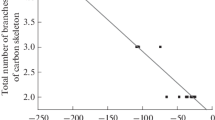
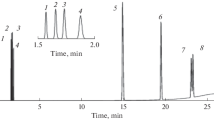
Conclusions
-
1.
A pulse microreactor was used in an admission system of a chromatomass spectrometer, and it was found that gas-phase hydrogenolysis (Pd/porous glass) of l-aryl-2-(2-furyl) cyclopropanes at 200–250°C proceeds with the formation of l-aryl-3-(2-furyl) propanes and 1(2)-aryl-2(l)-(2-furyl) propanes in 9:1 ratio.
-
2.
l-Aryl-3-(2-furyl) propanes undergo unusual fragmentation under electron impact to form stable cation radicals of alkylstyrenes, while fragmentation typical of alkylbenzenes and alkylfurans is less evident.
Similar content being viewed by others
Literature cited
A. I. Mikaya, L. P. Medvedkova, V. G. Zaikin, V. M. Vdovin, and A. A. Kamyshova, Izv. Akad. Nauk SSSR, Ser. Khim., 1181 (1983).
V. I. Smetanin, A. I. Mikaya, and V. G. Zaikin, Zavod. Lab.,49, No. 10, 24 (1983).
J. Newham, Chem. Revs.,63, 123 (1963).
Ya. R. Katsobashvili, M. V. Tsodikova, A. M. Krapivin, and M. V. Shishkina, Izv. Akad. Nauk SSSR, Ser. Khim., 2779 (1982).
Author information
Authors and Affiliations
Additional information
Translated from Izvestiya Akademii Nauk SSSR, Seriya Khimicheskaya, No. 11, pp. 2494–2497, November, 1986.
Rights and permissions
About this article
Cite this article
Mikaya, A.I., Zaikin, V.G. & Tsodikov, M.V. Reaction chromato-spectrometry in the series of 1-aryl-2-(2-furyl) cyclopropanes. Russ Chem Bull 35, 2282–2284 (1986). https://doi.org/10.1007/BF00953340
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00953340