Abstract
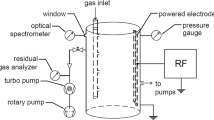
Processes which occur in microwave discharges of dilute mixtures of SF6 and O2 in He have been examined using a flow reactor sampled by a mass spectrometer. Two classes of experiments were performed. In the first set of experiments, mixtures containing 6×1011 cm−3 SF6, 6×1016 cm−3 He, and O2 in the range (0–3.6)×1013 cm−3 were passed through a 20-W 2450-MHz microwave discharge. The gas mixtures arriving at a sample point downstream from the discharge were examined for SF6, SF4, SOF2, SOF4, SO2F2, SO2, F, and O. In the second class of experiments, rate coefficients were measured for the reactions of SF4 with O and O2 and for the reaction of SF with O. The rate coefficient for the reaction of SF with O was found to be (4.2±1.5)×10−11 cm−3 s−1. SF4 was found to react so slowly with both oxygen atoms and oxygen molecules that only upper limits could be placed on the rate coefficients for these reactions. These values were 2×10−14 cm3 s−1 and 5×10−15 cm3 s−1 for reactions with O and O2 respectively. The observed distribution of products from the discharged mixtures is discussed in terms of the measured rate coefficients.
Similar content being viewed by others
References
D. L. Flamm,Solid State Technol. 22, 109 (1979).
M. J. Kushner,J. Appl. Phys. 53, 2923 (1982).
C. I. M. Beenakker, J. H. J. van Dommelen, and R. P. van de Poll,J. Appl. Phys. 52, 480 (1981).
D. L. Flamm,Plasma Chem. Plasma Process. 1, 37 (1981).
J. W. Coburn and H. F. Winters,J. Vac. Sci. Technol. 16, 391 (1979).
D. Edelson and D. L. Flamm,J. Appl. Phys. 56, 1522 (1984).
K. R. Ryan and I. C. Plumb,Plasma Chem. Plasma Process. 6, 231 (1986).
I. C. Plumb and K. R. Ryan,Plasma Chem. Plasma Process. 6, 205 (1986).
R. d'Agostino and D. L. Flamm,J. Appl. Phys.,52, 162 (1981).
G. Smolinsky and D. L. Elamm,J. Appl. Phys.,50, 4982 (1979).
I. C. Plumb and K. R. Ryan,Plasma Chem. Plasma Process. 6, 247 (1986).
M. C. Siddagangappa and R. J. Van Brunt,Proceedings of the 8th International Conference on Gas Discharge and Their Applications, Leeds University Press (1985), p. 247.
K. R. Ryan and I. C. Plumb,Plasma Chem. Plasma Process. 4, 271 (1984).
I. C. Plumb and K. R. Ryan,Plasma Chem. Plasma Process. 4, 271 (1984).
I. C. Plumb and K. R. Ryan,Int. J. Chem. Kinet. 13, 1011 (1981).
EPA/NIH Mass Spectral Data Base, S. R. Heller and G. W. A. Milne, eds., NSRDS-NBS 63, Suppl. 1 (1980).
J. Brunning,J. Chem. Soc. Faraday Trans. 2 81, 663 (1985).
D. C. Frost, S. T. Lee, C. A. McDowell, and P. C. Westwood,J. Electron Spectrosc. Relat. Phenom. 12, 95 (1977).
I. Sauers, H. W. Ellis, and L. G. Christophorou,IEEE Trans. Electr. Insul. EI-21, 111 (1986).
R. D. Levin and S. G. Lias, “Ionization Potential and Appearance Potential Measurements, 1971–1981” NSRDS-NBS 71 (1982).
M. W. Chase, J. L. Curnutt, R. A. McDonald, and A. N. Syverud,J. Phys. Chem. Ref. Data 7, 793 (1978).
M. W. Chase, J. L. Curnutt, J. R. Downey, R. A. McDonald, A. N. Syverud, and E. A. Valenzuela,J. Phys. Chem. Ref. Data 11, 695 (1982).
R. E. Winters, J. H. Collins, and W. F. Courchene,J. Chem. Phys. 45, 1931 (1966).
J. Brunning and M. A. A. Clyne,J. Chem. Soc. Faraday Trans. 2 80, 1001 (1984).
H. M. Anderson, J. A. Merson, and R. W. Light,IEEE Trans. Plasma Sci. PS 14, 156 (1986).
J. T. Herron,J. Phys. Chem. Ref. Data 16, 1 (1987).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Ryan, K.R., Plumb, I.C. Gas-phase reactions in plasmas of SF6 with O2 in He. Plasma Chem Plasma Process 8, 263–280 (1988). https://doi.org/10.1007/BF01020406
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF01020406