Summary
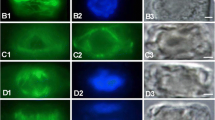
Tip cells of dark-grown protonemata of the mossCeratodon purpureus are negatively gravitropic (grow upward). They possess a unique longitudinal zonation: (1) a tip group of amylochloroplasts in the apical dome, (2) a plastid-free zone, (3) a zone of significant plastid sedimentation, and (4) a zone of mostly non-sedimenting plastids. Immunofluorescence of vertical cells showed microtubules distributed throughout the cytoplasm in a mostly axial orientation extending through all zones. Optical sectioning revealed a close spatial association between microtubules and plastids. A majority (two thirds) of protonemata gravistimulated for >20 min had a higher density of microtubules near the lower flank compared to the upper flank in the plastid-free zone. This apparent enrichment of microtubules occurred just proximal to sedimented plastids and near the part of the tip that presumably elongates more to produce curvature. Fewer than 5% of gravistimulated protonemata had an enrichment in microtubules near the upper flank, whereas 14% of vertical protonemata were enriched near one of the side walls. Oryzalin and amiprophos-methyl (APM) disrupted microtubules, gravitropism, and normal tip growth and zonation, but did not prevent plastid sedimentation. We hypothesize that a microtubule redistribution plays a role in gravitropism in this protonema. This appears to be the first report of an effect of gravity on microtubule distribution in plants.
Similar content being viewed by others
Abbreviations
- APM:
-
amiprophos-methyl
- DIC:
-
differential interference contrast
- DMSO:
-
dimethyl sulfoxide
- EGTA:
-
ethylene glycolbis-(β-amino-ethylether) N,N,N',N'-tetraacetic acid
- FITC:
-
fluorescein isothiocyanate
- GS:
-
gravitropic stimulus
- MT:
-
microtubule
- PIPES:
-
piperazine-N,N'-bis-2-ethanesulfonic acid
References
Banbury GH (1962) Geotropism of lower plants. In: Bünning E (ed) Physiologie der Bewegungen: Bewegungen durch Einflüsse der Temperatur, Schwerkraft, chemischer Faktoren und aus inneren Ursachen. Springer, Berlin Göttingen Heidelberg, pp 344–377 [Ruhland W etal (eds) Handbuch der Pflanzenphysiologie-Encyclopedia of plant physiology, vol 17, part 2]
Bartnik E, Sievers A (1988) In-vivo observations of a spherical aggregate of endoplasmic reticulum and of Golgi vesicles in the tip of fast-growingChara rhizoids. Planta 176: 1–9
Doonan JH, Cove DJ, Lloyd CW (1985) Immunofluorescence microscopy of microtubules in intact cell lineages of the moss,Physcomitrella patens. I. Normal and CIPC-treated cells. J Cell Sci 75: 131–147
— —, Corke FMK, Lloyd CW (1987) Pre-prophase band of microtubules, absent from tip-growing moss filaments, arises in leafy shoots during transition to intercalary growth. Cell Motil Cytoskeleton 7: 138–153
— —, Lloyd CW (1988) Microtubules and microfilaments in tip growth: evidence that microtubules impose polarity on protonemal growth inPhyscomitrella patens. J Cell Sci 89: 533–540
Hartmann E (1984) Influence of light on phototropic bending of moss protonemata ofCeratodon purpureus (Hedw.) Brid. J Hattori Bot Lab 55: 87–98
—, Klingenberg B, Bauer L (1983) Phytochrome-mediated phototropism in protonemata of the mossCeratodon purpureus Brid. Photochem Photobiol 38: 599–603
Jenkins GI, Courtice GRM, Cove DJ (1986) Gravitropic responses of wild-type and mutant strains of the mossPhyscomitrella patens. Plant Cell Environ 9: 637–644
Kiss JZ, Sack FD (1989) Reduced gravitropic sensitivity in roots of a starch-deficient mutant ofNicotiana sylvestris. Planta 180: 123–130
Morejohn LC, Bureau TE, Mole-Bajer J, Bajer AS, Fosket DE (1987) Oryzalin, a dinitroaniline herbicide, binds to plant tubulin and inhibits microtubule polymerization in vitro. Planta 172: 252–264
Sack FD, Kiss JZ (1989) Plastids and gravity perception. In: Boyer CD, Shannon JC, Hardison RC (eds) Physiology, biochemistry, and genetics of nongreen plastids. American Society of Plant Physiologists, Rockville, Maryland, pp 171–181
Schmiedel G, Schnepf E (1979) Side branch formation and orientation in the caulonema of the moss,Funaria hygrometrica: normal development and fine structure. Protoplasma 100: 367–383
— — (1980) Polarity and growth of caulonema tip cells of the mossFunaria hygrometrica. Planta 147: 405–413
Schnepf E (1986) Cellular polarity, Annu Rev Plant Physiol 37: 23–47
Tewinkel M, Volkmann D (1987) Observations on dividing plastids in the protonema of the mossFunaria hygrometrica Sibth. Planta 172: 309–320
Volkmann D, Sievers A (1979) Graviperception in multicellular organs. In: Haupt W, Feinleib ME (eds) Physiology of movements. Springer, Berlin Heidelberg New York, pp 573–600 [Pirson A, Zimmermann MH (eds) Encyclopedia of plant physiology, ns, vol 7]
Wacker I, Quader H, Schnepf E (1988) Influence of the herbicide Oryzalin on cytoskeleton and growth ofFunaria hygrometrica protonemata. Protoplasma 142: 55–67
Walker LM, Sack FD (1990) Amyloplasts as possible statoliths in gravitropic protonemata of the mossCeratodon purpureus. Planta 181: 71–77
White RG, Sack FD (1990) Actin microfilaments in presumptive statocytes of rootcaps and coleoptiles. Amer J Bot 77: 17–26
Young JC, Sack FD (1989) Videomicroscopy-based observation of gravitropism in individual protonema of the mossCeratodon. Am Soc Gravitat Space Biol Bull 2: 61
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Schwuchow, J., Sack, F.D. & Hartmann, E. Microtubule distribution in gravitropic protonemata of the mossCeratodon . Protoplasma 159, 60–69 (1990). https://doi.org/10.1007/BF01326635
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01326635