Abstract
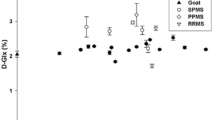
A TLC procedure which resolves two molecular species of ethanolamine plasmalogen, Pl-PE-1 and Pl-PE-2, was used to compare the ratio of these two species in myelin isolated from normal appearing white matter from brains of 17 multiple sclerosis (MS) patients, 17 normal (N) individuals, 1 patient with subacute sclerosing panencepha itis (SSPE) and 1 patient with a non-demyelinating neurological disease (OND). One of these species (Pl-PE-2) has been reported to be unique to myelin and has primarily 18∶1 in both the 1 and 2 positions of glycerol. The other species (Pl-PE-1) is also present in other membranes and has primarily a saturated chain in the 1 position and a polyunsaturated chain in the 2 position. The Pl-PE-1 to Pl-PE-2 ratio was quantitated by scanning the plates with a densitometer. The ratio was similar to normal in most of the MS samples, 0.88±0.09, but was much less than normal in 4 of the MS samples and the SSPE sample. This is attributed to increased decomposition or hydrolysis of Pl-PE-1 relative to Pl-PE-2 at some stage, either during the disease process or due to post-mortem decomposition. Although the reason for the enhanced decomposition of Pl-PE-1 is not known it suggests that Pl-PE-2 is more stable chemically. This may be related to the unique occurrence of Pl-PE-2 in myelin.
Similar content being viewed by others
References
Yanagihara, T., andCummings, J. N., 1969. Alterations of phospholipids, particularly plasmalogens, in the demyelination of multiple sclerosis as compared with that of cerebral edema. Brain 92:59–70.
Alling, C., Vanier, M-T., andSvennerholm, L., 1971. Lipid alterations in apparently normal white matter in multiple sclerosis. Brain Res. 35:325–336.
O'Brien, J. S., andSampson, E. L., 1965, Lipid composition of the normal human brain: grey matter, white matter, and myelin. J. Lip. Res. 6:537–544.
Sun, G. Y., andHorrocks, L. A., 1970. The acyl and alk-1-enyl groups of the major phosphoglycerides from ox brain myelin and mouse brain microsomal, mitochondrial and myelin fractions. Lipids 5:1006–1012.
Frosolono, M. F., and Marsh, M., 1973, Preparation and properties of phosphatidal ethanolamine from bovine white matter. Chem. Phys. Lipids 10:203–214.
Hack, M. H., andHelmy, F. M., 1977. Thin-layer chromatographic resolution of molecular species of ethenolamine plasmalogen quantitatively unique to myelin. J. Chromatogr. 135:229–234.
Hack, M. H., andHelmy, F. M., 1978. The diminution of the myelin ethanolamine plasmalogen in brain of the Jimpy mouse and brain and spinal cord of the Quaking mouse as visualized by thin-layer chromatography J. Chromatogr. 145:307–310.
Horrocks, L. A., 1972. Content, composition and metabolism of mammalian and avian lipids that contain ether groups, Pages 177–272,in Snyder, F. (ed.), Ether Lipids, Chemistry and Biology, Academic Press, N.Y.
Suzuki, K., Kamoshita, S., Eto, Y., Tourtellotte, W. W., andGonatas, J. O., 1973. Myelin in multiple sclerosis. Composition of myelin from normal-appearing white matter. Arch. Neurol. 28:293–297.
Fewster, M. E., Hirono, H., andMead, J. F., 1976. Lipid composition of myelin in multiple sclerosis. J. Neurol. 213:119–131.
Göpfert, E., Pytlik, S., andDebuch, H., 1980. 2′, 3′-cyclic nucleotide 3′-phosphohydrolase and lipids of myelin from multiple sclerosis and normal brains. J. Neurochem. 34:732–739.
Lowden, J. A., Moscarello, M. A., andMorecki, R., 1966. Isolation and characterization of an acid-soluble protein from myelin. Can. J. Biochem. 44:567–577.
Hack, M. H., andHelmy, F. M., 1975. Chromatographic analysis of the neutral and phosphatide glyceryl ethers from various biological sources. J. Chromatogr. 107:155–166.
Dittmer, J. C., andLester, R. L., 1964. A simple, specific spary for the detection of phospholipids on thin-layer chromatograms. J. Lipid Res. 5:126–127.
Touchstone, J. C., Chen, J. C., andBeaver, K. M., 1980. Improved separation of phospholipids in thin layer chromatography. Lipids 15:61–62.
Ansell, G. B., andSpanner, S., 1968. Plasmalogenase activity in normal and demyelinating tissue of the central nervous system. Biochem. J. 108:207–209.
Horrocks, L. A., Spanner, S., Mozzi, R., Fu, S. C., D'Amato, R. A., andKrakowka, S., 1978. Plasmalogenase is elevated in early demyelinating lesions. Adv. Exp. Med. Biol. 100:423–438.
Woelk, H., andPeiler-Ichikawa, K., 1974. Zur aktivitat der phospholipase A2 gegenuber verschiedenen 1-alk-1′-enyl-2-acyl-und 1-alkyl-2-acyl-verbindungen wahrend der multiplen sklerose. J. Neurol. 207:319–326.
Miller, S. L., Benjamins, J. A., andMorell, P., 1977. Metabolism of glycerophospholipids of myelin and microsomes in rat brain. J. Biol. Chem. 252:4025–4037.
Fishman, M. A., Madyastha, P., andPrensky, A. L., 1971. The effect of undernutrition on the development of myelin in the rat central nervous system. Lipids 6:458–465.
Svennerholm, L., Vanier, M-T., andJungbjer, B., 1978. Changes in fatty acid composition of human brain myelin lipids during maturation. J. Neurochem. 30:1383–1390.
Woelk, H., andBorri, P., 1973. Lipid and fatty acid composition of myelin purified from normal and MS brain. Eur. Neurol. 10:250–260.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Boggs, J.M., Stamp, D. & Moscarello, M.A. Comparison of two molecular species of ethanolamine plasmalogen in multiple sclerosis and normal myelin. Neurochem Res 7, 953–964 (1982). https://doi.org/10.1007/BF00965135
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00965135