolation, is also found to be independent of the CaCl2 concentration.
The necessary amount of phosphate to be added or removed per gram of soil in order to obtain a certain alteration of the phosphate potential is designated the differential phosphate potential buffering capacity, DPBC. For ten soils DPBC-values are determined on the basis of the Q/I relationships, (ΔQ/ΔI)Io, and found to be independent of the CaCl2 concentration. The content of colloids and of inorganic phosphate accounts for a significant part of the variation in the DPBC for different soils. The importance of the DPBC for characterization of the phosphate status of soils in respect to phosphate supply to plants is briefly discussed.
Similar content being viewed by others
References
Aslyng, H. C., The lime and phosphoric acid potential of soils, their determination and practical application. Dissertation, London, pp. 178 (1950). (not published).
Aslyng, H. C., The lime and phosphate potentials of soils; the solubility and availability of phosphates. Yearb. Roy. Vet. Agr. Coll. Copenhagen, 1–50 (1954).
Aslyng, H. C., Phosphate potential and phosphate status of soils. Acta Agr. Scand.14, 261–285 (1964).
Bache, B. W., Aluminium and iron phosphate studies relating to soils. — I. Solution and hydrolysis of variscite and strengite. J. Soil Sci.14, 113–123 (1963).
Barrow, N. J., Relationship between uptake of phosphorus by plants and the phosphorus potential and buffering capacity of the soil — an attempt to test Schofields hypothesis. Soil Sci.104, 99–106 (1967).
Barrow, N. J., Ozanne, P. G. and Shaw, T. C., Nutrient potential and capacity. I. The concepts of nutrient potential and capacity and their application to soil potassium and phosphorus. Australian J. Agr. Research16, 61–76 (1965).
Beckett, P. H. T. and White, R. E., Studies on the phosphate potentials of soils. III. The pool of labile inorganic phosphate. Plant and Soil21, 253–282 (1964).
Bjerrum, N., Calciumorthophosphate. I. Die Festen Calciumorthophosphate. Kgl. Danske Vidensk. Selsk. Math. Fys. Medd.31, 1–68 (1958).
Bjerrum, N. und Unmack, A., Die Dissoziationskonstanten der Phosphorsäure. Kgl. Danske Vidensk. Selsk. Math. Fys. Medd.9, 115–152 (1929).
Clark, J. S. and Peech, M., Influence of neutral salts on the phosphate ion concentration in soil solution. Soil Sci. Soc. Am. Proc.24, 346–348 (1960).
Fordham, A. W., The measurement of chemical potential of phosphate in soil suspensions. Australian J. Soil Research1, 144–156 (1963).
Greenwald, I., Redish, J. and Kibrick, A. C., The dissociation of calcium and magnesium phosphates. J. Biol. Chem.135, 65–76 (1940).
Hsu, P. H., Adsorption of phosphate by aluminium and iron in soils. Soil Sci. Soc. Am. Proc.28, 474–478 (1964).
International Society of Soil Science, Soil Physics Terminology. Bulletin23, 7–11 (1963).
Lamm, C. G., Soil library. The Technical University of Denmark. Report1 (not yet published (1967)).
Larsen, S. and Court, M. N., The chemical potentials of phosphate ions in soil solutions. Trans. 7th Intern. Congr. Soil Sci.2, 413–421 (1960).
Larsen, S., The influence of calcium chloride concentration on the determination of lime and phosphate potentials of soil. J. Soil Sci.16, 275–278 (1965).
Larsen, S., Soil phosphorus. Advances in Agronomy19, 151–210 (1967).
Le Mare, P. H., Observations on the phosphate potential of some tropical soils. Trans. 7th Intern. Congr. Soil Sci.3, 600–603 (1960).
Lindsay, W. L. and Moreno, E. C., Phosphate phase equilibria in soils. Soil Sci. Soc. Am. Proc.24, 177–182 (1960).
Muljadi, D., Posner, A. M. and Quirk, J. P., The mechanism of phosphate adsorption by kaolinite, gibbsite and pseudoboehmite. J. Soil Sci.17, 212–247 (1966).
Ramamoorthy, B. and Subramanian, T. R., Phosphate potential and phosphate ion activity in soils with special reference to phosphate availability. Trans. 7th Intern. Congr. Soil Sci.3, 590–599 (1960).
Rinno, G., The informative value of nutrient potentials in respect to nutrient dynamics in soil. Albrecht-Thaer-Arch.10, 867–888 (1966).
Schofield, R. K., Thermodynamic potentials of soil constituents. Rothamsted Expt. Sta. Rept.29, (1949).
Schofield, R. K., Can a precise meaning be given to „available” soil phosphorus. Soils and Fertilizers18, 373–375 (1955).
Taylor, A. W. and Gurney, E. L., The effect of lime on the phosphate potential and resin extractable phosphate in five soils. Soil Sci. Soc. Am. Proc.29, 482–483 (1965).
White, R. E., Studies on the phosphate potentials of soils. II. Microbial effects. Plant and Soil20, 184–193 (1964).
White, R. E. and Beckett, P. H. T., Studies on the phosphate potentials of soils. I. The measurement of phosphate potential. Plant and Soil20, 1–16 (1964).
Author information
Authors and Affiliations
Additional information
The author is indebted to professor, Dr. H. C. Aslyng, head of the department for his suggestions and helpful criticism during the progress of this work.
Rights and permissions
About this article
Cite this article
Jensen, H.E. Phosphate potential and phosphate capacity of soils. Plant Soil 33, 17–29 (1970). https://doi.org/10.1007/BF01378193
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF01378193